Main findings
The mortality rate among the first 22 patients to receive active treatment for COVID-19 in the intensive care department at Oslo University Hospital, Ullevål, is 9 %.
Half of the patients did not undergo intubation or mechanical ventilation, even though they had severe hypoxaemia and symptoms of acute respiratory distress.
The COVID-19 pandemic has placed a great strain on the Norwegian health care system. A number of patients have been hospitalised and the most severely ill have been treated in intensive care units. It is primarily when the disease attacks the lungs and the patient develops severe hypoxaemia that treatment in intensive care with ventilatory support becomes relevant. Early international reports described the rapid development of acute respiratory distress syndrome (ARDS) requiring early intubation and invasive mechanical ventilation (1, 2). Acute respiratory distress syndrome is defined according to the Berlin criteria on the basis of the degree of hypoxaemia measured in terms of the PaO2/FiO2 ratio with positive end-expiratory pressure (PEEP) > 5 cm H2O, acute onset and bilateral pulmonary opacities, and respiratory failure that cannot be explained by heart failure or hydrostatic oedema alone (3). Prolonged and complex lung-protective mechanical ventilation was described for COVID-19 patients, with use of high PEEP of up to 14–16 cm H2O (2).
Supplementary oxygen can also be administered to hypoxaemic patients by mask alone or in combination with non-invasive ventilation (NIV). However, non-invasive ventilation has previously proven to be of no benefit and to result in more complications in similar patients (4). The same experience was reported by Italian clinicians in their COVID-19 patients (2). There is currently no consensus regarding either the benefit or the risk of aerosol generation associated with the use of non-invasive ventilation or nasal high flow oxygen in COVID-19 patients (5, 6). Moreover, there is debate as to whether all hypoxaemic patients with COVID-19 pneumonia develop acute respiratory distress syndrome (7).
In the light of new findings, we will share our experiences of managing the critically ill COVID-19 patients who were admitted to our intensive care department. Our aim was to survey clinical status, treatment and outcomes among intensive care patients who received treatment with and without invasive mechanical ventilation.
Material and method
Oslo University Hospital, Ullevål is a local hospital for several boroughs in Oslo, as well as a regional hospital in the South-Eastern Norway Regional Health Authority. The hospital has about 1 200 beds, with approximately 45 000 admissions and more than 300 000 outpatient consultations each year, and is the country's largest acute care hospital and trauma centre. There are normally 33 adult intensive care beds with ventilator capacity available, distributed among six intensive care and observational units, but it is not unusual for capacity to be expanded due to high levels of activity.
In anticipation of a large number of intensive care patients during the pandemic, three intensive care units were established in early March 2020 that were ready to accommodate a total of 33 COVID-19 patients requiring intensive care, as well as four negative pressure isolation rooms that were reserved for procedures with an especially high risk of aerosol generation. An intermediate unit was also established, and several of the wards in the medical department served exclusively as COVID-19 wards. Here, up to 15 litres of oxygen were available via non-rebreather masks, while in the intensive care units, non-invasive ventilation was available in addition. On the wards, patients were monitored using the National Early Warning Score (NEWS) (8), up to 2–3 times per nursing shift. There were daily meetings between ward and intensive care clinicians to assess whether patients should be transferred to intensive care or to the intermediate unit. Patients who did not respond satisfactorily to oxygen therapy and who had increasing signs of fatigue/exhaustion with increased respiratory rate and tachycardia were admitted to intensive care. There were no universal criteria for transfer, but the decision was made by the clinician on duty on the ward or in Acute Admissions and by the attending intensive care clinician.
To oversee the treatment of patients in intensive care, new teams were set up comprising specialists in intensive care medicine and anaesthesia from the Division of Critical Care and the Department of Acute Medicine. Treatment was to primarily follow the guidelines for acute respiratory distress syndrome, with lung-protective ventilation with low tidal volume (4–6 ml/kg predicted body weight), plateau pressure <30 cm H2O, driving pressure <15 cm H2O and PEEP >5 cm H2O. In addition, there was to be deep sedation, neuromuscular blockade if necessary, restrictive fluid therapy and a low threshold for prone positioning (9, 10). However, we also allowed scope for individual adjustments. This meant that some patients could be offered non-invasive ventilation if indicated, with PEEP 5–8 cm H20, but with the lowest possible pressure support according to need and response. Patients who did not respond satisfactorily to oxygen were thus candidates for NIV, with the decision made by the intensive care clinician. The criteria for intubation were signs of exhaustion following prolonged hypoxaemia (rapid and increasing respiratory rate, tachycardia, subjective feelings of fatigue, decreasing PaO2 despite increasing oxygen supply, increasing PaCO2).
Data collection and presentation
All patients admitted to Oslo University Hospital with COVID-19 are registered on an ongoing basis in a local quality register as well as in the Norwegian Pandemic Registry. The internal quality register, as well as this publication of data from that register, were approved by the local Data Protection Officer (case number 20/07119). A description of the disease course and treatment was provided by the internal quality register for all patients admitted to the intensive care department at Ullevål in the period 5 March–28 May 2020. Three of the co-authors (TMO, KT, ARH) were responsible for this register and have full access to the data.
The internal quality register includes the parameters delivered to the Norwegian Pandemic Registry in addition to further information on comorbidities, daily information on patient status (blood pressure, heart rate, oxygen saturation, mental status), treatment (antimicrobial therapy, and organ support therapy such as respiratory and circulatory support) as well as biochemistry and radiology results. The time of admission and of any transfers between departments are recorded, with each day counted from midnight to midnight.
A summary of parameters related to status on arrival, intensive care treatment of respiratory failure, and outcomes was used in this publication. In addition to well-known clinical parameters, several scoring tools were also used. For general characterisation of the patient, we used the Charlson Comorbidity Index (predicts mortality on the basis of comorbidity, 0–37 points) and the Clinical Frailty Scale (predicts degree of frailty, from very well (1 point) to terminally ill (9 points), on the basis of cognitive status, mobility and need for assistance immediately prior to the acute illness). As a measure of physiological status upon hospitalisation in Acute Admissions, we used NEWS (respiratory rate, peripheral oxygen saturation, systolic blood pressure, pulse rate, level of consciousness/confusion, and temperature all scored from 1 to 3 points; NEWS ≥ 5 indicates severe acute illness) and the Quick Sequential Organ Failure Assessment (qSOFA) (one point each for respiratory rate ≥ 22/min, systolic blood pressure ≤ 100 mm Hg and altered mental status as revealed by the Glasgow Coma Scale (GCS) ≤ 15; qSOFA 2 and 3 indicate serious illness). The SOFA score includes the most abnormal value for each of circulation, respiration, renal function, mental status (measured with the GCS), coagulation, and liver function, and was used in this study to characterise disease severity upon arrival in Acute Admissions, as well as the worst total score at any point during the hospital stay.
Only patients who were admitted to the intensive care units were registered as intensive care patients and hence included in the study. The patients have been divided into three groups based on the treatment of their respiratory failure (oxygen alone, addition of non-invasive ventilation, and intubation with mechanical ventilation) and described using descriptive statistics (numbers with percentages, and means with minimum and maximum values). No statistical comparison of the groups was performed.
Results
During the period in question, 165 COVID-19 patients were admitted to Oslo University Hospital, Ullevål, of whom 26 (16 %) were treated in an intensive care unit. Four of these patients had treatment limitations owing to significant comorbidities, advanced age and frailty. They were therefore not candidates for invasive mechanical ventilation and have been excluded from the study (Figure 1). Of the 22 patients included, 11 received mechanical ventilation (two of whom had initially received non-invasive ventilation), 7 received oxygen alone and 4 received oxygen plus non-invasive ventilation (Figure 1, Table 1). The average age was 56 years (range 25–78 years), and 17 (77 %) were men. Overall, the patients had few comorbidities, with a mean Charlson Comorbidity Index of 1 (range 0–3), but nine of the patients had heart disease/hypertension. The mean body mass index was 30–35 in all groups (Table 1).
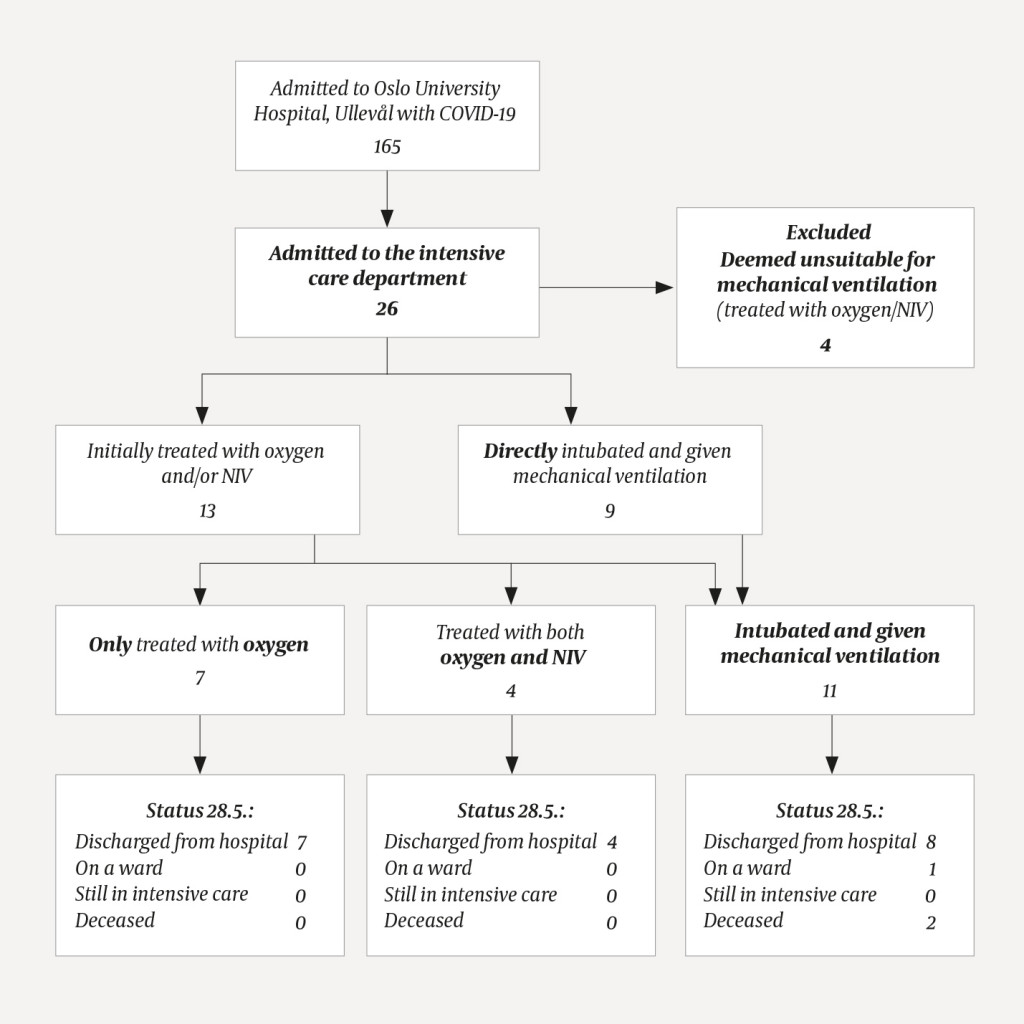
Table 1
COVID-19 patients who received active treatment in the intensive care department, Oslo University Hospital, Ullevål in the period 5 March–28 May 2020. Patient characteristics after treatment. Continuous variables are presented as mean (range). For categorical variables, the number in each group is shown. SOFA = Sequential Organ Failure Assessment, qSOFA = Quick-SOFA, NEWS = National Early Warning Score, SpO2 = oxygen saturation, PaO2 = partial pressure of oxygen in arterial blood, FiO2 = fraction of inspired oxygen.
| All (N = 22) | Oxygen (n = 7) | Non-invasive ventilation (n = 4) | Mechanical ventilation (n = 11) | ||
|---|---|---|---|---|---|
| Patient characteristics | |||||
| Age (years) | 59 (25–78) | 52 (25–76) | 70 (55–78) | 58 (43–74) | |
| Male | 17 | 5 | 4 | 8 | |
| Body mass index (kg/m2)1 (overweight > 25, obesity > 30) | 30 (24–51) | 32 (25–51) | 31 (27–36) | 30 (24–35) | |
| Charlson Comorbidity Index (0–37 points) | 1 (0–4) | 1 (0–4) | 2 (0–4) | 1 (0–3) | |
| Clinical Frailty Scale (1–9 points) | 3 (2–4) | 3 (2–4) | 3 (2–4) | 3 (2–4) | |
| Status in Acute Admissions | |||||
| SOFA (0–24 points) | 3 (0–7) | 3 (0–7) | 3 (2–3) | 4 (1–7) | |
| qSOFA (0–3 points) | 1 (0–2) | 1 (0–2) | 1 (1–1) | 1 (0–2) | |
| NEWS (0–20 points) | 8 (1–14) | 8 (3–14) | 9 (8–11) | 9 (1–13) | |
| Altered mental status (number of patients with change on Glasgow Coma Scale, GSC) | 5 | 3 | 0 | 2 | |
| Mean arterial pressure (MAP) (mm Hg) | 96 (68–132) | 93 (73–104) | 96 (68–132) | 98 (74–118) | |
| Tympanic temperature (°C) | 38.3 (36.0–40.3) | 38.1 (36.0–39.4) | 38.8 (37.7–39.5) | 38.2 (36.4–40.3) | |
| Respiratory rate (12–16 breaths/min) | 33 (20–52) | 29 (20–40) | 35 (23–45) | 36 (20–52) | |
| SpO26 (95–99 %) | 87 (56–100) | 88 (56–100) | 90 (86–95) | 85 (57–98) | |
| PaO27 (10.5–14 kPa) | 8.0 (4.0–12.8) | 7.6 (4.0–11.8) | 9.1 (8.4–10.1) | 7.9 (5.0–12.8) | |
| PaO2/FiO28-ratio2 (55–65 kPa) | 26 (6–56) | 32 (10–56) | 35 (21–42) | 21 (6–43) | |
| CRP (< 5 mg/l) | 164 (44–483) | 143 (44–428) | 139 (86–194) | 188 (47–483) | |
| Ferritin (30–400 µg/l)1 | 1 298 (163–4 994) | 1 834 (163–4 994) | 717 (178–1 432) | 1 167 (512–2 821) | |
| D-dimer (mg/L FEU)1 (age-adjusted decision thresholds) | 1.7 (0.2– > 4) | 1.7 (0.2– > 4) | 1.6 (0.7–2.6) | 2.0 (0.5– > 4) | |
| Leukocytes (3.5–10 · 109/l) | 8.8 (3.1–17.2) | 7.1 (3.1–14.4) | 8.1 (5.7–11.3) | 10.3 (5.0–17.2) | |
| Lymphocytes (1.1–3.3 · 109/l)1 | 0.9 (0.5–1.8) | 0.9 (0.7–1.4) | 0.9 (0.5–1.5) | 0.9 (0.6–1.8) | |
| Lactate (0.5–2.2 mmol/l) | 1.7 (0.7–5.4) | 1.8 (0.7–4.7) | 1.1 (1.0–1.2) | 1.9 (0.9–5.4) | |
| Treatment and outcome | |||||
| Worst SOFA score1 (0–24 points) | 6 (2–10) | 4 (2–6) | 7 (4–9) | 8 (6–10) | |
| Time from admission to treatment in intensive care (days) | 2 (0–4) | 2 (1–3) | 3 (2–4) | 1 (0–3) | |
| Time from admission to mechanical ventilation (days) | − | − | − | 1 (0–4) | |
| Time on ventilator (days) | − | − | − | 22 (9–49) | |
| Time in intensive care (days) | 15 (2–56) | 4 (2–8) | 5 (2–7) | 25 (10–56) | |
| Time in hospital (days) | 23 (6–65) | 14 (6–24) | 15 (12–18) | 32 (17–65) | |
| Discharged alive from intensive care | 203 | 7 | 4 | 9 3 | |
1Data for body mass index are not available for three patients, and ferritin, D-dimer and lymphocytes for one patient
2The PaO2/FiO2 ratio is an expression of the patient's oxygen requirements
3One patient is still hospitalised (on a ward)
Of the eleven patients who received mechanical ventilation, two had died as of 28 May 2020. The others had been discharged alive from intensive care, and one of them was still on a ward. Four patients were tracheotomised. All patients who did not receive mechanical ventilation survived and have been discharged from the intensive care department. There have been no readmissions to intensive care. As of 28 May 2020, mortality was 9 % (2/22) among all intensive care patients who received active treatment, and 18 % (2/11) among patients who received mechanical ventilation (Figure 1, Table 1).
Those who were intubated and given mechanical ventilation seem to have had a lower PaO2/FiO2 ratio upon hospitalisation in Acute Admissions (Table 1). All were admitted to intensive care during the second week of illness, and the time from admission to treatment in intensive care and intubation was 0–4 days. The average length of stay in intensive care was 4 days for patients who received oxygen, 5 days for those who received NIV, and 25 days for those who received mechanical ventilation (Table 1). Nine out of eleven patients who received mechanical ventilation were placed in the prone position. None of the patients who received non-invasive ventilation were placed in the classic prone position, but frequent turning and mobilisation are part of the treatment for all intensive care patients when possible. The average duration of mechanical ventilation was 22 days (9–49). Both of the patients who died suffered multiorgan failure with haemodynamic instability.
Discussion
This report reveals a mortality rate of 9 % among the first 22 patients to receive active treatment for COVID-19 in the intensive care department at Oslo University Hospital, Ullevål. Half of the patients were not intubated or given mechanical ventilation, even though they had severe hypoxaemia and symptoms of acute respiratory failure according to the Berlin criteria (3). These patients all seem to have had good outcomes, with relatively short stays in the intensive care department. A strikingly high proportion of the patients in intensive care were overweight.
Gattinoni et al. discuss whether mechanical ventilation with a traditional approach to acute respiratory distress syndrome may in some cases do more harm than good for COVID-19 patients. They recommend a more personalised approach based on patient phenotype, and have therefore divided patients into two groups: phenotype L with little lung stiffness and almost normal compliance, and phenotype H with increasing oedema, reduced aerated lung volume and significant lung stiffness with low compliance (11, 12). In type L patients, severe hypoxaemia is the result of both hypoxic vasoconstriction and failure of perfusion autoregulation. The proportion of collapsed lung tissue is low and there is limited potential for recruitment. The use of mechanical ventilation with overly high PEEP therefore seems unnecessary for these patients (11, 12). Initial interventions for these patients may include increasing the oxygen supply via various mask-based systems or non-invasive ventilation, although this is controversial (2), (4–6).
The patients in our ward cohorts received up to 10–15 litres of oxygen by mask with careful monitoring of NEWS, which was scored several times per nursing shift (8), along with measurement of arterial blood gases when indicated. In addition to daily meetings, clinicians on the wards contacted their counterparts in intensive care when needed, and any patients who showed increasing respiratory distress/signs of exhaustion were transferred to intensive care. Nevertheless, several of the patients were able to manage with only supplementary oxygen delivered via various mask-based systems. We found that despite clinically and radiologically severe respiratory failure, it was possible to use intermittent non-invasive ventilation successfully and thereby avoid intubation in some motivated patients (2), (4–6).
It is important to emphasise the need for the continuous presence of experienced intensive care nurses around all intensive care patients, especially hypoxaemic patients who are breathing independently with the aid of an oxygen mask or non-invasive ventilation. Active mobilisation with frequent changes in position, physiotherapy and personalised pain management/sedation are other key components of this intensive care treatment. It must also be stressed that a patient who is breathing spontaneously but with high respiratory effort and pronounced use of the respiratory muscles may generate a significant negative pressure in the pleural cavity, with an ensuing high transpulmonary pressure. A high transpulmonary pressure increases strain on the lungs and risks exacerbating the patient's respiratory failure (patient self-inflicted lung injury, P-SILI) (12, 13). Patients must therefore be closely monitored with respect to the need for intubation.
In line with international experience, in the early stages of mechanical ventilation therapy we used low tidal volume, low plateau pressure, low driving pressure, occasionally high PEEP (up to 14–16 cm H2O) and frequent prone positioning (2, 5, 8)(8–10). This was despite the fact that the lungs of most patients had almost normal compliance (> 50 ml/cm H2O). We eventually found that PEEP could often be reduced to 8–10 cm H2O without compromising oxygenation or compliance. However, some patients required increasing or sustained high PEEP and repeated prone positioning to maintain an adequate level of oxygenation. It is thus conceivable that in some patients we observed a progression from phenotype L to H (11–13). These patients had extensive pulmonary changes on CT, a large proportion of non-ventilated lung tissue and thus a greater potential to benefit from higher PEEP and prone positioning.
The theories of Gattinoni et al. are based on experience with COVID-19 as well as previous research on acute respiratory distress (syndrome) (7), (11–13). In any case, their description of the two phenotypes is consistent with our observations. We therefore believe that in many cases, even with significant hypoxaemia, intubation can be delayed if supplementary oxygen alone, non-invasive ventilation or nasal high flow produce satisfactory clinical improvement and the patient is not exhausted. However, the need for intubation must be continuously assessed, and any mechanical ventilation that follows intubation should be tailored to the patient's pulmonary physiology and clinical condition.
This requires round-the-clock care by experienced intensive care personnel with an understanding of pulmonary physiology and of advanced, careful mechanical ventilation, and who can closely monitor compliance, tidal volume, plateau pressure and titration of PEEP. All of this combined may potentially reduce the risk of phenotype L developing into phenotype H and life-threatening hypoxaemia (11–13). However, distinguishing between these two phenotypes in an individual patient is not necessarily straightforward, and there will be a degree of overlap. CT scans may be helpful, and we recommend systematic measurement of static compliance in all patients receiving mechanical ventilation.
It is important to recognise that the extent of this disease and the pandemic means that treatment must be viewed as a dynamic process in which we are open to changing course and strategy based on our own and others' experiences and knowledge.
Limitations
The main limitation of this study is the small number of patients in total, all of whom were treated at a single hospital. Thus, the generalisability of the findings may be low. We have not discussed other specific treatments beyond the different strategies for ventilation and oxygenation. Interventions such as circulatory support, fluid therapy, thrombosis prophylaxis, antiviral/anti-infective therapy, immunomodulatory therapy, immune response, nutrition, sedation/use of muscle relaxants, and general treatment of multiple organ failure have not been described in detail. These may of course affect therapeutic outcomes. We have not included a detailed discussion of ventilator settings and target values either, beyond stating that we endeavoured to achieve lung-protective ventilation with an oxygen saturation (SaO2) of 88–92 %, and acceptance of elevated arterial CO2 and moderate acidosis. We mainly used pressure- or volume-controlled ventilation, with a transition to pressure support ventilation when indicated on the basis of clinical status
Unfortunately, measurements of lung compliance were not systematically recorded in patients who received mechanical ventilation. Control of mechanical ventilation based on measurement of transpulmonary pressure, and techniques for CO2 removal via dialysis are not routinely used at Ullevål. The mean D-dimer levels are probably too low, as high values such as > 4 mg/l FEU were recorded.
In light of personal privacy considerations and the small number of patients, we cannot provide detailed information on the management of or data on individual patients. Nor do we have data on the physical and mental functional levels of patients discharged from the hospital.
Conclusion
The survival rate among the 22 COVID-19 patients actively treated in intensive care was high, at 91 %. Many hypoxaemic COVID-19 patients who were breathing independently were able to recover with an increased oxygen supply and non-invasive ventilation.
The article has been peer-reviewed.
- 1.
Wu F, Zhao S, Yu B et al. A new coronavirus associated with human respiratory disease in China. Nature 2020; 579: 265–9. [PubMed][CrossRef]
- 2.
Grasselli G, Zangrillo A, Zanella A et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy. JAMA 2020; 323: 1574–81. [PubMed][CrossRef]
- 3.
Ranieri VM, Rubenfeld GD, Thompson BT et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012; 307: 2526–33. [PubMed]
- 4.
Alraddadi BM, Qushmaq I, Al-Hameed FM et al. Noninvasive ventilation in critically ill patients with the Middle East respiratory syndrome. Influenza Other Respir Viruses 2019; 13: 382–90. [PubMed][CrossRef]
- 5.
Alhazzani W, Møller MH, Arabi YM et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med 2020; 46: 854–87. [PubMed][CrossRef]
- 6.
World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected: interim guidance. https://apps.who.int/iris/handle/10665/330893 Accessed 13.3.2020.
- 7.
Gattinoni L, Chiumello D, Rossi S. COVID-19 pneumonia: ARDS or not? Crit Care 2020; 24: 154. [PubMed][CrossRef]
- 8.
Royal College of Physicians. National Early Warning Score (NEWS) – Standardising the Assessment of Acute-Illness Severity in the NHS. London: Royal College of Physicians, 2012.
- 9.
Claesson J, Freundlich M, Gunnarsson I et al. Scandinavian clinical practice guideline on mechanical ventilation in adults with the acute respiratory distress syndrome. Acta Anaesthesiol Scand 2015; 59: 286–97. [PubMed][CrossRef]
- 10.
Fan E, Del Sorbo L, Goligher EC et al. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: Mechanical Ventilation in Adult Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 2017; 195: 1253–63. [PubMed][CrossRef]
- 11.
Gattinoni L, Chiumello D, Caironi P et al. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med 2020; 46: 1099–102. [PubMed][CrossRef]
- 12.
Marini JJ, Gattinoni L. Management of COVID-19 respiratory distress. JAMA 2020; 323: 2329–30. [PubMed][CrossRef]
- 13.
Brochard L, Slutsky A, Pesenti A. Mechanical ventilation to minimize acute lung injury in acute respiratory failure. Am J Respir Crit Care Med 2017; 195: 438–42. [PubMed][CrossRef]
Gundem og medarbeidere gir her en god illustrasjon på hvorfor man ikke bør basere anbefalinger om behandlingsvalg på små observasjonsstudier. Av 26 pasienter ble 11 behandlet med respirator og 15 med oksygen og/eller non-invasiv ventilasjon (NIV). Fire fikk ikke tilbud om respiratorbehandling. Disse er tatt ut av den videre analysen. Av de resterende 22 døde 2 pasienter. Dette gir en dødelighet på 9 prosent. Et meget godt resultat.
Behandlingsbegrensninger er vanlige i intensivavdelinger, og mange regner dette som en forutsetning for å kunne drive etisk forsvarlig intensivmedisin (1). Det er derfor gledelig at våre kollegaer opplyser leserne om dette. Men det er vanlig å inkludere disse pasientene i observasjonsstudier der død er endepunktet. Dersom vi forutsetter at de fire pasientene som ikke ble inkludert overlevde intensivoppholdet blir dødeligheten 7,7 %, og dersom de døde 23 %. Dersom de døde, og regnes med i gruppen som kun fikk oksygen eller NIV, blir dødeligheten i denne gruppen 26 %. Slik kan vi fortsette.
Forfatterne konkluderer «at man i mange tilfeller, selv med betydelig hypoksemi, kan avvente intubasjon dersom tilført oksygen alene, ikke-invasiv ventilasjonsstøtte eller nasal high flow gir tilfredsstillende klinisk bedring og pasienten ikke er utmattet». Forbeholdene er mange.
Forfatterne omtaler internasjonale evidensbaserte retningslinjer for behandling av pasienter med alvorlig lungesviktsyndrom (ARDS) som «internasjonale erfaringer».(2) På denne måten visker de ut skillet mellom egen synsing og anbefalinger som bygger på randomiserte studier. Forfatterne støtter seg på synspunkter som tidlig i pandemien ble fremsatt av en av intensivmedisinens berømtheter, Luciano Gattinoni, og medarbeidere, og som senere er blitt kraftig imøtegått (3). Sen erkjennelse av at pasienter har ARDS er assosiert med forsinket etterlevelse av evidensbaserte anbefalinger og høyere dødelighet (4).
Dødeligheten til kritisk syke Covid-19-pasienter er lav i Norge, lavere enn mange fryktet, og lavere enn i mange andre land. Vi er enige med forfatterne i at innsatsen fra intensivsykepleierne er en nøkkelfaktor bak suksessen. Men vi tror og håper at respekt for internasjonale evidensbaserte retningslinjer også har spilt en viktig rolle.
Litteratur:
1. Sprung CL, Ricou B, Hartog CS et al. Changes in End-of-Life Practices in European Intensive Care Units From 1999 to 2016. JAMA 2019;322:1692-1704.
2. Claesson J, Freundlich M, Gunnarsson I et al. Scandinavian clinical practice guideline on mechanical ventilation in adults with the acute respiratory distress syndrome. Acta anaesthesiologica Scandinavica. 2015; 59:286–97.
3. Fan E, Beitler JR, Brochard L et al. COVID-19-associated acute respiratory distress syndrome: is a different approach to management warranted? Lancet Respir Med 2020.
4. Needham DM, Yang T, Dinglas VD et al. Timing of low tidal volume ventilation and intensive care unit mortality in acute respiratory distress syndrome. A prospective cohort study. Am J Respir Crit Care Med 2015;191:177–85.
Vi takker Laake og medforfattere for deres interesse, engasjement og tilbakemeldinger vedrørende våre nylig publiserte erfaringer med kritisk syke covid-19 pasienter innlagt på intensivavdelingen ved OUS Ullevål. Vi er helt enige i at behandlingsvalg skal basere seg på internasjonale kunnskapsbaserte retningslinjer der dette foreligger. Vårt ønske med artikkelen var kun å beskrive og dele våre erfaringer med denne nye sykdommen, hvor vi fulgte intensivtrengende pasienter gjennom forskjellige faser av covid-19. I førsteutkastet av artikkelen inkluderte vi resultater fra pasientene som var ekskludert fra studien, men disse ble fjernet i henhold til tilbakemeldinger fra Tidsskriftet. Vi er enige i at disse pasientene også må medberegnes i helhetsbildet vedrørende behandling og utkomme.