Main findings
A total of 13 cases of leishmaniasis were registered in the period March 2014 – September 2017.
Visceral leishmaniasis was diagnosed late in the course of the disease, and the patients had been infected in Mediterranean countries, places clinicians rarely associate with tropical infections.
Visceral leishmaniasis and Leishmania species with the potential for causing mucocutaneous disease were diagnosed by PCR and sequencing and treated effectively with liposomal amphotericin B, in accordance with international guidelines.
Liposomal amphotericin B, and not local therapy, was administered as the first-line option to all cases of cutaneous leishmaniasis.
Leishmaniasis is caused by the protozoan Leishmania, which belongs to the class Kinetoplastida, the order Tryanosomatida and the family Trypanosomatidae. Leishmania is transmitted by sandfly bites, and depending on the species, dogs, rodents and humans are common reservoirs (1).
The manifestation depends on immune status and Leishmania species. It varies from a fatal course in visceral leishmaniasis if it goes untreated, to extensive lesions in the face and throat in the case of the mucocutaneous form, and to self-healing lesions with the cutaneous form (1).
Visceral leishmaniasis is endemic in 78 countries (2). The incidence has declined in recent years, and in 2018 about 17 000 new cases were reported. Of these, 90 % were in Brazil, Sudan, South-Sudan, Ethiopia, Kenya, Somalia and India (2). However, infections also occur in southern Europe. A study of travellers in Europe in the period 2000–2012 found 10 cases of visceral leishmaniasis and 30 cases of cutaneous leishmaniasis, the places of exposure being Spain, Malta and Italy (3). The mucocutaneous form, which is usually caused by the subgenus Leishmania (Viannia), is found mainly in South and Central America, while the cutaneous form is most common in South and Central America, Africa and Asia. The incidence of cutaneous leishmaniasis is increasing (2).
The Infectious Diseases Society of America (IDSA) has published guidelines for diagnosis and treatment in 2016 (4), and a European group of experts published recommendations for cutaneous leishmaniasis in 2014 (5). Polymerase chain reaction (PCR) testing and sequencing are recommended for species identification, which provides a basis for choice of treatment. This was not available in Norway as routine diagnostic testing before 2015.
Liposomal amphotericin B is recommended for visceral and mucocutaneous leishmaniasis, while local treatment is recommended for the cutaneous form in suitable cases (4, 5). Treatment for cutaneous leishmaniasis must be individualised, as there is no evidence providing a basis for universal recommendations. Drugs for local treatment do not have market authorisation in Norway and must be imported for compassionate use.
As this disease is rare in Norway, and the drugs are not readily available or are associated with potential adverse effects, there is a risk of inappropriate management. The aim of this study was to investigate the number of cases diagnosed, performance of diagnostic methods used and treatment of leishmaniasis at five university hospitals in Norway.
Material and method
Patients with suspected leishmaniasis at the university hospitals of Bergen, Oslo (Ullevål), Stavanger, Trondheim and Tromsø were included prospectively in the period 1.3.2014–30.9.2017. The initial plan was to include 50 patients, in order to compare PCR methods. In 2015 PCR was introduced as a routine diagnostic method at Ullevål Hospital, thereby reducing the need for method development. The method development part of the study was therefore terminated, but the incidence, diagnosis and treatment of leishmaniasis in Norway continued to be reported for a period of three years. Data on manifestation, place of exposure, immune status, diagnosis, treatment and outcomes were recorded.
Microscopy was performed locally, and the study hospitals decided where to send specimens for further analysis and species identification. At Oslo University Hospital, Ullevål, real-time PCR with 18S rDNA as the target gene was used for detection, and sequencing of the hsp70 gene was used for species identification (6). At the Public Health Agency of Sweden, non-species-specific serology was performed with L. (L.) donovani as antigen, conventional PCR was carried out with 18S rDNA as target gene, and species identification was performed with the aid of restriction fragment length polymorphism (RFLP).
In addition to routine diagnostics, the Norwegian National Advisory Unit on Tropical Infectious Diseases carried out PCR with 18S rDNA (7) and AAP3 (arginine permease 3) as target genes (8). Specimens from bone marrow, ethylenediamine tetraacetate (EDTA) blood, spleen biopsy, punch biopsy, fine-needle biopsy and absorptive filter paper swabs of the base of lesions (Whatman FTA, Sigma-Aldrich, St. Louis, USA) (see the appendix) were collected depending on clinical presentation.
Ethics
Patients gave written consent to taking part in the study. The study was approved by the Regional Ethics Committee (approval number: 2014/805).
Results
Cases diagnosed, place of exposure and clinical data
Thirteen patients diagnosed with leishmaniasis were registered during the period. Two had visceral leishmaniasis caused by Leishmania (L.) infantum. Both were elderly people (> 70 years) who had been infected in the Mediterranean area, one of whom, as previously reported in the Journal of the Norwegian Medical Association, was immunosuppressed by methotrexate due to rheumatoid arthritis (9). Both had persistent fever, weight loss, splenomegaly, elevated sedimentation rate and CRP, pancytopenia, hypoalbuminaemia and hypergammaglobulinaemia.
Cutaneous leishmaniasis was diagnosed in eleven patients. Two had been infected in Central America with the potentially mucotropic species L. (Viannia) braziliensis and L. (V.) naiffi, but neither had developed mucocutaneous leishmaniasis. Five had been infected in Mediterranean countries with L. (L.) tropica, L. (L.) infantum or L. (L.) major, two with L. (L.) major in the Middle East and two with L. (L.) mexicana in Central America. Four patients had two or fewer lesions, while seven had more than two lesions. Ten patients had lesions in areas exposed to the vector, such as face and extremities, while one had more atypical multiple lesions.
Diagnostic testing
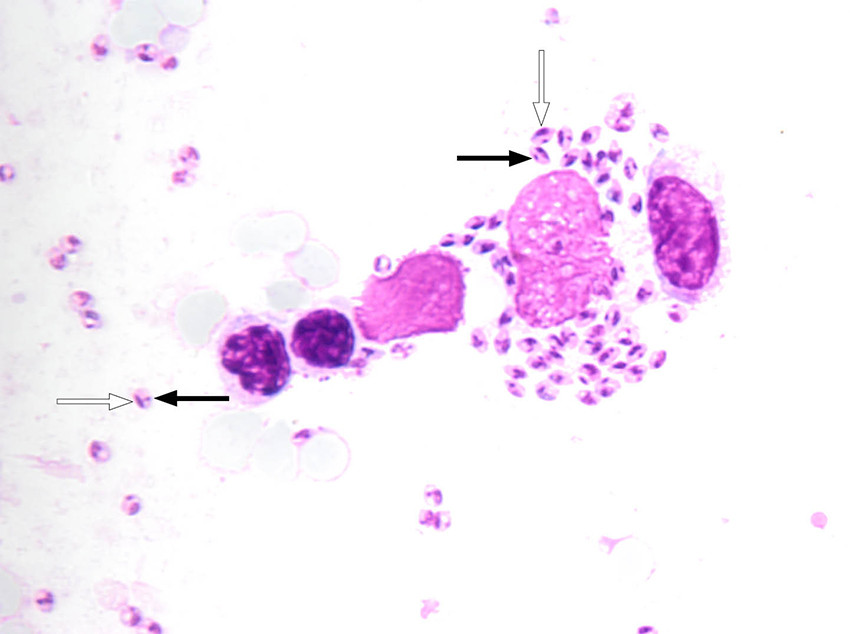
The patients with visceral leishmaniasis had positive serology and parasites were detected by PCR and/or microscopy of a spleen biopsy (Figure 1) and bone marrow. In both, Leishmania was also detected by PCR analysis of whole blood.
In ten patients with cutaneous leishmaniasis, PCR on punch biopsy (N = 9) or lesion scrapings identified Leishmania species. Microscopy revealed parasites in five of six patients with a positive PCR test. One patient had a negative PCR test, but L. (L.) major infection was diagnosed on the basis of exposure. Diagnostic testing was performed in line with guidelines (4, 5).
Specimens from 11 patients was collected for comparison of PCR methods. Two different target genes were used for PCR tests: 18S rDNA and AAP3. Leishmania was identified in more cases by using 18S rDNA than by using AAP3 as the target gene (Appendix).
Treatment
The patients with visceral leishmaniasis were treated with liposomal amphotericin B, as recommended in the guidelines (4), and both recovered. The cumulative dose of liposomal amphotericin B for the immunocompromised patient was 44 mg/kg (4 mg/kg days 1–6, 11, 17, 24, 31 and 38).
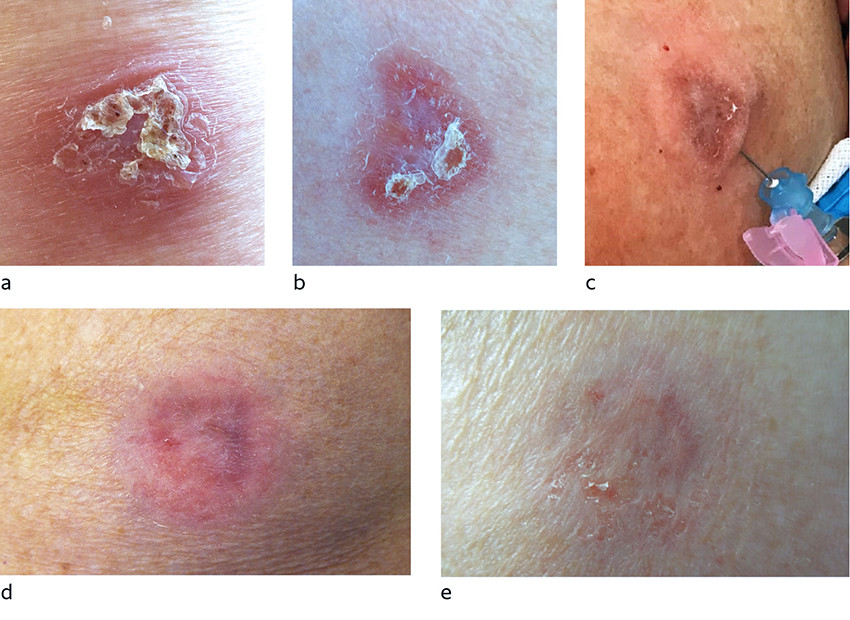
All patients who received drug therapy for cutaneous leishmaniasis (N = 8/11) were treated with liposomal amphotericin B as first-line choice. Seven received a cumulative dose of 18 mg/kg (3 mg/kg days 1–5 and 10). A standard dose recommendation for liposomal amphotericin B for treating cutaneous leishmaniasis has not been established, but the dosage was in accordance with recommendations in international guidelines (4). One patient with L. (L.) infantum infection did not respond to liposomal amphotericin B, but recovered after local treatment with sodium stibogluconate (4 mL at 100 mg/mL) combined with cryotherapy (Figure 2) administered in accordance with treatment recommendations (4, 5). The lesions of three patients with L. (L.) major or L. (L). Infantum infections healed spontaneously without treatment.
Discussion
Cases diagnosed, place of exposure and findings
Patients with visceral leishmaniasis had classic symptoms of persistent fever, weight loss, enlarged spleen, pancytopenia and hypergammaglobulinaemia (1), but correct diagnose and treatment were delayed. They were infected in the Mediterranean area, where L. (L.) infantum is endemic (10), but it is rarely encountered in travellers to Norway and the diagnosis is therefore often not considered until late in the disease course. However, in recent years there has been a relatively high incidence of infection with L. (L.) infantum in the Madrid region (11). Visceral leishmaniasis in Norway in patients infected in Spain has also been reported earlier (12, 13). One of the cases included in this study has also been reported earlier in the Journal of the Norwegian Medical Association (9). Given increasing travel activities and more refugees from endemic areas, it is probable that the incidence of leishmaniasis will increase in Norway (1, 14, 15).
The patients with visceral leishmaniasis in this study were elderly people, and one of them used immunosuppressive drugs. Compromised immunity is a known risk factor for visceral leishmaniasis, and the incidence of visceral leishmaniasis is increasing in immunocompromised travellers (15, 16).
Diagnostic testing
The patients with visceral leishmaniasis had positive serology and parasites were detected by PCR and/or microscopy of blood and tissue. In general, serology and PCR, microscopy and if relevant culture of bone marrow, spleen, lymph node, liver or blood are recommended for diagnostic testing, PCR being the most sensitive (4). Spleen aspirate is the gold standard, but due to risk of haemorrhagic complications (1 in 5 000), bone marrow analysis is often preferred (4, 17). Blood PCR is useful, and our findings are consistent with studies reporting that this test has high sensitivity (18). In cases of immunosuppression, there is a risk of false negative serology, and PCR is particularly useful in these cases (4).
Sequencing was used for species identification in patients with cutaneous leishmaniasis (4). PCR, microscopy and, if available, cultures from lesions or mucosa are recommended for the diagnosis of cutaneous and mucocutaneous leishmaniasis (4, 19). Molecular diagnostic methods are most sensitive, and are necessary for species identification. Species identification is particularly important for South and Central American infections, as the subgenus Leishmania (Viannia) can cause mucocutaneous leishmaniasis (4). Only one patient had a negative PCR test in this study, probably due to a low number of parasites in spontaneously resolving lesions.
Punch or fine-needle biopsies from the edge of a lesion or lesions scrapings are recommended as sample material (4, 19). Punch biopsy was performed in this study as a routine diagnostic test, in line with recommendations. In supplementary tests, PCR from a punch biopsy was positive in most cases compared with other specimens consisting of a smaller volume of tissue (Appendix). Filter paper swabbed directly on the base of the lesion is practical for sampling and dispatch, but PCR tests were negative for two of five such specimens in this study (Appendix).
Treatment
The recommended treatment for visceral leishmaniasis in immunocompetent patients is liposomal amphotericin B 3 mg/kg intravenously on days 1–5, 14 and 21 (4). A cumulative dose of 40 mg/kg is recommended for patients with immune deficiency. Patients with visceral leishmaniasis responded well to liposomal amphotericin B without exhibiting severe adverse effects. In endemic areas, liposomal amphotericin B is often not available because of its high price and refrigeration requirement, and miltefosine, sodium stibogluconate (Pentostam) and paromomycin are other options (1). In several Asian and African countries, monotherapy with miltefosine or sodium stibogluconate has been replaced with shorter courses and combination courses with other drugs because of increasing resistance and treatment failure, but the efficacy of the new courses varies across regions (20).
In cases of cutaneous leishmaniasis among immunocompetent persons infected with a Leishmania species that is not associated with mucocutaneous leishmaniasis, and where lesions are few and small, it is recommended to observe without drug treatment and wait for spontaneous healing (4, 5). Spontaneous healing may take 2–15 months (21). The options for active treatment of cutaneous leishmaniasis are: local treatment with sodium stibogluconate (Figure 2), cryotherapy, heat therapy and systemic treatment (4, 5). In cases of multiple or large lesions, facial lesions or infection with a species that can potentially cause mucocutaneous leishmaniasis, systemic treatment is recommended (4, 5).
All patients in this study who received drug therapy (N = 8/11) received liposomal amphotericin B as initial therapy, also in cases where the guidelines recommend local treatment (4, 5). Systemic treatment is expensive and has potentially serious adverse effects, but drugs for local therapy are less readily available, injections are painful and the methodology not well known among clinicians, which probably explains why this method is less widely used. Liposomal amphotericin B is less effective in cutaneous than in visceral leishmaniasis, probably because of lower drug concentrations in skin. At present there are no established recommendations based on controlled studies for using liposomal amphotericin B against cutaneous leishmaniasis (1). In a French study of cutaneous and mucocutaneous leishmaniasis, liposomal amphotericin B was only effective in 46 % (19/41) of patients (22). Similarly, we found that liposomal amphotericin B was not effective in one patient with L. (L.) infantum infection (Figure 2). This patient responded to two courses of treatment two weeks apart consisting of intracutaneous injections of sodium stibogluconate combined with cryotherapy.
Follow-up in case of treatment failure after three months or later recurrence is recommended for at least one year for cutaneous leishmaniasis and for ten years for potential mucocutaneous leishmaniasis (1, 4). After treatment for visceral leishmaniasis, the parasites remain latent in the body life-long, with risk of recurrence in the case of immunosuppression. Biochemical parameters for predicting recurrence are not available. We recommend that patients who have been treated be informed of the need to be alert to clinical signs of recurrence, and that they should not be blood donors (4).
Conclusion
The patients with visceral leishmaniasis were effectively treated, but diagnosed late in the course of the disease. Diagnosis of cutaneous leishmaniasis was in accordance with recommendations, but liposomal amphotericin B rather than local treatment was administered more frequently than recommended in the guidelines.
The article has been peer-reviewed.
- 1.
Burza S, Croft SL, Boelaert M. Leishmaniasis. Lancet 2018; 392: 951–70. [PubMed][CrossRef]
- 2.
Ruiz-Postigo JA, Grout L, Saurabh J. Global leishmaniasis surveillance, 2017-2018, and first report on 5 additional indicators. Wkly Epidemiol Rec 2020; 65: 265–80.
- 3.
Ehehalt U, Schunk M, Jensenius M et al. Leishmaniasis acquired by travellers to endemic regions in Europe: a EuroTravNet multi-centre study. Travel Med Infect Dis 2014; 12: 167–72. [PubMed][CrossRef]
- 4.
Aronson N, Herwaldt BL, Libman M et al. Diagnosis and Treatment of Leishmaniasis: Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clin Infect Dis 2016; 63: 1539–57. [PubMed][CrossRef]
- 5.
Blum J, Buffet P, Visser L et al. LeishMan recommendations for treatment of cutaneous and mucosal leishmaniasis in travelers, 2014. J Travel Med 2014; 21: 116–29. [PubMed][CrossRef]
- 6.
Van der Auwera G, Maes I, De Doncker S et al. Heat-shock protein 70 gene sequencing for Leishmania species typing in European tropical infectious disease clinics. Euro Surveill 2013; 18: 20543. [PubMed][CrossRef]
- 7.
Uliana SR, Nelson K, Beverley SM et al. Discrimination amongst Leishmania by polymerase chain reaction and hybridization with small subunit ribosomal DNA derived oligonucleotides. J Eukaryot Microbiol 1994; 41: 324–30. [PubMed][CrossRef]
- 8.
Tellevik MG, Muller KE, Løkken KR et al. Detection of a broad range of Leishmania species and determination of parasite load of infected mouse by real-time PCR targeting the arginine permease gene AAP3. Acta Trop 2014; 137: 99–104. [PubMed][CrossRef]
- 9.
Blomberg B, Müller KE, Helgeland L et al. En mann i 80-årene med leddgikt og vedvarende feber. Tidsskr Nor Legeforen 2019; 139. doi: 10.4045/tidsskr.18.0546. [PubMed][CrossRef]
- 10.
Ready PD. Leishmaniasis emergence in Europe. Euro Surveill 2010; 15: 19505. [PubMed]
- 11.
Arce A, Estirado A, Ordobas M et al. Re-emergence of leishmaniasis in Spain: community outbreak in Madrid, Spain, 2009 to 2012. Euro Surveill 2013; 18: 20546. [PubMed][CrossRef]
- 12.
Besada E, Njålla RJ, Nossent JC. Imported case of visceral leishmaniasis presenting as pancytopenia in a Norwegian patient treated with methotrexate and etanercept for psoriasis arthritis. Rheumatol Int 2013; 33: 2687–9. [PubMed][CrossRef]
- 13.
Moser KH, Rødevand E, Hammerstrøm J et al. En tidligere frisk mann med feberepisoder med kirkespirforløp. Tidsskr Nor Legeforen 2008; 128: 1845–7. [PubMed]
- 14.
Söbirk SK, Inghammar M, Collin M et al. Imported leishmaniasis in Sweden 1993-2016. Epidemiol Infect 2018; 146: 1267–74. [PubMed][CrossRef]
- 15.
Schwartz T, Jensenius M, Blomberg B et al. Imported visceral leishmaniasis and immunosuppression in seven Norwegian patients. Trop Dis Travel Med Vaccines 2019; 5: 16. [PubMed][CrossRef]
- 16.
Fletcher K, Issa R, Lockwood DNJ. Visceral leishmaniasis and immunocompromise as a risk factor for the development of visceral leishmaniasis: a changing pattern at the hospital for tropical diseases, london. PLoS One 2015; 10: e0121418. [PubMed][CrossRef]
- 17.
Sundar S, Rai M. Laboratory diagnosis of visceral leishmaniasis. Clin Diagn Lab Immunol 2002; 9: 951–8. [PubMed]
- 18.
de Ruiter CM, van der Veer C, Leeflang MMG et al. Molecular tools for diagnosis of visceral leishmaniasis: systematic review and meta-analysis of diagnostic test accuracy. J Clin Microbiol 2014; 52: 3147–55. [PubMed][CrossRef]
- 19.
Centers for Disease Control and Prevention. Practical guide for specimen collection and reference diagnosis of leishmaniasis. https://www.cdc.gov/parasites/leishmaniasis/resources/pdf/cdc_diagnosis_guide_leishmaniasis_2016.pdf Accessed 11.1.2021.
- 20.
Alves F, Bilbe G, Blesson S et al. Recent development of visceral leishmaniasis treatments: Successes, pitfalls, and perspectives. Clin Microbiol Rev 2018; 31: e00048–18. [PubMed][CrossRef]
- 21.
Reithinger R, Dujardin JC, Louzir H et al. Cutaneous leishmaniasis. Lancet Infect Dis 2007; 7: 581–96. [PubMed][CrossRef]
- 22.
Guery R, Henry B, Martin-Blondel G et al. Liposomal amphotericin B in travelers with cutaneous and muco-cutaneous leishmaniasis: Not a panacea. PLoS Negl Trop Dis 2017; 11: e0006094. [PubMed][CrossRef]