Metal ion levels in the blood of patients with metal-on-metal hip prostheses
Main findings
A group of patients who had received a BHR prosthesis experienced a significant increase in the median blood concentration of cobalt and chromium one to five years after surgery.
Five patients had metal ion levels that exceeded the threshold value of 7 μg/L, but were asymptomatic.
Five symptomatic patients who underwent revision surgery had metal ion levels below 7 μg/L. Metal ion concentration alone does not provide sufficient information to predict the need for revision surgery.
Metal-on-metal (MoM) hip prostheses, where both the femoral head and the hip cup are made of metal, were introduced in the 1960s. Two types were used: a total hip prosthesis with a stem, with either a large or small head (≤ 32 mm), and a resurfacing prosthesis without a stem (1). In a standard hip prosthesis, the femoral head and most of the femoral neck are removed and replaced with a prosthesis that has a smaller head diameter, and which is implanted in the marrow cavity of the femur. In such cases, the joint surfaces are normally made of metal-on-polyethylene or ceramic. However, with resurfacing prostheses, the femoral neck and most of the femoral head are retained, and the femoral head is given a new metal surface. Here the new femoral head is the same size as the original and rests in a cup made from the same material, normally a cobalt-chromium-molybdenum alloy. The Birmingham hip resurfacing prosthesis, referred to here as the BHR prosthesis, was developed in the 1990s (Figure 1) (2). It was designed to give better hip joint stability and mobility than a conventional prosthesis, and to reduce the problem of polyethylene wear and osteolysis (3).

According to the Norwegian Arthroplasty Register, 485 resurfacing prostheses were implanted in patients in Norway between 1987 and 2013, of which 466 were BHR prostheses (4, 5). Several countries have experienced an increased incidence of adverse soft tissue reactions which led to complex revision surgery of metal-on-metal prostheses. Pseudotumours (6), aseptic lymphocyte-dominated vasculitis-associated lesions (ALVAL) (7) and adverse reactions to metal debris (ARMD) (8) are all terms that have been used to describe potentially harmful reactions to metal wear products released from the bearing surfaces of such prostheses. The revision rate for several types of resurfacing prostheses was high, partly due to these soft tissue reactions, and partly due to other complications such as femoral neck fractures and prosthesis dislocation (9). These complications eventually led to a rapid reduction in the use of such prostheses (2). In many countries, including Norway, the use of metal-on-metal prostheses was discontinued in 2013.
A link has been found between the concentration of metal ions in the blood and local soft tissue reactions. Measuring the metal ion concentration in the blood of selected patients with metal-on-metal prostheses is therefore recommended, and in Norway a threshold value of 7μg/L has been set (10).
Systemic effects such as neurological problems, visual disturbances, hearing loss, memory loss, depression (11) and cardiomyopathy (12) are rare, but they have been reported in patients with some types of metal-on-metal prostheses, in particular those with toxic levels of cobalt (13).
The aim of this study was to analyse the release of metal ions in patients during a five-year period following implantation of a BHR prosthesis. In addition, we wanted to establish whether the trace metal levels were linked to clinical complications such as pain and reduced clinical function, or revision surgery.
Material and method
Ethics
The study protocol was approved by the Regional Committee for Medical and Health Research Ethics, REC West (project number 2010/2817). Written informed consent was obtained from the patients prior to taking blood samples. De-identified samples were used in the study.
Patients
Forty-four male patients at the Coastal Hospital at Hagevik in the period October 2009 to May 2013 were included and followed prospectively (Table 1). The median age was 53 years (min.–max. 36–69 years). Thirty-six patients received a BHR prosthesis in one hip, while 8 received a BHR prosthesis in both hips.
Table 1
Patients implanted with unilateral and bilateral BHR prostheses at the Coastal Hospital at Hagevik in the period October 2009 to May 2013. Median (min.–max.) age, prosthesis size, cup inclination angle and hip function scores five years after surgery.
| Patients with unilat./bilat. BHR prosthesis, n |
36/8 |
|---|---|
| Age, years |
53 (36–69) |
| Head size, mm |
54 (46–64) |
| Cup size, mm |
60 (54–66) |
| Cup inclination angle, ° |
38 (29–49) |
| Harris Hip Score |
96 (61–100) |
| Oxford Hip Score |
13 (12–31) |
The most common indication for surgery was primary osteoarthritis (n = 30) followed by secondary osteoarthritis (n = 11).
Clinical assessment
Two validated hip scoring systems were used for the clinical assessment of the prosthetic joint: the Harris Hip Score and the Oxford Hip Score. The Harris Hip Score was administered by a doctor, and consists of the following categories: pain, function, joint movement and joint deformity. The scores from the different categories are tallied, and the optimal score is 100, while 0 is the poorest score (14).
The Oxford Hip Score is based on a form consisting of 12 questions that measure function and pain, and which is completed by the patient. The optimal score is 12, while the poorest is 60 (15).
The size of the prosthesis was individually adapted to each patient. Pelvis and hip X-rays were taken of all patients within one week after surgery, and Sante DICOM Editor was used to measure the angle of inclination of the cup. Clinical data on patients and prosthetic components were retrieved from medical records at the hospital and checked against data from the Norwegian Arthroplasty Register.
Analysis of cobalt and chromium levels in whole blood
Blood samples were collected before surgery and again after three months, one year, three years and five years.
Blood was drawn from a vein in the forearm using an intravenous cannula (Becton Dickinson Venflon Pro) and stored at −20 °C in metal-free polypropylene tubes (VWR). Prior to analysis, 1.5 mL of whole blood was broken down with a mixture of 3 mL of 60 % ultrapure HNO3 and 2 mL of 30 % H2O2 (Merck) in a microwave-assisted system (Milestone 1200 Mega). After the digestion, the solution was diluted with double-distilled, deionized water (MilliQ).
The concentration of cobalt and chromium in whole blood was analysed using high resolution inductively coupled plasma mass spectrometry (HR-ICP-MS) (Thermo Scientific, Element XR).
The limit of detection was 0.04 μg/L for cobalt and 0.11 μg/L for chromium. The accuracy of the analytical method was monitored using a reference material (Seronorm Trace Elements Whole Blood L-2).
Statistical analysis
Non-parametric statistical methods were used to analyse the data in GraphPad Prism 8 software. Results are given here as a median and as lowest and highest values. The Wilcoxon matched-pairs signed rank test was used to compare preoperative and postoperative metal concentrations at patient level. Correlations between the blood metal level after five years and other parameters (angle of inclination, head size, clinical scores) were assessed using Spearman's rank correlation coefficient (rSp) in patients with a unilateral prosthesis. Five blood samples with chromium concentration below the detection limit were assigned a value of half the detection limit. The statistical significance level was defined as p <0.05.
Results
Blood metal levels
The concentration of cobalt (Figure 2a and Table 2a) and chromium (Figure 2b and Table 2b) in the blood increased between the surgery date and one year post-surgery (p <0.001) both for patients with a unilateral prosthesis and for patients with bilateral prostheses (Table 2). Neither the concentration of cobalt (p = 0.057) nor that of chromium (p = 0.261) changed significantly between one and five years after surgery.
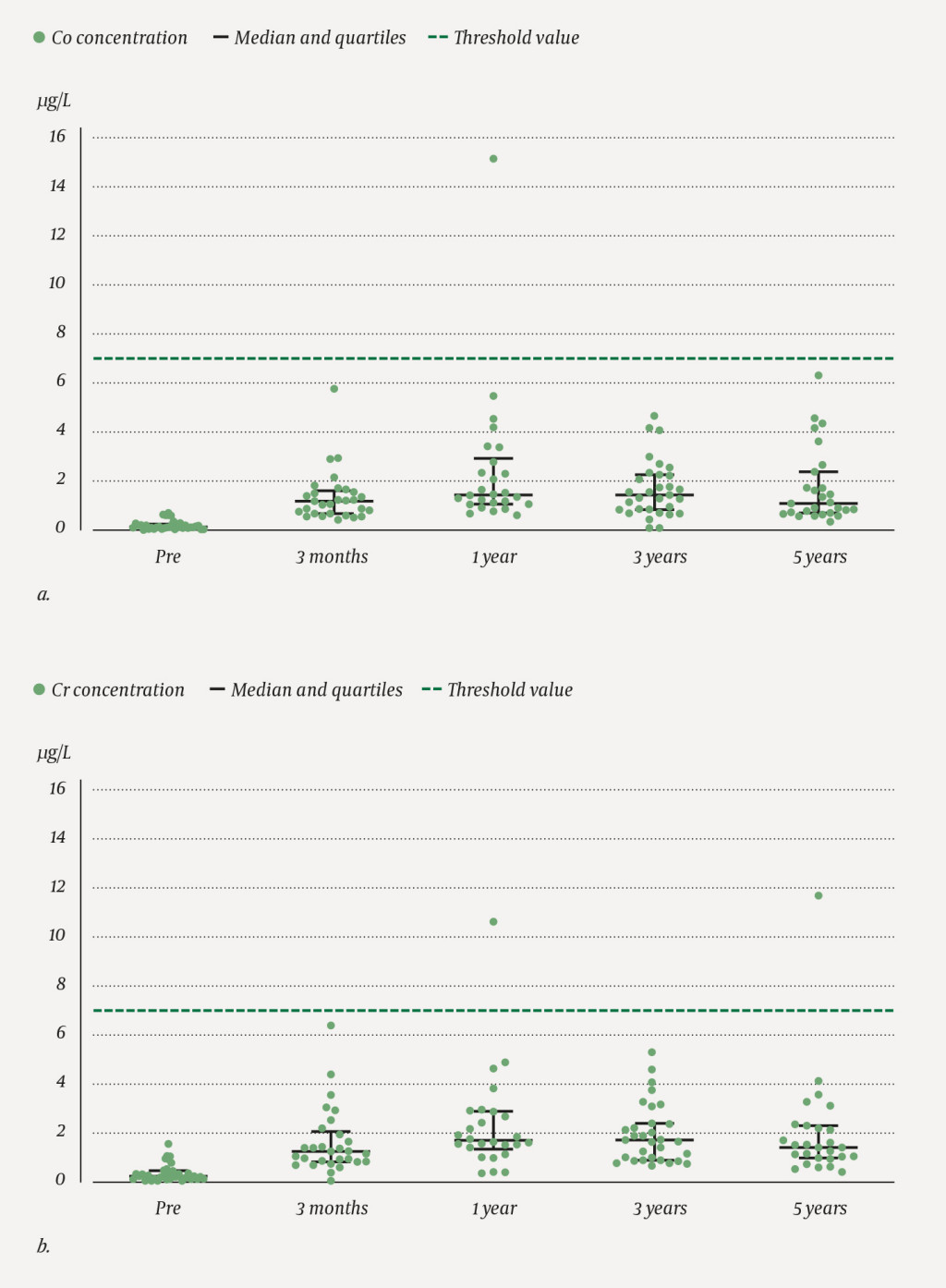
Table 2a
Number of blood samples available and whole blood concentrations, represented by the median and lowest and highest values, of cobalt in patients with unilateral (Uni) and bilateral (Bilat) BHR prostheses at all measurement times (before surgery and three months, one year, three years and five years afterwards). Cobalt in whole blood (μg/L).
| Preoperative |
3 months |
1 year |
3 years |
5 years |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
|
| No. of blood samples |
34 |
8 |
29 |
5 |
26 |
6 |
31 |
5 |
27 |
7 |
| Median (min.–max.) |
0.14 (0.02–0.71) |
0.84 (0.05–2.75) |
1.19 (0.43–5.78) |
3.72 (0.46–14.98) |
1.45 (0.62–15.16) |
3.20 (1.17–20.16) |
1.45 (0.10–4.68) |
3.25 (2.15–15.26) |
1.10 (0.36–6.32) |
2.29 (1.55–28.53) |
Table 2b
Number of blood samples available and whole blood concentrations, represented by the median and lowest and highest values, of chromium in patients with unilateral (Uni) and bilateral (Bilat) BHR prostheses at all measurement times (before surgery and three months, one year, three years and five years afterwards). Chromium in whole blood (μg/L).
| Preoperative |
3 months |
1 year |
3 years |
5 years |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
Uni |
Bilat |
|
| No. of blood samples |
34 |
8 |
29 |
5 |
26 |
6 |
31 |
5 |
27 |
7 |
| Median (min.–max.) |
0.23 (0.05–1.55) |
1.17 (0.05–3.23) |
1.25 (0.05–6.39) |
3.18 (0.05–12.32) |
1.70 (0.36–10.63) |
2.93 (1.34–11.07) |
1.71 (0.66–5.30) |
3.05 (2.71–10.07) |
1.41 (0.40–11.70) |
2.56 (1.65–14.07) |
Five patients had cobalt and/or chromium concentrations above the national threshold value of 7 μg/L at one or more of the postoperative measurement times. Three of them had bilateral prostheses. The highest cobalt concentration observed was 28.5 μg/L, and the same patient also had the highest chromium concentration (14.1 μg/L).
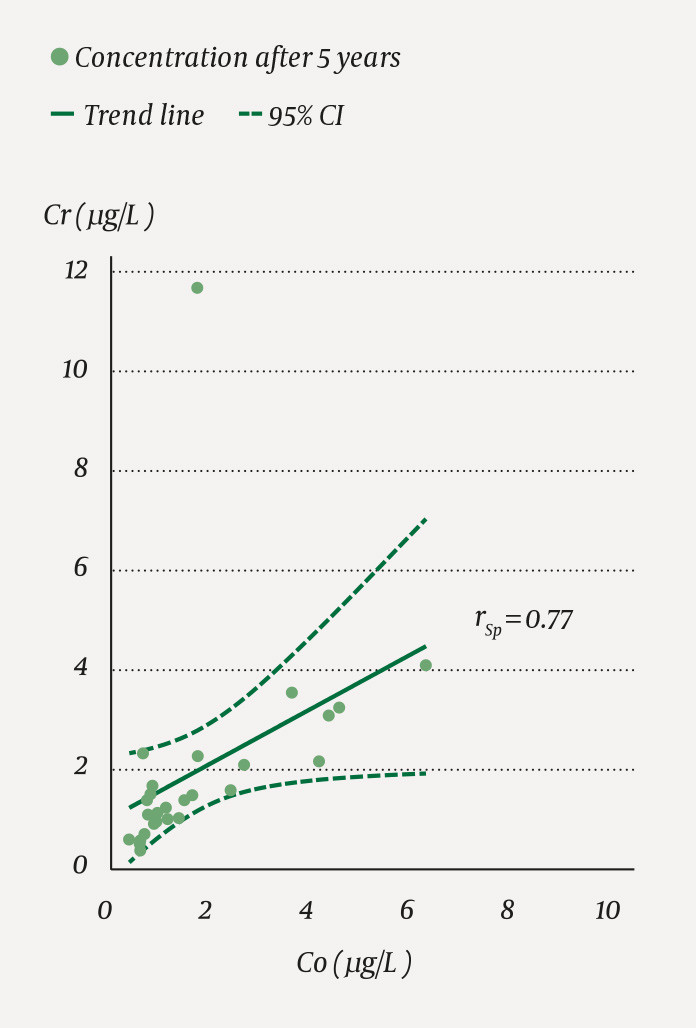
There was a correlation (rSp = 0.77; p <0.001) between the concentration of cobalt and chromium in whole blood in patients with a unilateral prosthesis five years after surgery (Figure 3).
Clinical findings
The Harris Hip Score improved from 60 points (36–91 points) preoperatively to 96 points (61–100 points) after five years. The median Oxford Hip Score improved from 33 points (21–45 points) to 13 points (12–31 points) five years after surgery.
We found no correlation between the clinical scores of patients with a unilateral prosthesis and the metal levels in the blood five years after surgery. The five patients with metal levels above the threshold value of 7 μg/L had a median Harris Hip Score of 97 points (93–100 points) five years after the procedure.
Five revision surgeries were performed during the five-year period due to pain (n = 3), prosthetic dislocation (n = 1) and pseudotumour (n = 1). All of these patients had metal levels below 7 μg/L.
Prosthesis head size, cup inclination angle and correlation with metal level
The median head size was 54 mm (46–64 mm), and only one femoral component was less than 50 mm (Table 1).
The median cup inclination angle was 38°, and the angle varied between 29° and 49°, which is within the recommended range in the literature.
We found no statistically significant correlation between the metal ion levels five years after surgery and the size of the prosthesis head or the inclination angle of the cup.
Discussion
In this study, patients' blood metal concentration levels peaked after one year, and remained at this level at the three and five-year measurements. The median values are well below the Norwegian threshold value of 7 μg/L, and are comparable to the levels reported in other studies of patients with a BHR prosthesis (2, 16). In a multi-centre study of 416 patients with a BHR prosthesis, a median concentration in whole blood of 1.4 μg/L for cobalt and 1.29 μg/L for chromium was found in patients with a unilateral prosthesis, and 2.1 μg/L for cobalt and 2.3 μg/L for chromium in patients with bilateral prostheses (17).
Only five patients in our study exceeded the Norwegian threshold value. All five had good clinical results assessed by hip scores, but they are considered to be at risk of developing a pseudotumour and are therefore being closely monitored.
The metal ion level in blood is used as an indicator of potential complications associated with the joint prosthesis, but sensitivity and specificity in this respect are relatively low (18), which is illustrated by our findings to some extent.
In our study, 5 of 44 patients underwent revision surgery, and in 4 of the reoperated patients, the prosthesis was replaced with no objective findings indicating a pseudotumour. All reoperated patients had metal levels below the threshold value of 7 μg/L.
The overall revision rate of resurfacing prostheses (mainly BHR prostheses) in Norway has been shown to be 5 % on average (95 % confidence interval 3.0 to 7.0 %) after five years (5). In comparison, standard total hip prostheses had a lower revision risk of 3.8 % (95 % CI 3.6 to 4.0 %) after five years (5).
There are few longitudinal studies documenting the lifespan of BHR prostheses exceeding ten years. A retrospective study of 95 people with a BHR prosthesis found an overall revision rate of 12 % at ten-year follow-up (16). The revision rate was higher in women (16 %) than in men (7 %). Australia's National Joint Replacement Registry reported a 6.6 % revision of BHR prostheses at ten-year follow-up. Other resurfacing prostheses, such as the ASR (articular surface replacement, DePuy), have had far poorer results, with up to 30 % revision surgeries after ten years, most likely due to design differences that increase the release of metal (19).
Risk factors have been described for the development of pseudotumours and other soft tissue reactions in patients with metal-on-metal hip prostheses. In addition to patients with elevated concentrations of cobalt and chromium in the blood, women and patients with smaller femoral head sizes (< 50 mm diameter) and a high cup inclination angle (> 50°) have an increased risk (20). It has been claimed that localised edge wear between the head and cup at a high angle of inclination or anteversion angle of the cup results in increased metal ion levels (8). In our study, both cup positioning and component size are within acceptable limits. Only one prosthesis had a femoral head size of less than 50 mm.
The Norwegian National Advisory Unit on Arthroplasty and Hip Fractures recommends that hospitals regularly follow up patients with all types of metal-on-metal prostheses with a head diameter of more than 32 mm, throughout the life of the prosthesis (10). The frequency of follow-up varies according to the patient's gender (women are followed more closely), the size of the femoral head (small heads are followed more closely) and the documented long-term results of the prosthesis. Standard X-rays are supplemented with measurements of metals in whole blood, and if these tests produce adverse findings or there is clinical suspicion (pain, palpable mass, reduction in function), MRI or ultrasound scans are also performed (5).
It has proved difficult to set threshold values for the concentrations of cobalt and chromium in the blood that indicate an increased risk of soft tissue reactions and revision surgery, and no consensus has been reached (17). In most European countries, including Norway, the threshold value is set at 7 μg/L (10). Some have chosen a lower threshold, all the way down to 3 μg/L (5). A lower limit will naturally result in poorer specificity. The debate on this continues.
Increased pseudotumour volume, poorer hip function scores and bilateral metal-on-metal prosthesis have also been associated with future revision surgery (5). There is therefore a need for further investigation into the relationship between metal ion levels and biological causal mechanisms for the development of tissue reactions.
One weakness of the study is that only men were included. At the start of the study, it was shown that women had poorer results with such prostheses, and the prosthesis was only used on men during the inclusion period. The strength of the study is that it was a prospective study with preoperative measurements and several postoperative follow-ups of a cohort from the same hospital. The cohort consists of approximately 10 % of all patients who have received a BHR prosthesis in Norway.
The authors would like to thank Western Norway Regional Health Authority, Bergen Hospital Trust, the Grieg Foundation, Annlaug Drange Steinsland, Roel Bierling, the physiotherapists at the Coastal Hospital at Hagevik, Irene Ohlen Moldestad, Siv Hjorth Dundas, the Norwegian Arthroplasty Register and all the study participants.
The article has been peer reviewed.
- 1.
Head WC. The Wagner surface replacement arthroplasty. Orthop Clin North Am 1982; 13: 789–97. [PubMed]
- 2.
Holland JP, Langton DJ, Hashmi M. Ten-year clinical, radiological and metal ion analysis of the Birmingham Hip Resurfacing: from a single, non-designer surgeon. J Bone Joint Surg Br 2012; 94: 471–6. [PubMed][CrossRef]
- 3.
Logishetty K, van Arkel RJ, Ng KCG et al. Hip capsule biomechanics after arthroplasty: the effect of implant, approach, and surgical repair. Bone Joint J 2019; 101-B: 426–34. [PubMed][CrossRef]
- 4.
Nasjonalt Kompetansesenter for Leddproteser. Årsrapport 2018. Bergen: Helse Bergen HF, Ortopedisk klinikk, Haukeland universitetssjukehus, 2018. http://nrlweb.ihelse.net/Rapporter/Rapport2018.pdf Accessed 27.10.2020.
- 5.
Pijls BG, Meessen JMTA, Tucker K et al. MoM total hip replacements in Europe: a NORE report. EFORT Open Rev 2019; 4: 423–9. [PubMed][CrossRef]
- 6.
Pandit H, Glyn-Jones S, McLardy-Smith P et al. Pseudotumours associated with metal-on-metal hip resurfacings. J Bone Joint Surg Br 2008; 90: 847–51. [PubMed][CrossRef]
- 7.
Langton DJ, Sidaginamale RP, Joyce TJ et al. Aseptic lymphocyte-dominated vasculitis-associated lesions are related to changes in metal ion handling in the joint capsules of metal-on-metal hip arthroplasties. Bone Joint Res 2018; 7: 388–96. [PubMed][CrossRef]
- 8.
Langton DJ, Jameson SS, Joyce TJ et al. Early failure of metal-on-metal bearings in hip resurfacing and large-diameter total hip replacement: A consequence of excess wear. J Bone Joint Surg Br 2010; 92: 38–46. [PubMed][CrossRef]
- 9.
Matharu GS, Pandit HG, Murray DW. Poor survivorship and frequent complications at a median of 10 years after metal-on-metal hip resurfacing revision. Clin Orthop Relat Res 2017; 475: 304–14. [PubMed][CrossRef]
- 10.
Oppfølging av pasienter med metall-på-metall hofteproteser. Anbefaling fra Nasjonalt Register for Leddproteser. http://nrlweb.ihelse.net/Anbefalinger/Metall%20på%20metall.pdf Accessed 20.4.2020.
- 11.
Green B, Griffiths E, Almond S. Neuropsychiatric symptoms following metal-on-metal implant failure with cobalt and chromium toxicity. BMC Psychiatry 2017; 17: 33. [PubMed][CrossRef]
- 12.
Mosier BA, Maynard L, Sotereanos NG et al. Progressive cardiomyopathy in a patient with elevated cobalt ion levels and bilateral metal-on-metal hip arthroplasties. Am J Orthop (Belle Mead NJ) 2016; 45: E132–5. [PubMed]
- 13.
Pelclova D, Sklensky M, Janicek P et al. Severe cobalt intoxication following hip replacement revision: clinical features and outcome. Clin Toxicol (Phila) 2012; 50: 262–5. [PubMed][CrossRef]
- 14.
Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am 1969; 51: 737–55. [PubMed][CrossRef]
- 15.
Dawson J, Fitzpatrick R, Carr A et al. Questionnaire on the perceptions of patients about total hip replacement. J Bone Joint Surg Br 1996; 78: 185–90. [PubMed]
- 16.
Hartmann A, Lützner J, Kirschner S et al. Do survival rate and serum ion concentrations 10 years after metal-on-metal hip resurfacing provide evidence for continued use? Clin Orthop Relat Res 2012; 470: 3118–26. [PubMed][CrossRef]
- 17.
Matharu GS, Berryman F, Judge A et al. Blood metal ion thresholds to identify patients with metal-on-metal hip implants at risk of adverse reactions to metal debris: An external multicenter validation study of birmingham hip resurfacing and corail-pinnacle implants. J Bone Joint Surg Am 2017; 99: 1532–9. [PubMed]
- 18.
Hart AJ, Sabah SA, Bandi AS et al. Sensitivity and specificity of blood cobalt and chromium metal ions for predicting failure of metal-on-metal hip replacement. J Bone Joint Surg Br 2011; 93: 1308–13. [PubMed][CrossRef]
- 19.
Hip K, Arthroplasty S. 2019 Annual Report. Adelaide: Australian Orthopaedic Association National Joint Replacement Registry, 2019. https://aoanjrr.sahmri.com/annual-reports-2019 Accessed 10.3.2020.
- 20.
The safety of metal-on-metal joint replacements with a particular focus on hip implants. Luxembourg: Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR), 2014. https://ec.europa.eu/health/sites/health/files/scientific_committees/emerging/docs/scenihr_o_042.pdf Accessed 14.8.2019.