A young woman arrived at hospital after a new-onset seizure. Over the following months, she was hospitalised multiple times with serious neurological deficits, which turned out to be due to a relatively newly described neurological disorder.
A previously healthy woman in her early twenties arrived at Acute Admissions by ambulance after a new-onset seizure. The woman's boyfriend reported that she had suddenly become unwell, lost consciousness and shown asymmetrical shaking of her entire body for two minutes. She had bitten her lip, but had not experienced enuresis or frothing at the mouth, and had recovered quickly afterwards. For two weeks prior to the seizure, she had had headaches and muscle pain but had otherwise been well. Upon arrival, the examining doctor found normal vital signs, and normal somatic and neurological function with no photophobia or neck stiffness. Blood tests showed only CRP 60 mg/l (reference range 0–5 mg/l) and slightly elevated leukocyte levels.
A new-onset seizure with loss of consciousness may be the result of systemic disturbances such as hypoglycaemia or withdrawal, or a brain disorder such as stroke, tumour or infection (1). In the current case, the history of muscle pain and elevated CRP suggested an inflammatory origin. Head CT was performed to rule out serious intracranial pathology prior to lumbar puncture.
The cerebrospinal fluid showed a moderately elevated leukocyte count, consistent with meningitis or meningoencephalitis (Table 1). Aciclovir was administered until PCR for herpes simplex virus was found to be negative. EEG suggested generalised epileptiform activity. Head MRI showed leptomeningeal enhancement and possible swelling of certain cortical folds.
Table 1
Cerebrospinal fluid results. Pathological results are shown in bold.1
| Reference range |
Weeks after symptom onset |
||||||
|---|---|---|---|---|---|---|---|
| 0 |
1 |
2 |
8 |
19 |
|||
| Leukocytes (∙ 106 cells/l) |
0–4 |
22 |
292 |
475 |
61 |
146 |
|
| Percentage mono-/ polymorphonuclear |
- |
91/9 |
92/8 |
93/7 |
79/21 |
94/6 |
|
| Protein (g/l) |
0.15–0.50 |
0.29 |
0.83 |
1.4 |
0.69 |
0.75 |
|
| Glucose (mmol/l) |
approx. ⅔ of blood level |
3.4 |
- |
2.5 |
4.4 |
2.7 |
|
| Albumin (mg/l) |
0–350 |
153 |
439 |
763 |
315 |
394 |
|
| Albumin index |
0–9 |
3.8 |
9.5 |
17 |
7.5 |
8.8 |
|
| IgG (mg/l) |
0–34 |
24 |
56 |
- |
37 |
38 |
|
| IgG index |
< 0.7 |
0.45 |
0.5 |
- |
0.51 |
0.46 |
|
| Oligoclonal bands |
- |
Negative |
- |
- |
Negative |
Negative |
|
| Cytology |
- |
- |
- |
Normal |
- |
- |
|
| Antibodies2 |
- |
- |
Encephalitis, neuronal, anti-MOG and anti-aquaporin-4 |
- |
Anti-MOG and anti-aquaporin-4 |
- |
|
1PCR for varicella zoster virus, herpes simplex virus 1 and 2 and enterovirus, as well as serology for Borrelia, were negative in all tests. Negative cultures two and eight weeks after disease onset.
2Negative results
The medical history was consistent with serous meningitis or meningoencephalitis. The patient's good general condition, moderately increased CRP, mild pleocytosis, and normal ratio of cerebrospinal fluid/serum glucose meant that bacterial meningitis appeared unlikely, and virology results were negative for herpes encephalitis. There are many other possible causes of serous meningitis, including rare infections, malignancy, autoimmune diseases, medications and haemorrhage. Because the patient's condition improved spontaneously and the workup did not suggest any serious treatment-requiring illness, the patient was discharged after three days without further tests.
Four days later, the patient gradually developed an increasing sensation of tingling in her hands, followed by hypersensitivity and then numbness in her abdomen, back, genital area, buttocks and legs, as well as urinary retention, constipation and impaired balance. Upon readmission one week after discharge, she had unsteady gait, reduced sensitivity distal to the lower thorax, reduced sphincter tone and 800 ml residual urine. Motility, tendon reflexes, Romberg's test and plantar reflexes were normal.
The patient's sensory symptoms distal to the thorax and difficulty urinating were consistent with subacute myelopathy. Guillain-Barré syndrome can also cause rapidly progressive sensory abnormalities, but most often causes paresis and extinction of tendon reflexes. Common causes of subacute myelopathy are demyelinating disorders and infections as well as post-infectious and paraneoplastic disease (2).
MRI of the brain and total medulla showed enhancement in more than 50 % of the medulla diameter with weak contrast enhancement at levels C4–C5, C6–C7 and Th11–Th12 (Figure 1), but improvement in the lesions that had previously suggested meningitis. The radiologist considered the images to be most consistent with acute disseminated encephalomyelitis. A new lumbar puncture (Table 1) showed an increasing cell count and moderately increased total protein and albumin consistent with blood-brain barrier failure. On the basis of the increasing cell count in the cerebrospinal fluid, aciclovir was again administered until the PCR result for herpes simplex virus came back negative.
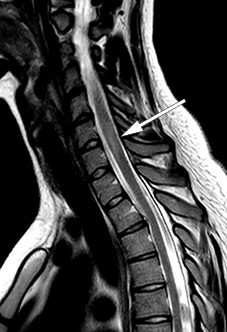
The anamnesis, clinical examination, MRI and cerebrospinal fluid were all consistent with encephalomyelitis. Common causes, such as an infection or multiple sclerosis, appeared less likely given the negative test results for infection and the absence of intrathecal IgG production (oligoclonal bands and IgG index). In such cases, it is necessary to investigate other possible causes: in addition to a broad pathogen detection assay, tests for paraneoplastic and encephalitis antibodies in serum were therefore performed. Angiotensin converting enzyme (ACE), which is a marker of sarcoidosis, and antibodies against aquaporin-4 and myelin oligodendrocyte glycoprotein (MOG), which are markers of neuromyelitis optic spectrum disorders, were quantified in both serum and cerebrospinal fluid. Cerebrospinal fluid cytology was also performed to exclude malignancy.
The following week, the patient developed increasing paralysis of the legs and difficulty walking, numbness of the hands and left-sided plantar inversion. She developed a headache, especially around her left eye upon eye movement, and subjectively reduced visual acuity in that eye. To rule out infection, another lumbar puncture was performed, which showed a further increase in leukocytes as well as signs of blood-brain barrier failure, but still no evidence of infection (Table 1, two weeks after symptom onset).
Subacute unilateral reduction in visual acuity and pain upon eye movement suggested optic neuritis. The combination of myelitis and optic neuritis is typical of demyelinating disease. In multiple sclerosis, the most common demyelinating disease in adults, there are usually also characteristic periventricular, infratentorial or juxtacortical lesions, which were absent in our patient. In addition, oligoclonal bands and an increased IgG index are typically seen in the cerebrospinal fluid, as well as a lower cell count. Neuromyelitis optica is usually accompanied by more longitudinally extensive myelitis changes, and often by optic neuritis with concurrent or sequential involvement of both optic nerves (3). Acute disseminated encephalomyelitis most commonly affects children in the wake of a viral infection of the respiratory tract and is characterised by altered consciousness, fever, and neurological deficits that progress over a few days, with MRI showing multifocal demyelinating lesions of roughly the same age (4). In both acute disseminated encephalomyelitis and neuromyelitis optica, an increased cell count in the cerebrospinal fluid is common whereas oligoclonal bands are rare.
High-dose corticosteroids are the acute treatment for all inflammatory demyelinating diseases, and 1 g of methylprednisolone was therefore given intravenously for five days followed by tapering of prednisolone over two weeks. A marked improvement was seen after a few hours. The headache, photophobia and pain upon eye movement disappeared, and urination and movement of the lower extremities improved. However, MRI showed new infratentorial lesions medially in both cerebellar peduncles as well as anteriorly in the pons and medulla oblongata, but a reduction in contrast enhancement in the medulla.
In acute disseminated encephalomyelitis, cerebral lesions are usually seen as diffuse white matter changes, but lesions in the cortex and basal ganglia also occur. While awaiting test results, our patient was discharged for rehabilitation with a diagnosis of acute disseminated encephalomyelitis.
A follow-up MRI one month later showed a new non-enhancing lesion in the right thalamus (Figure 2) as well as two new, small, enhancing lesions in the medulla at C5, C6 and possibly also Th5. The infratentorial lesions had resolved completely, and those in the medulla oblongata were reduced.
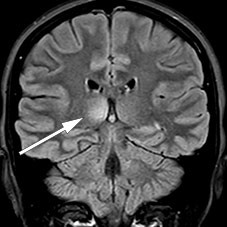
Acute disseminated encephalomyelitis is usually monophasic and is typically treated with steroids for four to six weeks. The changes on MRI could thus be interpreted as suggesting both a need to reconsider the diagnosis and to continue with anti-inflammatory treatment. However, the patient reported feeling well, and tests for ACE, neuronal antibodies, encephalitis antibodies, aquaporin-4 antibodies and MOG antibodies were negative, as was cerebrospinal fluid cytology. It was therefore decided to wait with any further treatment, and the patient was sent for rehabilitation for continuing difficulties with urination.
To months after discharge from the neurology department, the on-duty doctor was contacted by the rehabilitation centre because the patient had experienced gradually increasing headache and nausea as well as reduced vision in her left eye over the past 24 hours. The on-duty ophthalmologist measured visual acuity of 0.05 in the left eye and 1.2 in the right, and suspected optic neuritis. The on-duty neurologist noted deviation of the uvula to the left and bilateral inversion of the plantar reflexes. Lumbar puncture showed moderately increased cell count, total protein and IgG (Table 1). Head MRI showed bilateral hyperintensity in the thalamus in the absence of any other brain lesions, as well as bilateral hyperintensity in the optic nerve and hyperintensity in the optic chiasm (Figure 3). The patient was again given methylprednisolone, which produced a rapid improvement.
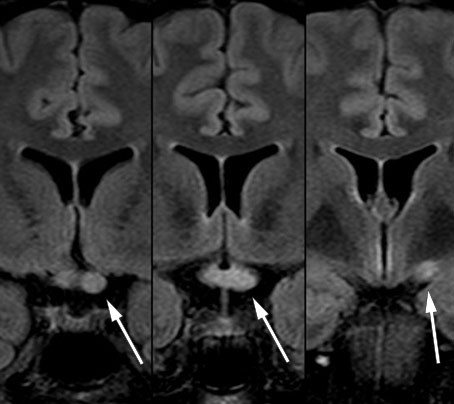
Myelitis and bilateral optic neuritis are the hallmarks of neuromyelitis optica spectrum disorders. Although the patient had previously tested negative for antibodies against aquaporin-4 and MOG, this now seemed the most likely diagnosis, and new samples of serum and cerebrospinal fluid were therefore obtained for antibody testing. PET-CT and mammography were also performed, as neuromyelitis optica spectrum disorders in older individuals may be associated with breast cancer.
When tests showed no signs of malignancy, treatment was initiated with 1 000 mg rituximab infusions two weeks apart, as well as 60 mg of prednisolone with gradual tapering prior to scheduled discontinuation after two months.
Neuromyelitis optica spectrum disorders are usually treated with immunosuppressants such as azathioprine, mycophenolate mofetil or rituximab in combination with corticosteroids and intravenous immunoglobulins or plasmapheresis (5, 6). Rituximab is an anti-CD20 antibody that produces prolonged suppression of memory B cells in the blood. Rituximab is also effective in multiple sclerosis, which had still not been fully ruled out.
After discharge, the second serum anti-MOG test was found to be positive. Towards the end of the prednisolone taper, the patient had two brief admissions to the medical department owing to persistent headache and urinary tract infection. MRI of the brain and spinal cord and neurological examinations revealed no new findings at this point. Immediately after discontinuation of prednisolone, the patient was admitted to the neurology department with increasing visual impairment, diplopia, right-beating nystagmus, and abduction paresis in the right eye. An MRI 20 days after the previous examination and almost two months after rituximab initiation, revealed new bilateral frontal subcortical lesions as well as in the medulla, left cerebellar peduncle and corpus callosum (Figure 4). Lumbar puncture still showed moderate pleocytosis (Table 1, last column). As before, the patient received methylprednisolone with good effect, and was then discharged home with long-term prednisolone tapering and prompt follow-up at the outpatient clinic.
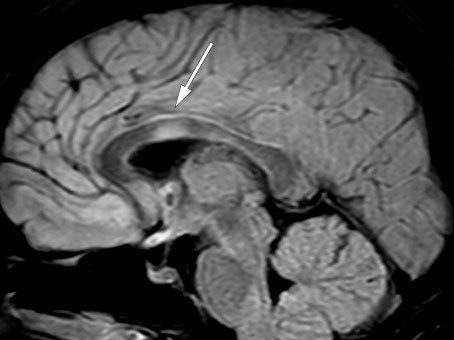
A third test confirmed positive anti-MOG status. The patient's overall medical history, including exacerbations shortly after discontinuation of prednisolone, was also consistent with anti-MOG encephalomyelitis (7). Lesions in the corpus callosum are common in multiple sclerosis, but may also occur in cases of adult anti-MOG encephalomyelitis (8).
The patient has received semi-annual infusions of rituximab for the past two years. Prednisolone has been slowly reduced to 2.5/5 mg every other day, with no clinical deterioration or new MRI findings. She experienced significant cushingoid adverse effects until the prednisolone dose fell below 10 mg/day. She has persistent urination difficulties, but has otherwise regained all functions that were affected during the course of her illness.
Discussion
The detection of new autoantibodies has changed our understanding of many neurological diseases (6). The discovery of antibodies against aquaporin-4 led to neuromyelitis optica, previously considered a subtype of multiple sclerosis, instead being considered a disease in its own right (5). The detection of antibodies against NMDA and other neuronal cell-surface proteins led to the recognition and characterisation of multiple types of autoimmune encephalitis.
Anti-MOG encephalomyelitis is an even more recent example (6). Anti-MOG antibodies were first detected in multiple sclerosis, but their presence turned out to be an artefact resulting from unsatisfactory laboratory techniques (6). In recent years, radiological and clinical features of anti-MOG encephalomyelitis have been characterised using techniques in which native MOG protein is expressed on the cell surface (6, 8, 9). The disease occurs most frequently in children and young adults. In children, the disease often resembles acute disseminated encephalomyelitis, but with more frequent relapses (6). In adults, the disease is characterised by myelitis and optic nerve inflammation and may resemble neuromyelitis optica, but it often responds better to treatment and gives rise to fewer sequelae. Because neuromyelitis optica and anti-MOG encephalomyelitis may have similar clinical presentations, they are often referred to collectively as neuromyelitis optica spectrum disorders (6), even though they are associated with different autoantibodies. Anti-aquaporin-4 binds to astrocytes, whereas anti-MOG binds to the surface of oligodendrocytes. Antibodies against aquaporin-4 from patients with neuromyelitis optica can induce a similar disease in laboratory animals and are therefore assumed to be pathogenic. This is not the case for anti-MOG antibodies (3, 6, 9).
Anti-MOG encephalomyelitis frequently gives rise to longitudinally extensive myelitis and bilateral optic neuritis, and oligoclonal bands are rarely detected in the cerebrospinal fluid (3, 6). Meningoencephalitis with epileptic seizures and acute disseminated encephalomyelitis-like MRI findings are also common, as well as involvement of the medullary cone with neurogenic bladder (7). Our patient thus exhibits almost all the typical signs and symptoms. She illustrates, too, that there is often a very good response to steroids with near-complete resolution of symptoms, but that frequent relapses occur if prednisolone is rapidly tapered to less than 10 mg per day (9). When treating multiple sclerosis, it takes at least two months for the anti-inflammatory effect of anti-CD20 treatment to become established. This is also likely to be the case in anti-MOG encephalomyelitis. The patient should receive steroid cover for this period at least and probably for longer (7).
While some immunomodulatory drugs for multiple sclerosis can exacerbate neuromyelitis optica and anti-MOG encephalomyelitis, observational studies suggest that immunosuppression with azathioprine, mycophenolate mofetil or rituximab as well as immunomodulation with intravenous immunoglobulins is effective (6). Such treatment should be started immediately upon diagnosis. It is currently unknown when, and indeed whether, treatment should be discontinued. In our experience rituximab is very well tolerated by patients with demyelinating diseases. Although the prognosis for anti-MOG encephalomyelitis is better than for neuromyelitis optica, attacks can still produce significant sequelae. Monoclonal antibodies directed against interleukin-6 or complement protein C5 may eventually offer new therapeutic options. We have therefore chosen to continue rituximab until more robust data are available regarding discontinuation and treatment alternatives.
The patient has consented to the publication of this article.
This article has been peer-reviewed.
- 1.
Realfsen MS, Bø SM, Lossius MI et al. Førstegangs generalisert tonisk-klonisk krampeanfall. Tidsskr Nor Legeforen 2015; 135: 1256–8. [PubMed][CrossRef]
- 2.
Frohman EM, Wingerchuk DM. Clinical practice. Transverse myelitis. N Engl J Med 2010; 363: 564–72. [PubMed][CrossRef]
- 3.
Kvistad SA, Wergeland S, Torkildsen Ø et al. Neuromyelitis optica. Tidsskr Nor Legeforen 2013; 133: 2057–61. [PubMed][CrossRef]
- 4.
Pohl D, Alper G, Van Haren K et al. Acute disseminated encephalomyelitis: Updates on an inflammatory CNS syndrome. Neurology 2016; 87 (suppl 2): S38–45. [PubMed][CrossRef]
- 5.
Kessler RA, Mealy MA, Levy M. Treatment of neuromyelitis optica spectrum disorder: acute, preventive, and symptomatic. Curr Treat Options Neurol 2016; 18: 2. [PubMed][CrossRef]
- 6.
Reindl M, Waters P. Myelin oligodendrocyte glycoprotein antibodies in neurological disease. Nat Rev Neurol 2019; 15: 89–102. [PubMed][CrossRef]
- 7.
Ramanathan S, Mohammad S, Tantsis E et al. Clinical course, therapeutic responses and outcomes in relapsing MOG antibody-associated demyelination. J Neurol Neurosurg Psychiatry 2018; 89: 127–37. [PubMed][CrossRef]
- 8.
Jarius S, Ruprecht K, Kleiter I et al. MOG-IgG in NMO and related disorders: a multicenter study of 50 patients. Part 2: Epidemiology, clinical presentation, radiological and laboratory features, treatment responses, and long-term outcome. J Neuroinflammation 2016; 13: 280. [PubMed][CrossRef]
- 9.
Dos Passos GR, Oliveira LM, da Costa BK et al. MOG-IgG-associated optic neuritis, encephalitis, and myelitis: lessons learned from neuromyelitis optica spectrum disorder. Front Neurol 2018; 9: 217. [PubMed][CrossRef]