Long-term follow-up after surgery for atrial fibrillation with concomitant open heart surgery
Main findings
Six months after undergoing surgery for atrial fibrillation using the Cox-Maze IV technique during concomitant open heart surgery, 14 of 19 patients had sinus rhythm.
The frequency of longer-term recurrence was high, particularly in patients with long pre-operative duration of atrial fibrillation.
Peri- and postoperative complications were observed in seven patients, but none were clearly related to the ablation treatment.
The prevalence of atrial fibrillation is high among patients who are to undergo cardiac surgery (1). Modern surgical ablation methods, such as Cox-Maze IV (CMIV) have made it simpler to combine surgical treatment of atrial fibrillation with other cardiac surgery (2). The treatment can now be performed much faster and more safely than previously, when the atrial tissue had to be cut with scissors and scalpel before being sewn up again. In the short term (one year), a success rate of 89 % has been reported after a CMIV procedure (2). In the longer term, such ablation is associated with a reduced rate of post-operative atrial fibrillation, stroke and heart failure (3), and improved long-term survival (4).
The multi-centre trial Concomitant Utilization of Radio Frequency Energy for Atrial Fibrillation (CURE-AF) was conducted in the United States in the period 2007–2011 to document the efficacy and safety of irrigated uni- and bipolar radiofrequency ablation for treating atrial fibrillation according to the CMIV protocol during concomitant open heart surgery. After 6–9 months, the results showed that 66 % of a total of 150 patients were free of atrial tachyarrhythmias (5).
Surgical ablation of atrial fibrillation is now regarded as a class I recommendation for selected patients (1). It is therefore important to decide whether patients with atrial fibrillation should be offered surgical ablation in connection with planned cardiac surgery. The short- and long-term results for patients who took part in the Norwegian part of the CURE-AF trial are presented in this article.
Material and method
Inclusion and early follow-up
Two Norwegian cardiac surgery centres took part in the CURE-AF trial in the period 2008–2010. Nineteen patients (ten in Trondheim and nine in Bergen) were included, underwent surgery and were followed up according to the CURE-AF protocol by the same cardiac surgeon at each centre. Data from the Norwegian part of the trial were not included in the report by Damiano et al. (5). Both the primary CURE-AF trial in Norway (European Companion Protocol to the US CURE-AF study, IDE G060220/S4 Permanent AF Rev E and IDE G070046/S1 Persistent AF Rev B) and the subsequent long-term follow-up were regarded as quality assurance projects by the Regional Committee for Medical and Health Research Ethics (REK) West (067.08 and 2015/257). The CURE-AF study was registered in 2007 (NCT00431834). The European arm of the study was registered retrospectively on 27 November 2020 in ClinicalTrials.gov (NCT04645615).
Patients intended for elective cardiac surgery on ordinary indications and who in addition had atrial fibrillation and met the inclusion criteria for the CURE-AF trial were included successively. Transthoracic echocardiography was used by a cardiologist to measure left ventricular function and the diameter of the left atrium. Patients were given detailed information about the risks and benefits of participating in the trial, and they signed an informed consent form.
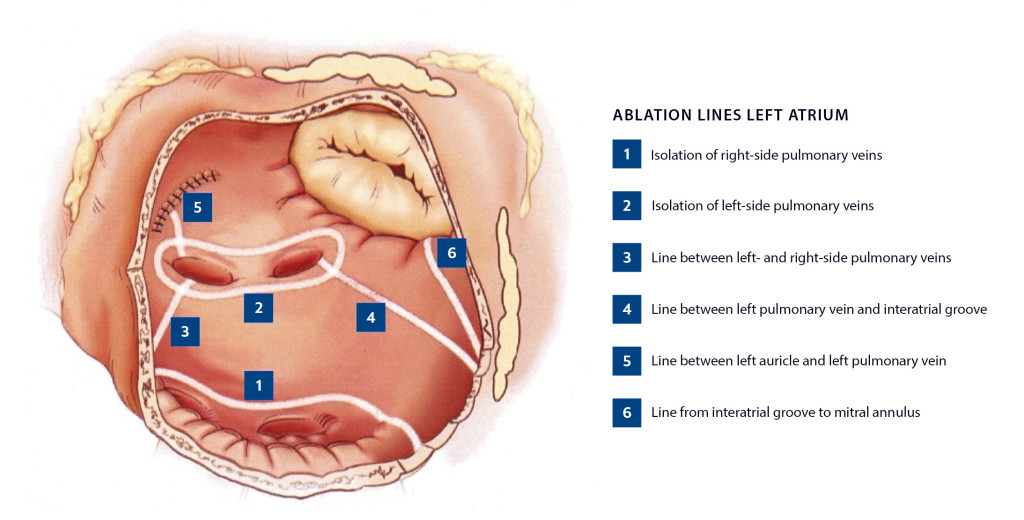
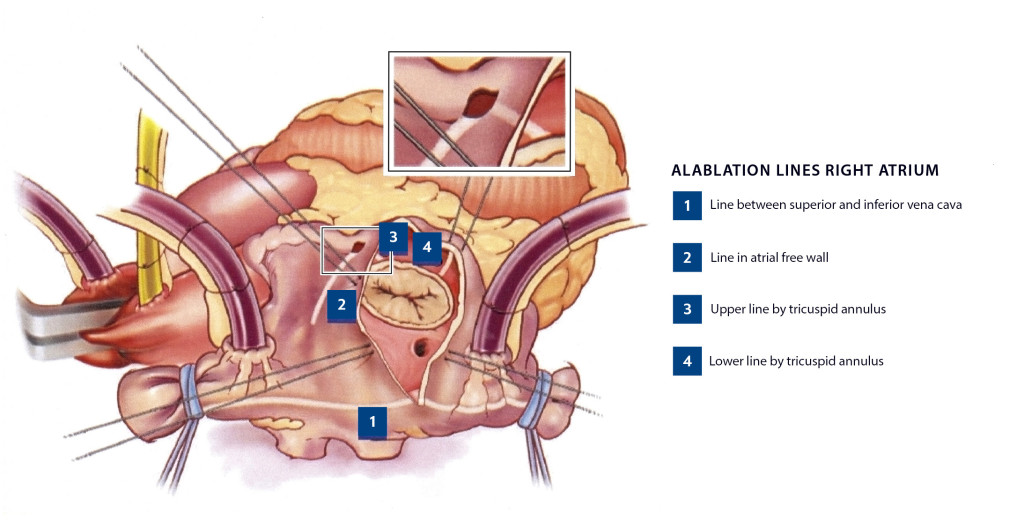
The ablations were performed using bipolar equipment (Cardioblate LP, Medtronic, Minneapolis, MN, USA) and/or unipolar equipment (Cardioblate Monopolar Pen, Medtronic) linked to a radio-frequency generator (Cardioblate 68000 RF Generator, Medtronic). During ablation, the tissue was cooled with saline infusion. The ablation lines are visualised schematically in Figures 1 and 2. All patients had a full set of ablation lines applied, irrespective of the type of atrial fibrillation and principal surgical intervention. Bipolar equipment was used for pulmonary vein ablation and the line between the superior and inferior vena cava. The other lines were generated using combinations of unipolar and bipolar equipment. Pulmonary vein ablation was performed on the beating heart (before cardioplegia). A primary conduction block between the pulmonary veins and the left atrium was achieved in 16 patients, while three needed more ablations in order to achieve a block. In addition the left auricle was amputated (n = 9) or ligated/closed with sutures (n = 8) or a suture machine (n = 2).
Medication with amiodarone commenced within a few days postoperatively and continued (200 mg daily) for three months. Eighteen of 19 patients were already using warfarin before the procedure. According to the CURE-AF protocol, warfarin was to be used for at least six months after the intervention. However, warfarin was found to have been continued on clinical indication in 17 of 18 patients until the end of the study.
The term permanent atrial fibrillation should not be used for patients who undergo surgical ablation (6). In this study, the results will be presented collectively for the whole group and separately for the two sub-groups persistent atrial fibrillation (duration > 7 days or requiring electroconversion) and long-standing persistent atrial fibrillation (duration > 12 months) where relevant. The endpoint for rhythm status in the CURE-AF trial, documented by means of 24-hour Holter monitoring, was six months for long-standing persistent atrial fibrillation and nine months for persistent atrial fibrillation. At these points in time, class I and class III antiarrhythmic agents had been discontinued. Follow-up was then gradually transferred to general practitioners (GPs) and outpatient clinics.
Long-term follow-up
One patient died a few months after the procedure. The remaining 18 were followed for 5–6 years after the procedure with clinical examination, detailed history of heart rhythm and 12-lead ECG. Patient records, including all ECG records, were obtained from GPs, cardiologists and local hospitals.
In the period after the Holter monitoring (6–9 months postoperatively), two patients in the group with persistent atrial fibrillation had catheter ablation performed 11 months and 65 months, respectively, after the surgical procedure. They subsequently maintained sinus rhythm. Two patients in the group with long-standing persistent atrial fibrillation had permanent pacemakers implanted. During the follow-up examination, some patients were found to have already developed chronic atrial fibrillation, while in others evidence was found of recurrence that needed further diagnostics and treatment. This was initiated in the course of autumn/winter 2015. A final summing up of rhythm status was made in December 2015.
Statistics
Data are presented as median values ± standard deviation for normally distributed data, otherwise as medians (1st quartile and 3rd quartile). The two-sample t-test and Mann-Whitney U-test for continuous variables and Fisher's exact test for nominal data were used to compare the data for the groups with long-standing persistent atrial fibrillation and persistent atrial fibrillation.
Results
Preoperative data are presented in Table 1. With the exception of median duration of atrial fibrillation, which was longer in the group with long-standing persistent atrial fibrillation compared with the persistent atrial fibrillation group (40 versus 6 months), there were no statistically significant differences between the two groups. The median age was 72 years, and the majority of the patients were in New York Heart Association (NYHA) class II (mild symptoms). Left ventricular function was normal. The prevalence of coronary disease was the same in both groups. Almost all (18/19) had undergone anticoagulation therapy with warfarin, and 13 were on beta-blocker treatment.
Table 1
Preoperative characteristics of a patient population (n = 19) who had surgical ablation for atrial fibrillation during concomitant open heart surgery in Bergen or Trondheim in 2008–2010. Number unless otherwise specified
| Variable | Data | |
|---|---|---|
| Age (years), median (1st quartile; 3rd quartile) | 72 (68; 73) | |
| Sex (no. of men/women) | 15/4 | |
| Hypertension | 3 | |
| Duration atrial fibrillation (months), median (1st quartile; 3rd quartile) | 20 (9; 53) | |
| Beta-blocker | 13 | |
| Warfarin | 18 | |
| NYHA1 class, no. of patients | ||
| Class I | 1 | |
| Class II | 12 | |
| Class III | 6 | |
| Left ventricular ejection fraction (per cent), mean value ± SD | 62 ± 5 | |
| Diameter left atrium (mm), mean value ± SD | 53 ± 8 | |
| Surgical conditions2 | ||
| Mitral insufficiency | 10 | |
| Coronary disease | 9 | |
| Aortic valve stenosis | 3 | |
1 New York Heart Association
2 Some patients had several conditions requiring surgery
The main procedures were mitral valve surgery (n = 10), aortocoronary bypass procedure (n = 6) and aortic valve prosthesis (n = 3) (Table 2). Mitral valvuloplasty was carried out significantly more frequently (p = 0.01) in the group with persistent atrial fibrillation. The left internal mammary artery was used as a bypass in eight of nine who underwent coronary surgery. In the group with long-standing persistent atrial fibrillation, five patients also underwent secondary procedures (closure of foramen ovale, resection of aneurism in ascending aorta, tricuspid valvuloplasty). Median ablation time was 379 seconds. There was no difference in ablation time between the groups, but considerable variation among individual patients. Peri- and postoperative complications occurred in seven patients (37 %), none clearly related to the CMIV procedure. The median stay in hospital was eight days, and there was no mortality within 30 days.
Table 2
Peri- and postoperative data on a patient population (n = 19) who had surgical ablation performed for atrial fibrillation in connection with open heart surgery in Bergen or Trondheim in 2008–2010. Number unless otherwise specified.
| Variable | Data | |
|---|---|---|
| Main procedure | ||
| Mitral valve procedures, total | 10 | |
| Reconstruction/ring | 6 | |
| Prosthesis | 4 | |
| Aortic valve prosthesis | 3 | |
| Coronary bypass surgery1 | 6 | |
| Cardiopulmonary bypass time (min), mean ± SD | 192 ± 71 | |
| Aortic clamping (min), mean ± SD | 129 ± 65 | |
| Total radiofrequency ablation time (sec), median (1st quartile; 3rd quartile) | 379 (325; 552) | |
| Hospital stay (days), median (1st quartile; 3rd quartile) | 8 (5; 9) | |
1 Coronary bypass surgery was performed in a total of nine patients, in three as part of a combined procedure
Upon discharge, 58 % (11/19) had sinus rhythm; 5 of 7 in the group with persistent atrial fibrillation and 6 of 12 in the group with long-standing persistent atrial fibrillation (p = 0.63) (Figure 3 a). Over the following 6–9 months, 14 of 19 patients (74 %) were electroconverted because of persistent or recurring supraventricular tachycardias (SVT).
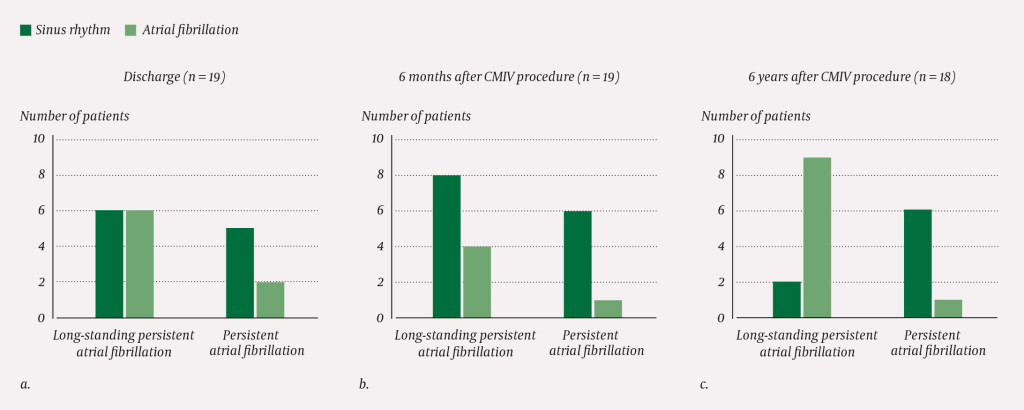
Six months after surgery (Figure 3b), 14/19 patients (74 %) had sinus rhythm; 8 of 12 in the group with long-standing persistent atrial fibrillation and 6 of 7 in the group with persistent atrial fibrillation (p = 0.60).
Long-term follow-up
When the patients were examined after five years, 12 of 18 proved to have atrial fibrillation. Nine of these 12 were in sinus rhythm 6 months postoperatively, while three were in atrial fibrillation. Six patients, all in the group with persistent atrial fibrillation, had sinus rhythm.
Seven patients with recurrence were assessed as needing further arrhythmia diagnostics and possibly electroconversion. During the observation period in autumn 2015, four of these seven proved to have paroxysmal atrial fibrillation. Atrial flutter with 2:1 conduction was found in two patients in the group with long-standing persistent atrial fibrillation. These two were successfully electroconverted, and retained sinus rhythm to the end of the survey. One patient was referred for electroconversion, but the cardiologist judged that it was not indicated.
The final result for the whole population (Figure 3c) was restored sinus rhythm in 44 % (8/18), with a significant difference (p = 0.013) between the group with persistent atrial fibrillation (6/7) and the group with long-standing persistent atrial fibrillation (2/11).
Discussion
Atrial fibrillation has been found to be present in almost one third of patients with heart disease requiring cardiac surgery in the three years prior to surgery (1). Surgical treatment of atrial fibrillation during concomitant cardiac surgery is today a class I recommendation for symptomatic patients with atrial fibrillation of both short and long standing (6). Cardiologists and cardiac surgeons should therefore decide whether concomitant ablation is to be performed during cardiac surgery.
Our study supports radiofrequency ablation for atrial fibrillation according to the CMIV protocol as a safe procedure with few complications. In the short term, and with follow-up according to a standard monitoring regime, the procedure can be expected to restore sinus rhythm in more than 2/3 of patients. These findings are consistent with the results of the CURE-AF trial published by Damiano et al. (5). They found that the number of patients with sinus rhythm (without supraventricular tachycardia) on Holter monitoring after 6–9 months (our figures in parentheses) was 66 % (74 %) for the whole group, 63 % (67 %) for the group with long-standing persistent atrial fibrillation and 82 % (86 %) for the group with persistent atrial fibrillation. The patient population that was studied and followed up in this trial is small, and we had no control group to verify the benefit of the procedure. However, the patients were closely followed up for a long time after the procedure. Although the execution of the study and the follow-up survey took place a while back, neither indications, interventional method nor follow-up of patients who have undergone surgical ablation have changed substantially. The results are therefore still regarded as relevant.
Increased left atrial diameter, short ablation time and number of additional procedures have all been found to increase the risk of recurrence of atrial fibrillation (5). We were unable to confirm these relationships, probably because of the selection criteria for the study and the small population size. A complete ablation pattern with transmural lesions is a prerequisite for a good result in all ablation for atrial fibrillation (7). Pulmonary vein ablation was conducted with bipolar equipment, and transmurality was confirmed by blocked impulse conduction between pulmonary veins and left atrium (exit block). We found that repeated ablation procedures were necessary in some patients to achieve exit block. Even where this was documented, one patient was found to experience recurrence after six months in the form of atrial flutter from an ablated pulmonary vein. Short ablation time (< 360 seconds) has proved to be a risk factor for recurrence (5). The median ablation time in our study (379 seconds) was higher than this, but varied substantially across patients. Incomplete ablation can probably explain some of the recurrences in this study.
Long duration of preoperative atrial fibrillation is a risk factor for recurrence of atrial fibrillation (8), which is confirmed in our study by significantly different recurrence rates between the groups with long-standing persistent atrial fibrillation and persistent atrial fibrillation. Long-term results after a CMIV procedure for atrial fibrillation show considerable variation. The CURE-AF trial in the United States was stopped after nine months, and to the best of our knowledge follow-up data have not been published. We investigated heart rhythm five years after the procedure and found a high rate of atrial fibrillation recurrence, particularly in the group with long-standing persistent atrial fibrillation. The recurrences often generated limited symptoms, and were not always picked up early enough by the primary health service (data not shown).
At the end of the study, after 5–6 years, 44 % of our patients had sinus rhythm. This is considerably lower than reported in two recent studies from the United States, where 66 % and 64 % had sinus rhythm five years after a similar procedure (8, 9). In both these studies, the patients were closely followed up and appropriately treated for recurrence of atrial fibrillation in the years following the procedure. Since early results in our study were in line with those of other centres, we suspect that one important cause of the high recurrence rate is deficiencies in follow-up and electroconversion, particularly in the group with long-standing persistent atrial fibrillation. This is supported by findings in a Danish study, where follow-up was also left to the primary health service, and where only 33 % and 25 % of the patients were without supraventricular tachycardia after five and seven years, respectively (10).
The recurrence rate has proved to be substantial after 18–36 months (9), which again underscores the importance of long-term follow-up and prompt intervention at a specialist centre in the event of recurrence of atrial fibrillation (9, 10). This emerged clearly during the follow-up survey in our study, as over a third of the patients (7/18) proved to need further arrhythmia diagnostics. In addition, some patients with long-standing persistent atrial fibrillation had already developed permanent atrial fibrillation during the first few years after the operation.
In order to improve the long-term results after ablation for atrial fibrillation concomitant with heart surgery, it seems reasonable to recommend a regular follow-up programme, for example with an annual ECG and sometimes 24-hour ECG monitoring, allowing recurrences to be revealed earlier and adequate treatment initiated.
The article has been peer-reviewed.
Retrospective registration of the European arm of the study took place in connection with the preparation of the present article. The editorial board has no reason to believe that this has influenced the reporting of the results.
- 1.
McCarthy PM, Davidson CJ, Kruse J et al. Prevalence of atrial fibrillation before cardiac surgery and factors associated with concomitant ablation. J Thorac Cardiovasc Surg 2020; 159: 2245–2253.e15. [PubMed][CrossRef]
- 2.
Damiano RJ, Schwartz FH, Bailey MS et al. The Cox maze IV procedure: predictors of late recurrence. J Thorac Cardiovasc Surg 2011; 141: 113–21. [PubMed][CrossRef]
- 3.
Dunning J, Nagendran M, Alfieri OR et al. Guideline for the surgical treatment of atrial fibrillation. Eur J Cardiothorac Surg 2013; 44: 777–91. [PubMed][CrossRef]
- 4.
Musharbash FN, Schill MR, Sinn LA et al. Performance of the Cox-maze IV procedure is associated with improved long-term survival in patients with atrial fibrillation undergoing cardiac surgery. J Thorac Cardiovasc Surg 2018; 155: 159–70. [PubMed][CrossRef]
- 5.
Damiano RJ, Badhwar V, Acker MA et al. The CURE-AF trial: a prospective, multicenter trial of irrigated radiofrequency ablation for the treatment of persistent atrial fibrillation during concomitant cardiac surgery. Heart Rhythm 2014; 11: 39–45. [PubMed][CrossRef]
- 6.
Calkins H, Hindricks G, Cappato R et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm 2017; 14: e275–444. [PubMed][CrossRef]
- 7.
Gaita F, Ebrille E, Scaglione M et al. Very long-term results of surgical and transcatheter ablation of long-standing persistent atrial fibrillation. Ann Thorac Surg 2013; 96: 1273–8. [PubMed][CrossRef]
- 8.
Henn MC, Lancaster TS, Miller JR et al. Late outcomes after the Cox maze IV procedure for atrial fibrillation. J Thorac Cardiovasc Surg 2015; 150: 1168–76, 1178.e1–2.
- 9.
Ad N, Holmes SD, Stone LE et al. Rhythm course over 5 years following surgical ablation for atrial fibrillation. Eur J Cardiothorac Surg 2015; 47: 52–8, discussion 58. [PubMed][CrossRef]
- 10.
Engelsgaard CS, Pedersen KB, Riber LP et al. The long-term efficacy of concomitant maze IV surgery in patients with atrial fibrillation. Int J Cardiol Heart Vasc 2018; 19: 20–6. [PubMed][CrossRef]