Here we describe pulmonary embolism in three patients at different stages of COVID-19. The development of pulmonary embolism can be easy to overlook if respiratory failure and hypoxemia are attributed to SARS-CoV-2 pneumonia.
The clinical picture associated with COVID-19 is complicated by severe acute phase reactions and widespread inflammation in the lungs. Pulmonary embolism can also occur in COVID-19 patients who receive thromboprophylaxis with low molecular weight heparin, and infection control considerations should not be an obstacle to essential diagnostic imaging.
Patient 1
A man in his seventies with type 2 diabetes was hospitalised after three days of fever with lower back and abdominal pain. Upon admission, he was found to have respiratory alkalosis and mild tachypnoea. A chest X-ray showed subtle opacities in basal areas of the right lung, suggestive of pneumonia. SARS-CoV-2 was detected in bronchial secretions. The patient remained tachypnoeic despite receiving supplemental O2 at 2 l/min. He was also given enoxaparin 40 mg × 1 as thromboprophylaxis. A chest X-ray on day 4 revealed increased opacities bilaterally, which had progressed further by day 8.
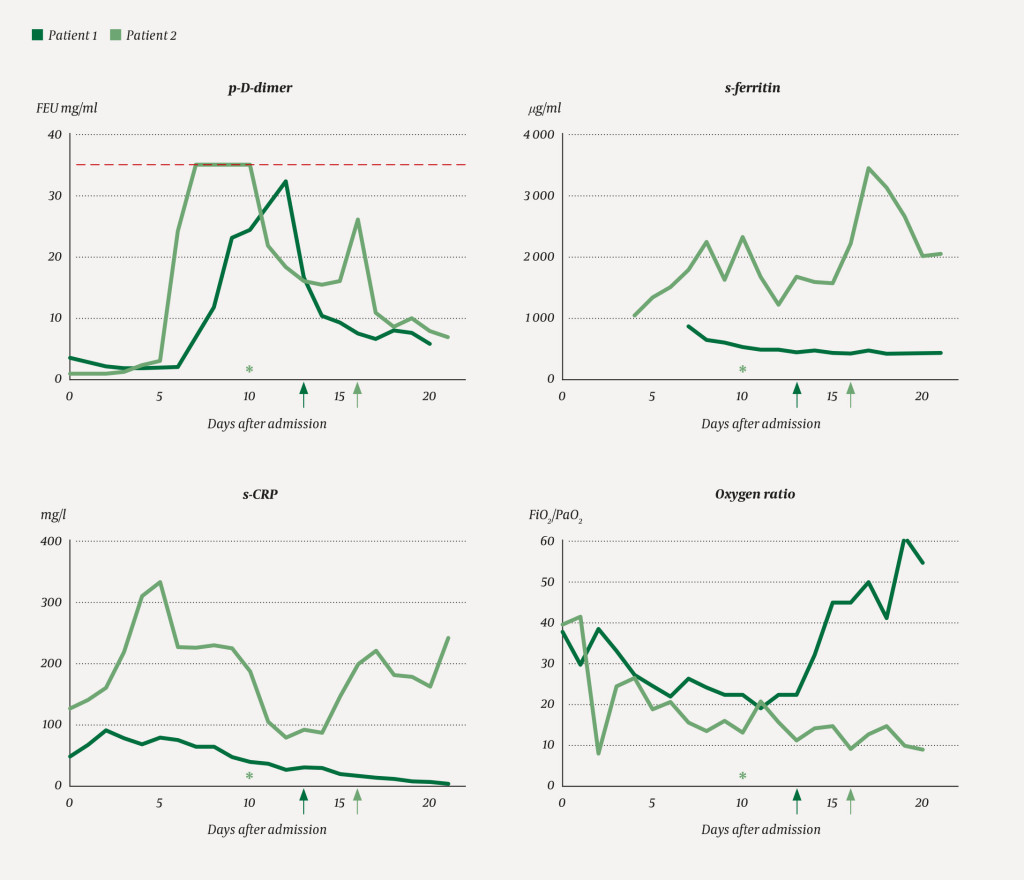
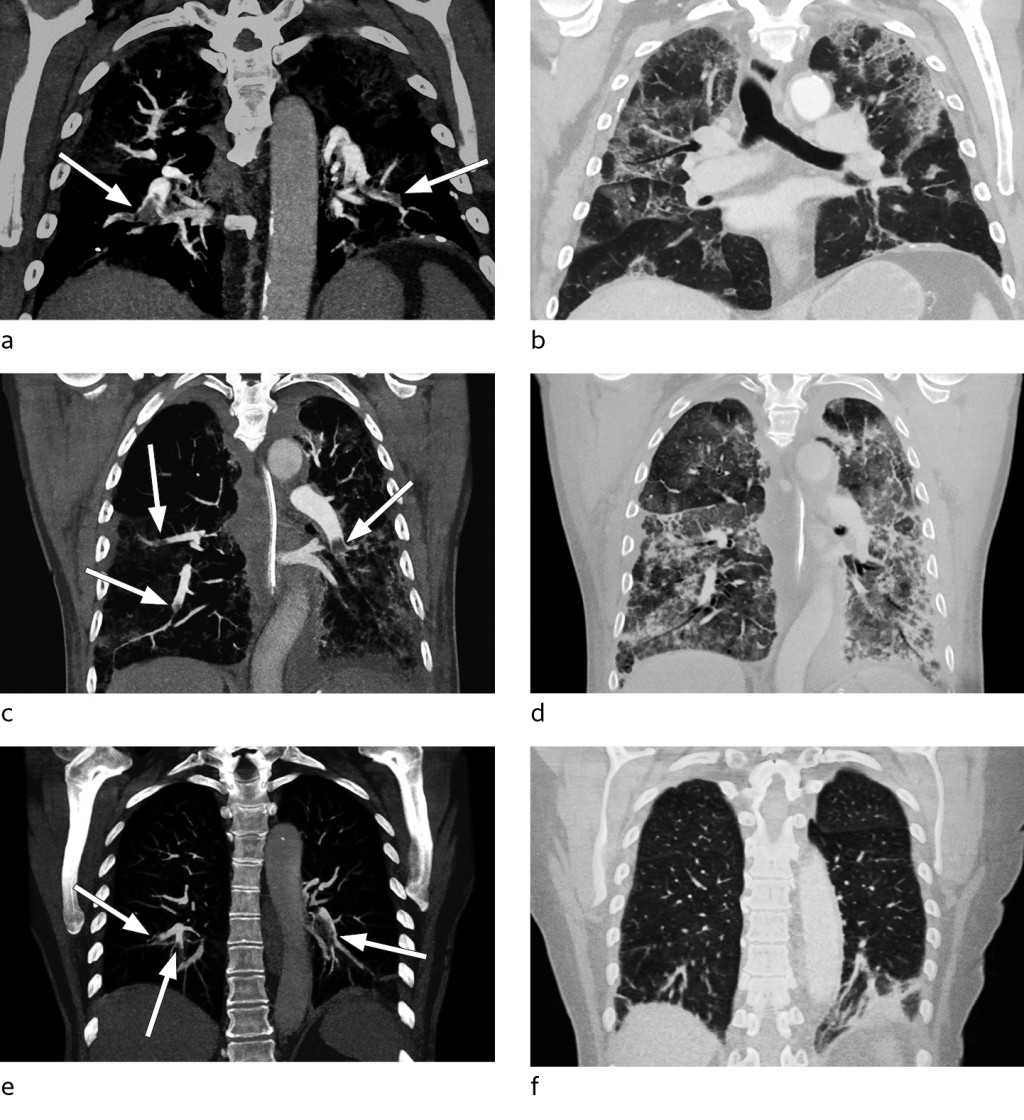
From day 9, the patient showed increasing oxygen demand, and required 10 l/min via a non-rebreathing mask to maintain SpO2 > 94 %. He was gradually becoming increasingly tired and weak. A chest X-ray on day 12 showed the lung opacities to be largely unchanged. Owing to a lack of improvement in oxygenation and a strong and sustained increase in D-dimer levels to > 10 mg/ml (Figure 1), CT pulmonary angiography was performed on day 15. This revealed bilateral segmental and subsegmental pulmonary embolisms, in addition to widespread infection-related opacities (Figures 2a–b). After initiation of anticoagulation therapy (enoxaparin 100 mg × 2), the patient's oxygen demand slowly decreased and his general condition rapidly improved.
Patient 2
A previously healthy man in his late seventies was hospitalised after three days of high fever, dry cough and reduced general condition. He was not dyspnoeic, but had hypoxemia with SpO2 90 % in room air. A chest X-ray upon admission was normal. SARS-CoV-2 was detected in bronchial secretions. He was given enoxaparin 40 mg × 1 as thromboprophylaxis. A further chest X-ray on day 1 revealed new bilateral opacities. By day 2, he was weaker and showed greater oxygen demand. He became tachypnoeic on day 3, and an X-ray showed progression of the pulmonary opacities. He was transferred to the intensive care unit and intubated.
Based on reports of increased incidence of venous thromboembolism in intensive care patients with COVID-19, we decided to increase the prophylactic dose of enoxaparin to 40 mg × 2. On day 7, the patient's D-dimer levels increased from 2.8 to 24 mg/l. His ferritin level was > 2 000 µg/ml and treatment with an interleukin-1 receptor antagonist (anakinra) was initiated on day 10. Radiological progression of the pulmonary opacities was seen up to day 13, while D-dimer levels decreased after initiation of anakinra (Figure 1). However, owing to a renewed increase in D-dimer from 16 mg/ml to 26 mg/ml, CT pulmonary angiography was performed on day 16. This revealed bilateral peripheral pulmonary embolisms and widespread opacities in all lobes (Figures 2c–d). Anticoagulation therapy (enoxaparin 100 mg × 2) was initiated, but after a long disease course with severe respiratory failure that did not improve, the patient died on day 22.
Patient 3
A previously healthy woman in her seventies experienced a week of illness with mild dry cough and upper respiratory tract symptoms. SARS-CoV-2 was detected in bronchial secretions after two days. After being free of symptoms for two weeks, the patient again developed a dry cough and was hospitalised 23 days after symptom onset, with activity-related dyspnoea and tachypnoea. Upon admission, she had respiratory alkalosis and a D-dimer level of 1.9 mg/ml. Chest X-ray was normal. CT pulmonary angiography was performed, and revealed bilateral segmental and subsegmental pulmonary embolisms affecting all lobes, but no opacities suggestive of infection (Figures 2e–f). She received oral anticoagulant therapy (apixaban 10 mg × 2) and was discharged when she showed improvement.
Discussion
These case studies demonstrate that pulmonary embolism may contribute to hypoxemia at various stages of the disease resulting from SARS-CoV-2 infection. It is difficult to pinpoint exactly when in the disease course this complication arose. In patients 1 and 2, high D-dimer levels were first attributed to a strong acute phase response to the infection (Figure 1). As the patients had been treated with low molecular weight heparin throughout their time in hospital, pulmonary embolism was not suspected until later in the disease course. Several studies have reported that high D-dimer levels (> 1.0 mg/ml) upon admission are associated with increased risk of death from COVID-19 (1–3), but the causal relationship is likely to be multifactorial.
In patient 2, a sharp rise in serum levels of C-reactive protein (CRP) and ferritin (Figure 1) was observed in the course of the disease. It has been reported that severe disease related to the coronavirus may be associated with the emergence of a hyperinflammatory state (cytokine storm) as part of the immune response to the virus (4), and there has been speculation over whether immunomodulatory therapy may be helpful in this regard. Given our patient's severe and persistent respiratory failure, we therefore decided to try immunomodulatory therapy with anakinra. After treatment initiation, we observed rapidly decreasing levels of D-dimer, CRP and ferritin (Figure 1), but no sign of any clinically significant improvement. This, in conjunction with a renewed increase in the D-dimer level, raised suspicion of pulmonary embolism.
In many patients COVID-19 leads to a prolonged disease course with high fever, reduced general condition and pulmonary involvement, all of which contribute to immobilisation. Treatment in intensive care in itself represents a significant risk factor for thromboembolic disease. Acute infections are associated with a significant but transient increase in the risk of venous thromboembolic events (5). Understanding of the pathophysiology of COVID-19-associated thromboembolic disease is still limited. Both the viral infection itself and the accompanying antiviral immune response entail a risk of vigorous activation of the coagulation system as a result of endothelial damage, platelet activation, and the release of potent proinflammatory cytokines (6). It is also suspected that endothelial damage may result in marked complement activation, thereby triggering a thrombotic microangiopathy similar to that seen in atypical haemolytic-uraemic syndrome (7). The high incidence of pulmonary embolism in cases of COVID-19 is presumably due to a combination of inflammation-mediated damage to pulmonary tissue (3) and systemic hypercoagulability.
Several publications have reported a strikingly high incidence of pulmonary embolism in COVID-19 patients. The condition has been detected in 20–30 % of patients in whom CT pulmonary angiography was performed on the basis of clinical indication (8–11). Biochemical and functional signs of hypercoagulability have been described in seriously ill COVID-19 patients and seem to be associated with a poor prognosis (1, 2, 8, 12, 13).
Among 184 intensive care patients in the Netherlands, 27 % had CT/ultrasound-confirmed venous thromboembolic events, 81 % of whom (25 patients) had pulmonary embolism (8). Deep vein thrombosis (DVT) was detected in one patient. Increased global coagulation parameters (INR, activated partial thromboplastin time) were predictors of thromboembolic complications. This suggests that coagulopathy may contribute to the development of pulmonary embolism in cases of COVID-19 (8). It is worth noting that these patients received thromboprophylaxis with low molecular weight heparin. However, the doses differed across the various hospitals in the study, and also increased over time according to the article. At one of the centres, the prophylactic dose was doubled over the course of the observation period to two daily doses, and the authors argue in light of the strikingly high incidence of pulmonary embolism that this practice must be considered for intensive care patients with COVID-19 (8).
In the Dutch study, diagnostic imaging was performed only on clinical suspicion, and the overall incidence of thromboembolic disease may therefore be even higher (8). In seven of 25 patients with pulmonary embolism, only subsegmental embolisms were found. The clinical consequences of such peripheral thrombosis probably vary, depending on the extent of the infection-triggered parenchymal damage.
We have been informed by infectious disease and intensive care communities in Norway that a strikingly high incidence of thromboembolic complications has also been observed in COVID-19 patients here, despite the use of standard prophylactic doses of low molecular weight heparin (at our hospital, enoxaparin 40 mg × 1). Based on this information and our own experience, we have decided to use an increased prophylactic dose of enoxaparin (40 mg × 2) for some of these patients in our department. Systematic studies of the incidence of deep vein thrombosis and pulmonary embolism will be valuable for revealing the extent of such disease in hospitalised COVID-19 patients and for clarifying the underlying pathophysiology.
In our hospital, CT scans have only been used to a very limited degree for routine diagnostics in COVID-19 patients, primarily because inflammatory changes can readily be seen on a standard chest X-ray, but also because of infection control considerations. In view of the high incidence of pulmonary embolism in this patient group, more widespread use of CT angiography should be considered in patients with persistently high oxygen demand and biochemical signs of hyperinflammation, as well as in the event of D-dimer levels that are either very high or show marked variation.
Conclusion
Physicians who manage patients with COVID-19 both in and outside hospitals must be alert to the high incidence of thromboembolic complications in this patient group. Effective prophylaxis and rapid detection of any pulmonary embolisms will probably enable more patients to be treated successfully. CT pulmonary angiography should therefore be considered for patients with severe disease even if they are already being treated with low molecular weight heparin.
With limited understanding of the underlying pathophysiology and an absence of data from randomised controlled trials, it is difficult to formulate guidelines for thromboprophylaxis in cases of COVID-19. The incidence of bleeding complications should also be further surveyed. Whether increased prophylactic or therapeutic doses of low molecular weight heparin are indicated for the entire patient population, or only for those groups of patients thought to be at particularly high risk, has still to be determined. While we await the results of systematic studies, clinical practice should be based on consensus and experience from different centres nationally and internationally.
The patients have consented to the publication of this article.
The article has been peer-reviewed.
- 1.
Zhou F, Yu T, Du R et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: 1054–62. [PubMed][CrossRef]
- 2.
Tang N, Li D, Wang X et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 2020; 18: 844–7. [PubMed][CrossRef]
- 3.
Fogarty H, Townsend L, Ni Cheallaigh C et al. COVID-19 coagulopathy in Caucasian patients. Br J Haematol 2020; 189: bjh.16749. [PubMed][CrossRef]
- 4.
Mehta P, McAuley DF, Brown M et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395: 1033–4. [PubMed][CrossRef]
- 5.
Smeeth L, Cook C, Thomas S et al. Risk of deep vein thrombosis and pulmonary embolism after acute infection in a community setting. Lancet 2006; 367: 1075–9. [PubMed][CrossRef]
- 6.
Giannis D, Ziogas IA, Gianni P. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J Clin Virol 2020; 127: 104362. [PubMed][CrossRef]
- 7.
Campbell CM, Kahwash R. Will complement inhibition be the new target in treating COVID-19 related systemic thrombosis? Circulation 2020; 141: CIRCULATIONAHA.120.047419. [PubMed][CrossRef]
- 8.
Klok FA, Kruip MJHA, van der Meer NJM et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res 2020; 191: S0049-3848(20)30120-1. [PubMed][CrossRef]
- 9.
Poissy J, Goutay J, Caplan M et al. Pulmonary embolism in COVID-19 patients: Awareness of an increased prevalence. Circulation 2020; 141: CIRCULATIONAHA.120.047430. [PubMed][CrossRef]
- 10.
Leonard-Lorant I, Delabranche X, Severac F et al. Acute pulmonary embolism in COVID-19 patients on CT Angiography and relationship to d-dimer levels. Radiology 2020; 295: 201561. [PubMed][CrossRef]
- 11.
Grillet F, Behr J, Calame P et al. Acute pulmonary embolism associated with COVID-19 pneumonia detected by pulmonary CT angiography. Radiology 2020; 295: 201544. [PubMed][CrossRef]
- 12.
Ranucci M, Ballotta A, Di Dedda U et al. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost 2020; 18: jth.14854. [PubMed][CrossRef]
- 13.
Panigada M, Bottino N, Tagliabue P et al. Hypercoagulability of COVID-19 patients in Intensive Care Unit. A Report of Thromboelastography Findings and other Parameters of Hemostasis. J Thromb Haemost 2020; 18: jth.14850. [PubMed][CrossRef]
Det beskrives i artikkelen tre kasuistikker med lungeembolier hos pasienter med covid-19 infeksjon. For alle tre ble det beskrevet at de hadde for lav oksygenmetning i blodet. For pasient 1 og pasient 3 ble det beskrevet at de har respiratorisk alkalose, for pasient 1 ved innkomst, for pasient 3 noe senere i sykdomsforløpet.
Er det mulig å forklare hvordan det kan ha seg at disse pasientene har respiratorisk alkalose? Man skulle umiddelbart tro at de hadde respiratorisk acidose, ikke respiratorisk alkalose.
Hadde pasient 1 diabetes og infeksjonen førte til metabolsk acidose, kan respiratorisk alkalose være kompensatorisk. Hvis lungeembolusene ikke er for store og sitter i underlappene, vil tachypnoe og hyperventilering kunne senke CO2 og øke pH i midtfelt og overlapper.
Forekomst av DVT var uventet lav i de nederlandske dataene. I en fersk rapport i Thrombosis Research rapporterer Corrado Lodigiano og medarbeidere fra et universitetssykehus i Milano, The Humanitas Covid-19 Task Force, om 388 pasienter, innlagt i perioden 13.02.20 – 10.04.20. Median alder var 66 år, 68 % av pasientene var menn. 16 % trengte intensivbehandling (ICU), hvorav alle fikk tromboseprofylakse. 75 % fikk tromboseprofylakse på sengepost. 44 pasienter fikk billeddiagnostikk. Tromboemboliske komplikasjoner ble påvist hos 28 pasienter: 16 (36%) hadde DVT. CT-angiografi av 30 pasienter viste lungeembolisme (LE) hos 10 (33 %). 2,5 % hadde cerebrovaskulære og 1,1 % akutt coronarsyndrom/hjerteinfarkt. 2,2 % hadde disseminert intravaskulær koagulasjon (DIC). Det er verdt å merke seg at flere pasienter (36 %) fikk DVT enn LE. Halvparten av DVT-pasientene fikk påvist dette innen 24 timer etter innleggelse. Raskt økende D-dimer og DIC var forbundet med høy mortalitet.
Forfatterne konkluderer med at det nå haster med å forbedre spesifikk diagnostikk og behandling av venøs tromboembolisme, samt kartlegge effekt og risiko ved tromboseprofylakse hos Covid-19 pasienter. Dersom over en tredel av pasienten utvikler DVT, vil dette lett underdiagnostiseres i en situasjon hvor fokus er rettet mot luftveiene.