Main findings
Escherichia coli was confirmed in 85 % of urinary isolates from children in Norway.
We found a significantly higher proportion of trimethoprim resistance and a significantly lower proportion of ciprofloxacin resistance in urinary isolates of E. coli taken from children compared to adults.
For E. coli in urine samples in children with a urinary tract infection, there was a low incidence of resistance to nitrofurantoin (0.5 %), mecillinam (4.0 %), cephalexin (4.3 %) and amoxicillin-clavulanic acid (7.2 %).
Urinary tract infections (UTIs) are common in children. Approximately 2 % of boys and 8 % of girls will have had a UTI before the age of eight (1). In paediatrics, there is a tradition of distinguishing between upper (febrile) and lower (afebrile) UTIs (1–3). The incidence is highest among infants, who mainly contract upper UTIs (1, 3, 4). A meta-analysis found that the incidence of upper UTIs among febrile children < 2 years was 7 % (4). In older children, the incidence of upper and lower UTIs is relatively similar (1). Most children can be treated with peroral antibiotics, but choosing the right antibiotic is crucial for preventing severe symptoms (3). However, unnecessary use of broad-spectrum antibiotics also needs to be avoided in order to curb the development of antibiotic resistance (5).
Escherichia coli is reported to cause at least 70 % of UTIs in children (1, 3). Globally, there is concern about the high rates of resistance to a number of antibiotics in E. coli urinary isolates from children (6–8). Norway has seen a slow but gradual increase in antibiotic resistance among E. coli in urine over the last 10–20 years (9).
Both the Norwegian Directorate of Health and the Norwegian Society of Pediatricians issue recommendations for the treatment of UTIs in children in Norway (2, 10), but these are partly based on microbiological resistance data that have been collated for all age groups (9). Reports from other countries show clear differences in resistance patterns in E. coli isolates from children compared with isolates from adults (11–13). One study showed that access to data specifically relating to resistance among E. coli in urine from children could potentially improve the quality of treatment for UTIs (13).
We wanted to strengthen the evidence base for choosing antibiotics to treat paediatric UTIs in Norway by using national data to describe the presence of bacteria and resistance patterns in urinary isolates.
Material and method
Data collection and bacterial isolates
This is a national observational study based on register data from the Norwegian Surveillance System for Antimicrobial Drug Resistance (NORM) covering the period 2013–2017. The register collects resistance data from bacterial isolates at all clinical microbiological laboratories in Norway (9). A urine sample must be provided if bacteria are detected in significant numbers in a patient with symptoms of a UTI. In urine aspiration sampling, ≥ 102 colony-forming units (CFU)/mL is regarded as significant, while a midstream sample or disposable catheter requires ≥ 103 CFU/mL. For mixed flora, ≥ 104 CFU/mL is required, while ≥ 105 CFU/mL is required from urine collection bags and permanent catheters. Only one isolate per patient is included during the collection period. Resistance data are interpreted according to clinical breakpoints set by the European Committee on Antimicrobial Susceptibility Testing (EUCAST), based on minimum inhibitory concentration (MIC) levels or zone diameter (14). In order to assess whether the bacterium is sensitive (S), sensitive to increased exposure (I) or resistant (R), each bacterium has a fixed limit for the breakpoint of different antibiotics. Increased exposure (I) means that a higher than standard dosage is needed for the bacterium to be sensitive.
We included all urine samples among the entire Norwegian population that met the NORM criteria, both from the primary and specialist health services, in defined periods in 2013–2017 (Table 1). In total, 13 211 urine samples were included, each representing one patient. Data from urine samples in children (0–17 years) were extracted from the NORM database and included the following: the patient's month of birth, sample date and antibiotic sensitivity with MIC levels/zone diameter. We divided the children into three age groups: infants (< 1 year, up to and including the month they turned one year old); preschool children (1–5 years, from the month following their first birthday up to and including the month of their sixth birthday); school children (6–17 years, from the month following their sixth birthday up to and including the month they turned 18). Urine samples from adults were separated from the year-specific NORM data by subtracting paediatric isolates from the total number of isolates presented in the report. This was done for each combination of bacteria and antibiotics, so that all paediatric isolates were eliminated.
Species
Table 1
Demographic overview of bacterial isolates from urine samples in children up to age 17 and adults, collected periodically from the Norwegian Surveillance System for Antimicrobial Drug Resistance (NORM) in the period 2013–2017. Spp. = (several unspecified species within a genus)
|
| Children aged 0–17 (crude rate/100 000 inhabitants1) | Adults ≥ 18 years (crude rate/100 000 inhabitants) |
|---|---|---|
| Total number of isolates2 | 589 (49) | 12 622 (311) |
| Age groups | ||
| < 1 year | 94 (158) | - |
| 1–5 years | 139 (45) | - |
| 6–17 years | 356 (43) | - |
| Bacteria | ||
| Escherichia coli | 403 (34) | 6 105 (150) |
| Klebsiella spp. | 101 (8) | 4 751 (117) |
| K. pneumoniae | 74 (6) | 2 811 (69) |
| K. oxytoca | 14 (1) | 1 227 (30) |
| Other Klebsiella spp. | 13 (1) | 713 (18) |
| Enterococcus spp. | 59 (5) | 1 201 (30) |
| E. faecalis | 59 (5) | 1 117 (27) |
| E. faecium | 0 (0) | 82 (2) |
| Other Enterococcus spp. | 0 (0) | 2 (0) |
| Proteus spp. | 22 (2) | 411 (10) |
| Enterobacter spp. | 4 (0) | 154 (4) |
1Number of inhabitants in the specific age groups in 2017, based on data from Statistics Norway.
2Number of days vary where data have been collected for NORM in the period 2013–17: E. coli 14 days (2013–17), Klebsiella spp. 105 days (2013–17), Enterococcus spp. 21 days (2015), Proteus spp. 21 days (2017), Enterobacter spp. 21 days (2016)
Cephalexin resistance is not reported to the NORM register, but cephalexin is recommended as an alternative antibiotic for upper UTIs in children (2). Since 2015, however, Haukeland University Hospital and the University Hospital of North Norway have routinely tested for cephalexin resistance in E. coli urinary isolates from children. We therefore chose to include resistance data for cephalexin in the period 2015–19 for samples taken from children aged 0–15 (from birth to 15 years) and analysed at the two hospitals. Only one isolate per child was included.
Data processing and statistics
We categorised antibiotic sensitivity as either S and I (sensitive isolate) or R (resistant isolate), according to the breakpoint defined by EUCAST that corresponded to the year of the NORM analysis.
Demographic data were presented as absolute numbers and as the crude rate per 100 000 inhabitants based on population data from Statistics Norway (15). The crude rates were calculated for adults and for children in different age groups. The population was based on the total number of inhabitants for the respective age groups in 2017. The length of the data collection period varied for the different bacteria. In order to estimate the relative distribution of the different bacteria, we therefore weighted the number of days that data were collected for each microbe.
Resistance data were presented as percentages with a corresponding 95 % confidence interval (95 % CI) based on the standard deviations of the percentages. All isolates from children were analysed for sensitivity to clinically relevant antibiotics. Bacteria that included ≥ 50 isolates were compared with adults as a percentage of resistant isolates. The resistance rate for Enterococcus faecalis was calculated separately, while for Klebsiella species (spp.), we included all the species in the comparison.
We used the chi-squared test to compare percentages and Fisher's exact test in cases where the expected frequency was ≤ 5. Stata SE version 16.1 was used for the analysis. A p-value < 0.05 was considered significant.
Ethics
The data were collected in accordance with the Resistance Register Regulations, and the study was approved by the Data Protection Officer at Haukeland University Hospital (ID 1075).
Results
We included a total of 589 urinary isolates from children. These accounted for 4.5 % of all isolates reported to the NORM register in 2013–17. A comparison to the background population showed approximately three times as many isolates from infants as from older children, and compared with adults, we found more E. coli and less Klebsiella spp. in children (Table 1). We found the following distribution of bacterial species among children: E. coli 85.2 % (95 % CI 84.0–86.3), Enterococcus spp. 8.3 % (7.4–9.3), Proteus spp. 3.1 % (2.6–3.7), Klebsiella spp. 2.8 % (2.3–3.4) and Enterobacter spp. 0.6 % (95 % CI 0.3–0.9). Figure 1 shows the distribution by æ.
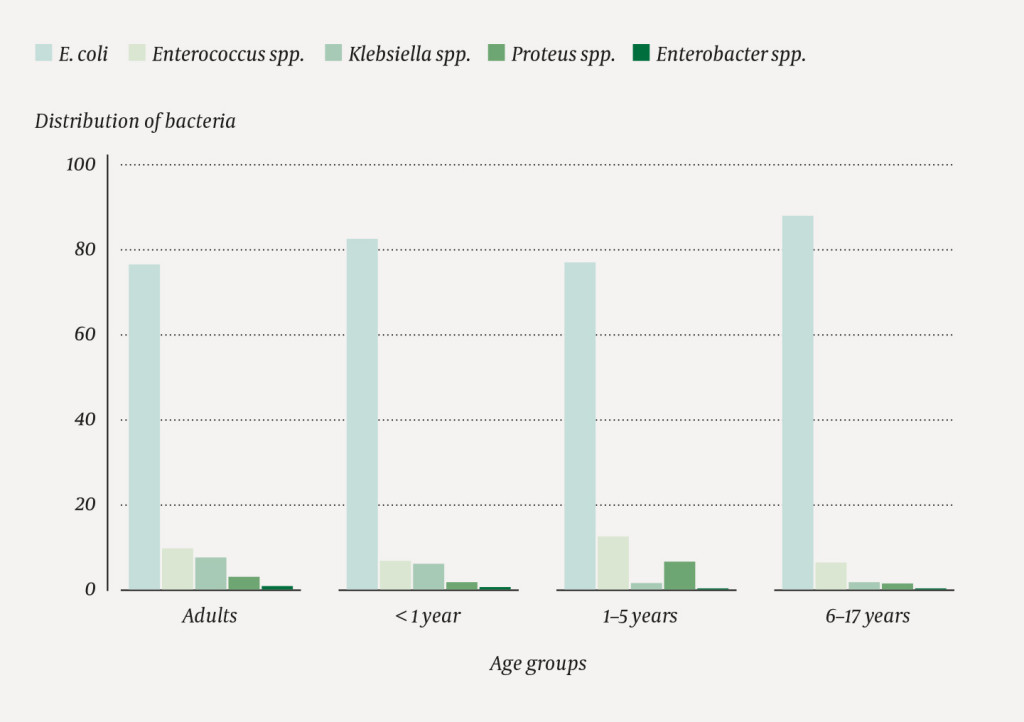
Table 2 shows resistance rates in isolates from children compared with isolates from adults. Among the 16 E. coli urinary isolates from children that were resistant to mecillinam, 12 (75.0 %) were also resistant to amoxicillin-clavulanic acid. Of the 22 Proteus spp. collected from children, all were sensitive to gentamicin and amoxicillin-clavulanic acid, while one isolate was resistant to mecillinam. Of four Enterobacter spp. collected from children, all were sensitive to gentamicin, cefotaxime and trimethoprim-sulfamethoxazole, while three (75.0 %) were resistant to amoxicillin-clavulanic acid.
Table 2
Resistance rates in urinary isolates from children up to and including 17 years of age compared with adults, based on data from the Norwegian Surveillance System for Antimicrobial Drug Resistance (NORM) in the period 2013–17.
| Bacteria |
| Age 0–17 years | Age ≥ 18 years |
| Difference, % | ||||
|---|---|---|---|---|---|---|---|---|---|
| No. | Per cent (95 % CI) | No. | Per cent (95 % CI) | ||||||
| Escherichia coli | 403 | − | 6 105 | − | − | ||||
| Ampicillin | 142 | 35.2 (30.6–40.1) | 2 139 | 35.0 (33.8–36.2) | 0.2 | ||||
| Mecillinam | 16 | 4.0 (2.3–6.4) | 351 | 5.7 (5.2–6.4) | 1.7 | ||||
| Amoxicillin-clavulanic acid | 29 | 7.2 (4.9–10.2) | 441 | 7.2 (6.6–7.9) | 0.0 | ||||
| Meropenem | 0 | 0.0 (0.0–0.9) | 0 | 0.0 (0.0–0.1) | 0.0 | ||||
| Gentamicin | 11 | 2.7 (1.4–4.8) | 241 | 3.9 (3.5–4.5) | 1.2 | ||||
| Ciprofloxacin | 23 | 5.7 (3.7–8.4) | 533 | 8.7 (8.0–9.5) | 3.01 | ||||
| Nitrofurantoin | 2 | 0.5 (0.1–1.8) | 81 | 1.3 (1.1–1.6) | 0.8 | ||||
| Trimethoprim | 109 | 27.0 (22.8–31.7) | 1 396 | 22.9 (21.8–23.9) | 4.11 | ||||
| Trimethoprim-sulfamethoxazole | 97 | 24.1 (20.0–28.5) | 1 264 | 20.7 (19.7–21.7) | 3.4 | ||||
| Extended spectrum beta-lactamases (positive = resistant)2 | 8 | 2.0 (0.9–3.9) | 190 | 3.1 (2.7–3.6) | 1.1 | ||||
| Cephalexin (N = 2 064)3 | 88 | 4.3 (3.4–5.2) | − | − | − | ||||
| Klebsiella spp. | 101 | − | 4 751 | − | − | ||||
| Mecillinam | 5 | 5.0 (1.6–11.2) | 479 | 10.1 (9.2–11.0) | 5.1 | ||||
| Amoxicillin-clavulanic acid | 12 | 11.9 (6.3–19.8) | 344 | 7.2 (6.5–8.0) | 4.7 | ||||
| Meropenem | 0 | 0.0 (0.0–3.6) | 2 | 0.0 (0.0–0.2) | 0.0 | ||||
| Gentamicin | 2 | 2.0 (0.2–7.0) | 89 | 1.9 (1.5–2.3) | 0.1 | ||||
| Ciprofloxacin | 2 | 2.0 (0.2–7.0) | 256 | 5.4 (4.8–6.1) | 3.4 | ||||
| Trimethoprim-sulfamethoxazole | 14 | 13.9 (7.8–22.2) | 592 | 12.5 (11.5–13.4) | 1.4 | ||||
| Extended spectrum beta-lactamases (positive = resistant) | 4 | 4.0 (1.1–9.8) | 142 | 3.0 (2.5–3.5) | 1.0 | ||||
| Enterococcus faecalis | 59 | − | 1 117 | − | − | ||||
| Ampicillin | 0 | 0.0 (0.0–6.1) | 0 | 0.0 (0.0–0.3) | 0.0 | ||||
| Gentamicin (high-level resistance) | 5 | 8.5 (2.8–18.7) | 160 | 14.3 (12.3–16.5) | 5.8 | ||||
| Vancomycin (screening) | 0 | 0.0 (0.0–6.1) | 0 | 0.0 (0.0–0.3) | 0.0 | ||||
1P-value < 0.05. For ciprofloxacin: 0.03. For trimethoprim: 0.02. Chi-squared test was used.
2Bacteria that produce extended spectrum beta-lactamases will generally also be resistant to third-generation cephalosporins such as cefotaxime.
3Based on 2064 urinary isolates from children up to and including 15-year-olds at the University Hospital of North Norway and Haukeland University Hospital in the period 2015–19.
Discussion
We have described national epidemiology and resistance rates in paediatric UTIs in Norway and used corresponding data from adults as a reference. E. coli was the predominant microbe in urine samples from children, with low resistance rates (< 10 %) for nitrofurantoin, mecillinam, cephalexin and amoxicillin-clavulanic acid.
The higher proportion of urinary isolates from infants compared to older children reflects the higher incidence of upper UTIs in this age group (1). The predominance of E. coli in urinary isolates from children is well known (1, 3), but a high incidence of other bacteria is also reported internationally (16). The higher proportion of Enterococcus spp. and Proteus spp. among preschool children in our study has also been observed in the United States (8).
Use of ciprofloxacin in children in Norway is very low (17, 18), but overuse among adults is viewed as problematic (19). Lower ciprofloxacin resistance rates in E. coli in children compared to adults have also been observed internationally (11–13) and are likely to be linked to the lower usage in children (9).
We found greater resistance of E. coli to both trimethoprim and trimethoprim-sulfamethoxazole in children compared to adults, which also corresponds with the findings in a US study (13). In the Norwegian Directorate of Health's guidelines for antibiotic use, trimethoprim and trimethoprim-sulfamethoxazole are among the empirical first choices for children with lower and upper UTIs respectively (10). Consumer statistics from the Norwegian Prescription Database show that trimethoprim and trimethoprim-sulfamethoxazole have been frequently used among children ≤ 5 years in recent years (17). We can assume that a significant proportion of the prescriptions were for treatment of a UTI. We can also assume that a significant proportion were prescribed empirically on the basis of the guidelines. In general, however, empirical antibiotics are not recommended for the treatment of UTIs if the E. coli resistance rate exceeds 20 % (6).
Our data support current recommendations that pivmecillinam and amoxicillin-clavulanic acid should be among the first choices for empirical treatment of upper UTIs in children (2, 10). We found lower resistance rates among both E. coli and Klebsiella spp. to pivmecillinam than to amoxicillin-clavulanic acid. Furthermore, E. faecalis is sensitive to amoxicillin-clavulanic acid, and has inherent resistance to pivmecillinam. Despite the lack of good clinical evidence for the use of pivmecillinam in upper UTIs, it is our view that the available literature, low resistance rates, favourable ecological profile and good clinical experience provide an adequate foundation for recommending this in Norway (20–22). Pivmecillinam is not available as an oral solution. The tablets can be crushed and mixed with food such as jam etc., but crushed tablets have an unpleasant taste. Permission was granted to market amoxicillin-clavulanic acid as an oral solution in Norway in 2019, and this is well established as a treatment for upper UTIs in children (23). However, there are concerns about high resistance rates in several countries (6, 7), and increasing use could also lead to a further development of resistance in Norway (24).
At Haukeland University Hospital and the University Hospital of North Norway, we found low cephalexin resistance rates in E. coli urinary isolates from children. These are not national data, but they nevertheless provide a good indication of the likely level of resistance nationally. Cephalexin is available as an oral solution and is recommended in other high-income countries as the first choice for upper UTIs in children (25, 26). A large observational study showed good clinical efficacy in UTIs in children (27). However, consideration should also be given to the possible selection of extended spectrum beta-lactamases in cases of excessive use (28). Overall, it is our view that cephalexin should be considered on an equal footing with amoxicillin-clavulanic acid in the empirical guidelines for upper UTIs, also to avoid excessive use of amoxicillin-clavulanic acid.
Nitrofurantoin is not for use in upper UTIs due to poor tissue penetration, but is a good first choice for lower UTIs. In 2009, a Norwegian literature review called for approval of nitrofurantoin in drop form (22), but it is still only available as an unpleasant-tasting water-soluble tablet.
Intravenous gentamicin and ampicillin are recommended for clinically weak patients, and there should be a liberal threshold for their use in children younger than 3–6 months (2). The gentamicin resistance rate was < 3 % for all bacteria included except enterococci. This confirms that gentamicin and ampicillin represent a potent empirical regimen for Norwegian children.
One of the strengths of this study is that using national data from a reliable register gave us a basis for assessing the level of resistance in children in relation to the current guidelines for choosing antibiotics in the treatment of UTIs in children in Norway.
The lack of clinical data is a weakness of the study. Randomised controlled clinical trials investigating the efficacy of relevant peroral antibiotics for the treatment of upper UTIs would be useful. Different data registration periods meant that we had to estimate the distribution of the microbes, but we consider this to be sufficiently precise for our purposes. Some UTIs are likely to have been treated without a urine culture sample. Our data are therefore probably not representative of all clinical UTIs during the registration periods.
Some of the urine samples in the NORM register are likely to be collection bag samples. This increases the risk of contamination, but the register's strict inclusion criteria mean that the isolates in our material are most likely uropathogenic. Resistance data for cephalexin are only available from two hospitals, but data on cephalexin and E. coli in urine samples will be included in future NORM reporting.
Conclusion
Our description of resistance data from urinary isolates from children in Norway strengthens the knowledge base for recommendations for empirical treatment of UTIs among this group. In the oral treatment of upper UTIs, pivmecillinam and amoxicillin-clavulanic acid should still be among the first choices, but consideration should also be given to whether cephalexin should be included as an equivalent first choice. Pivmecillinam and nitrofurantoin should be the first choice for lower UTIs. Trimethoprim-sulfamethoxazole and trimethoprim should preferably only be used following resistance determination.
The authors would like to thank Ellen Blakstad, Department of Quality and Research, Northern Norway Regional Health Authority for retrieving data from the University Hospital of North Norway on E. coli and sensitivity to cephalexin.
The article has been peer reviewed.
- 1.
Tullus K, Shaikh N. Urinary tract infections in children. Lancet 2020; 395: 1659–68. [PubMed][CrossRef]
- 2.
Norsk barnelegeforening. Akuttveileder i pediatri. https://www.helsebiblioteket.no/pediatriveiledere/ Accessed 1.11.2020.
- 3.
Kaufman J, Temple-Smith M, Sanci L. Urinary tract infections in children: an overview of diagnosis and management. BMJ Paediatr Open 2019; 3: e000487. [PubMed][CrossRef]
- 4.
Shaikh N, Morone NE, Bost JE et al. Prevalence of urinary tract infection in childhood: a meta-analysis. Pediatr Infect Dis J 2008; 27: 302–8. [PubMed][CrossRef]
- 5.
Vernaz N, Huttner B, Muscionico D et al. Modelling the impact of antibiotic use on antibiotic-resistant Escherichia coli using population-based data from a large hospital and its surrounding community. J Antimicrob Chemother 2011; 66: 928–35. [PubMed][CrossRef]
- 6.
Bryce A, Costelloe C, Wootton M et al. Comparison of risk factors for, and prevalence of, antibiotic resistance in contaminating and pathogenic urinary Escherichia coli in children in primary care: prospective cohort study. J Antimicrob Chemother 2018; 73: 1359–67. [PubMed][CrossRef]
- 7.
Bryce A, Hay AD, Lane IF et al. Global prevalence of antibiotic resistance in paediatric urinary tract infections caused by Escherichia coli and association with routine use of antibiotics in primary care: systematic review and meta-analysis. BMJ 2016; 352: i939. [PubMed][CrossRef]
- 8.
Edlin RS, Shapiro DJ, Hersh AL et al. Antibiotic resistance patterns of outpatient pediatric urinary tract infections. J Urol 2013; 190: 222–7. [PubMed][CrossRef]
- 9.
Helsedirektoratet. Nasjonal faglig retningslinje for antibiotikabruk i primærhelsetjenesten. http://www.antibiotikaiallmennpraksis.no/ Accessed 15.10.2020.
- 10.
NORM/NORM-VET 2013–2019. Usage of antimicrobial agents and occurrence of antimicrobial resistance in Norway. Årlige rapporter. https://unn.no/fag-og-forskning/norm-norsk-overvakingssystem-for-antibiotikaresistens-hos-mikrober#rapporter/ Accessed 20.10.2020.
- 11.
Adam HJ, Baxter MR, Davidson RJ et al. Comparison of pathogens and their antimicrobial resistance patterns in paediatric, adult and elderly patients in Canadian hospitals. J Antimicrob Chemother 2013; 68 (suppl 1): i31–7. [PubMed][CrossRef]
- 12.
Bielicki JA, Lundin R, Sharland M. Antibiotic resistance prevalence in routine bloodstream isolates from children's hospitals varies substantially from adult surveillance data in Europe. Pediatr Infect Dis J 2015; 34: 734–41. [PubMed][CrossRef]
- 13.
Boggan JC, Navar-Boggan AM, Jhaveri R. Pediatric-specific antimicrobial susceptibility data and empiric antibiotic selection. Pediatrics 2012; 130: e615–22. [PubMed][CrossRef]
- 14.
European Commitee on Antimicrobial Susceptibility testing (EUCAST). Clinical breakpoints – breakpoints and guidance. https://eucast.org/clinical_breakpoints/ Accessed 30.9.2020.
- 15.
Statistisk sentralbyrå. 07459: Population, by sex and one-year age groups (M) 1986–2021. https://www.ssb.no/en/statbank/table/07459/ Accessed 1.6.2020.
- 16.
Frumkin K. Bacteriology of urinary tract infections in emergency patients aged 0-36 months. J Emerg Med 2015; 48: 405–15. [PubMed][CrossRef]
- 17.
Størdal K, Mårild K, Blix HS. Bruk av antibiotika hos barn i perioden 2005 – 16. Tidsskr Nor Legeforen 2017; 137. doi: 10.4045/tidsskr.17.0272. [PubMed][CrossRef]
- 18.
Thaulow CM, Berild D, Eriksen BH et al. Potential for more rational use of antibiotics in hospitalized children in a country with low resistance: Data from eight point prevalence surveys. Pediatr Infect Dis J 2019; 38: 384–9. [PubMed][CrossRef]
- 19.
Andreassen V, Waagsbø B, Blix HS. Ciprofloxacin usage at a local hospital. Tidsskr Nor Legeforen 2020; 140. doi: 10.4045/tidsskr.20.0146. [PubMed][CrossRef]
- 20.
Dewar S, Reed LC, Koerner RJ. Emerging clinical role of pivmecillinam in the treatment of urinary tract infection in the context of multidrug-resistant bacteria. J Antimicrob Chemother 2014; 69: 303–8. [PubMed][CrossRef]
- 21.
Jansåker F, Frimodt-Møller N, Benfield TL et al. Mecillinam for the treatment of acute pyelonephritis and bacteremia caused by Enterobacteriaceae: a literature review. Infect Drug Resist 2018; 11: 761–71. [PubMed][CrossRef]
- 22.
Klingenberg C, Småbrekke L, Døllner H et al. Peroral antibiotikabehandling av urinveisinfeksjon hos barn. Tidsskr Nor Legeforen 2009; 129: 1342–4. [PubMed][CrossRef]
- 23.
Montini G, Toffolo A, Zucchetta P et al. Antibiotic treatment for pyelonephritis in children: multicentre randomised controlled non-inferiority trial. BMJ 2007; 335: 386. [PubMed][CrossRef]
- 24.
Leflon-Guibout V, Ternat G, Heym B et al. Exposure to co-amoxiclav as a risk factor for co-amoxiclav-resistant Escherichia coli urinary tract infection. J Antimicrob Chemother 2002; 49: 367–71. [PubMed][CrossRef]
- 25.
National Instutute for Health and Care Excellence (NICE). Pyelonephritis (acute): antimicrobial prescribing. https://www.nice.org.uk/guidance/ng111/chapter/Recommendations/ Accessed 22.10.2020.
- 26.
The Royal’s Children Hospital Melbourne. Gudineline for treatment of urinary tract infections. https://www.rch.org.au/clinicalguide/guideline_index/Urinary_tract_infection/ Accessed 19.10.2020.
- 27.
Beus JM, Cowden CL, Metjian TA et al. Cephalexin for outpatient urinary tract infections in children. Open Forum Infect Dis 2015; 2 (suppl 1): 1572. [CrossRef]
- 28.
Cheng CH, Tsai MH, Huang YC et al. Antibiotic resistance patterns of community-acquired urinary tract infections in children with vesicoureteral reflux receiving prophylactic antibiotic therapy. Pediatrics 2008; 122: 1212–7. [PubMed][CrossRef]