Mycobacteria are aerobic, non-spore-forming rod bacteria, are non-motile and acid-fast, i.e. they cannot be decolourised by acid-alcohol in special staining. Their growth rate is generally low, but varies. This forms the basis for their subdivision into fast- and slow-growing species, defined as growth in a solid medium before or after seven days, when spread from a liquid bacterial culture. Non-tuberculous mycobacteria (NTM) have varying pathogenicity and are normally found in the environment, e.g. in soil and water, but can cause infections in both humans and animals. The bacteria may also colonise medical equipment. The most common causes of NTM pulmonary infections in the USA and Europe are the Mycobacterium avium complex (MAC), followed by M. kansasii, the M. abscessus complex and M. malmoense, in slightly varying order, depending on the geographical area. In some places, M. fortuitum and M. xenopi are more common (1, 2). The M. abscessus complex and M. fortuitum are fast-growing species, the others are slow-growing.
With the introduction of new molecular methods, a growing number of non-tuberculous mycobacteria have been identified: to date, 186 species and 13 subspecies (3, 4). The classification into groups/complexes, species and subspecies is constantly changing, and may vary depending on method. This may be of clinical significance and influence the choice of antibiotics. This applies in particular to the fast-growing species M. abscessus, which is now classified as a complex and divided into three different subspecies: abscessus, bolletii and massiliense. Inducible macrolide resistance is found in the first two subspecies and is associated with poorer treatment outcomes (5).
The probability of mycobacterial infection varies with the pathogenicity of the species, the degree of exposure and the person's susceptibility. It was formerly believed that human-to-human infection did not occur, but recent case histories indicate that it can take place between patients with cystic fibrosis (6, 7). To the best of our knowledge, however, there is no consensus on whether this calls for special infection control measures.
Up to the 1950s, M. tuberculosis was the predominant mycobacterial pulmonary infection, and non-tuberculous mycobacteria were generally regarded as non-pathogenic. As the prevalence of tuberculosis fell, an increasing number of patients were diagnosed with NTM pulmonary infections. During the HIV epidemic of the 1980s, the prevalence of systemic NTM infections increased, and the need for better treatment received more attention.
NTM infections are not notifiable in most countries, including Norway, and detection of pulmonary infections is based on clinical, radiological and microbiological criteria. It is therefore difficult to obtain a good overview of prevalence. Nevertheless, studies from other countries indicate an increase in recent years that cannot be explained by the HIV epidemic or improved diagnostics (1, 8). Many reasons have been proposed for this, such as a larger proportion of elderly in the population, lower BCG vaccination coverage, more people living longer with structural pulmonary diseases (bronchiectasis, chronic obstructive pulmonary disease) and diseases that impair the immune system (malignancy, diabetes mellitus etc.) (9), in addition to increased use of immunomodulatory therapy (10). In contrast to tuberculosis, it is difficult to say whether the prevalence of NTM infections is higher in people from countries other than Norway, as we do not have an overview of the prevalence in Norway.
Diagnosing and treating NTM pulmonary infections is a challenge. International guidelines build largely on experience and case studies (11–13). There is a brief general account of the subject in the Norwegian Institute of Public Health's tuberculosis guidelines. Other than that, there are no national guidelines on the subject. This article summarises the most recent knowledge on the subject, with the emphasis on diagnosis and treatment.
Method
In 2007, the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA) issued joint guidelines for the diagnosis, treatment and prevention of non-tuberculous mycobacterial diseases (11). These guidelines form the basis for many of the recommendations regarding diagnosis and treatment that are presented in this article. More recent findings and guidelines are also included. Guidelines for the treatment of patients with cystic fibrosis (CF) came in 2016 (12), and in 2017 the British Thoracic Society (BTS) issued its own recommendations (13), which are based on a review of relevant literature and the conclusions of a panel of experts.
We searched in PubMed, Embase and Cochrane for all reviews and systematic reviews on non-tuberculous mycobacteria as a cause of pulmonary disease published in the period 2007–2017 (see search strings in the appendix). The search cut-off date was 28 September 2017. There were a total of 215 hits. Articles in languages other than English or that were considered not to cover the topic (infections in animals, local epidemiological data and infections in children) and infections in HIV patients were excluded – 103 articles in all. A total of 112 articles were reviewed in their entirety. In addition, relevant original articles cited in the literature were reviewed – a total of 50 articles.
Diagnosis
The recommendations from the American Thoracic Society and the Infectious Diseases Society of America provide criteria for clinical, radiological and bacteriological findings for determining whether a suspected NTMpulmonary infection lends itself to treatment (11) (Box 1).
Clinical/radiological criteria
-
Pulmonary symptoms and nodular or cavitary opacities on chest radiographs or multifocal bronchiectasis with multiple small nodules on a CT thorax
and
-
Appropriate exclusion of other diagnoses
Microbiological criteria
-
Positive culture result in at least two separate expectorated sputum samples. If the results are non-diagnostic, consider repeat direct microscopy samples for acid-fast bacteria and culture
or
-
Positive culture result in one bronchial wash or bronchoalveolar lavage
or
-
Transbronchial or other lung biopsy with histological findings consistent with mycobacterial infection (granulomatous inflammation or acid-fast bacteria) and growth of non-tuberculous mycobacteria, or biopsy with findings consistent with mycobacterial infection and one or more sputum samples or bronchoalveolar washings that are culture-positive.
A doctor with experience in the field should be consulted in the event of growth of unusual microbes or microbes that usually represent contamination.
Patients suspected of having NTM pulmonary disease but who do not meet the criteria should be followed until the diagnosis has been confirmed or excluded.
The diagnosis NTM pulmonary disease does not, per se, mean that therapy must be instituted. This must be decided after carefully weighing up the potential risks and benefits to the individual patient.
Clinical examination
Symptoms of NTM lung infection are non-specific as a rule. Coughing, increased mucous formation, weight loss, low-grade fever, haemoptysis and dyspnoea are common. In addition, patients often have an underlying disease that may produce similar symptoms. The clinical presentation may resemble that found with tuberculosis (14, 15).
Cystic fibrosis or bronchiectasis are overrepresented among patients with NTM pulmonary infections, and in a recently published meta-analysis, the condition was found in 9.3 % of patients with bronchiectasis (16). Overrepresentation of patients with mutation in the cystic fibrosis transmembrane regulator gene (CFTR gene) is also found, although these do not have clinical cystic fibrosis (heterozygous mutation) (17–20). The reason for this, and the part played by the CFTR gene, are still unknown.
An unusual disease manifestation is hypersensitivity pneumonitis. The cause is assumed to be an immunological reaction to inhaled non-tuberculous mycobacteria in aerosols from infected water, including swimming pools and hot tubs (21, 22); it is also called "hot tub lung". Clinical and radiological findings are similar to other types of hypersensitivity pneumonitis.
Radiological examination
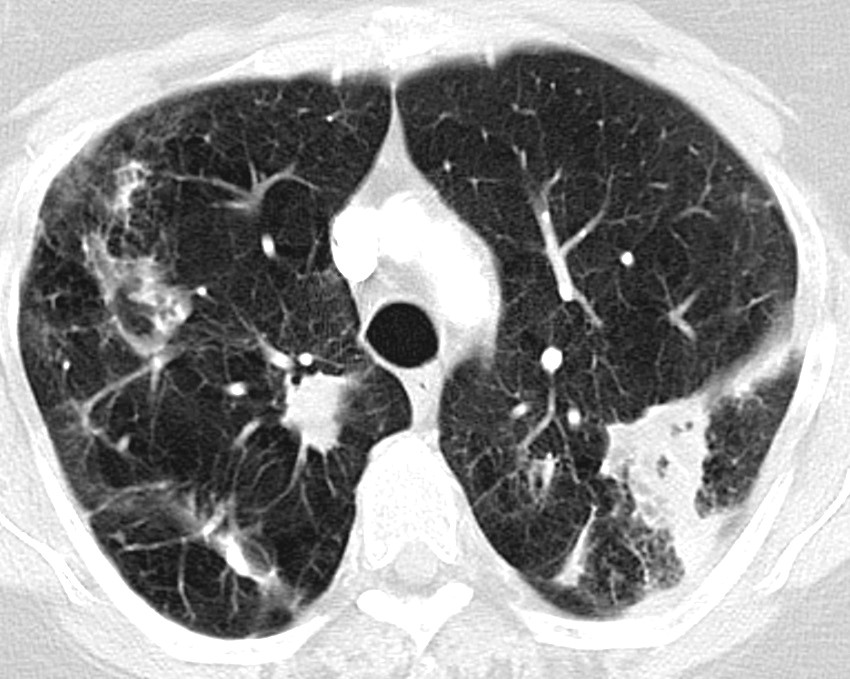
When NTM pulmonary disease is suspected, the patient should undergo a CT thorax. The two most common radiological presentations are the fibrocavitary and nodular bronchiectasis forms (23, 24). The fibrocavitary form has cavitary lesions, most frequently in the upper lobes, with radiological findings that resemble tuberculosis (Fig. 1). This form often has a more aggressive course, and is seen most frequently in elderly men who smoke or have another pulmonary disease, such as chronic obstructive pulmonary disease, or have had tuberculosis. The nodular bronchiectasis form is characterised by multifocal bronchiectasis and small nodules, most frequently in non-smoking, elderly women. This form is also over-represented in patients with a low body mass index, scoliosis, pectus excavatum, mitral prolapse, and in tall persons (17, 19).
Microbiological examination
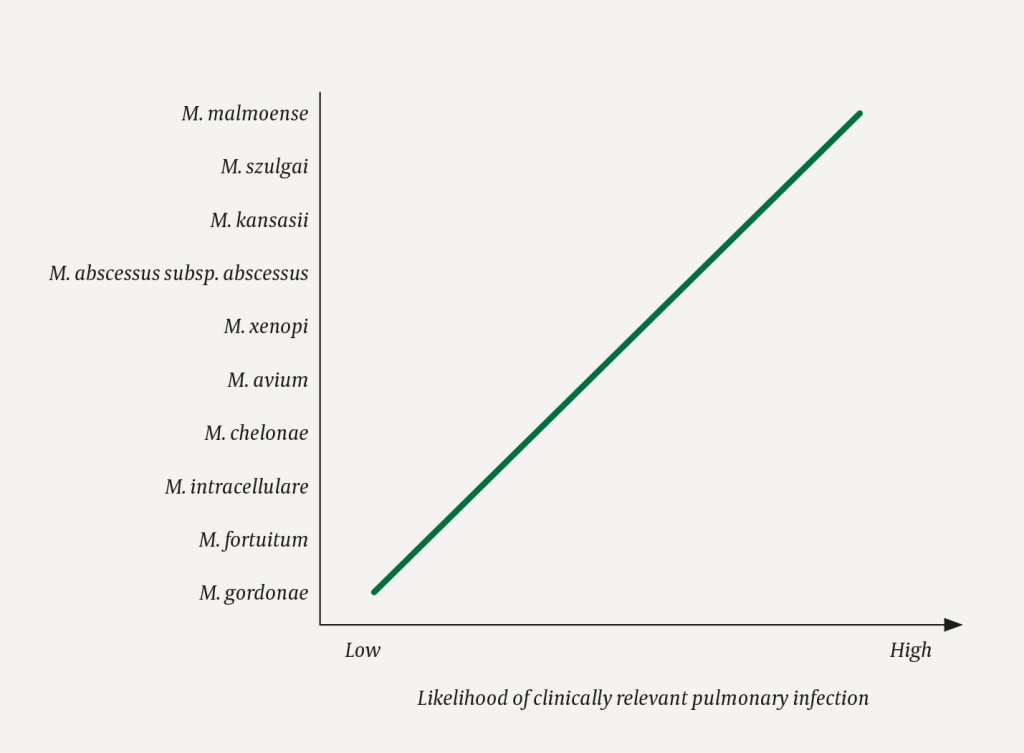
Microbiological diagnosis of pulmonary disease due to non-tuberculous mycobacteria may be difficult because these bacteria occur naturally in the environment. Unlike tuberculosis, where findings of the bacteria confirm the disease, findings of non-tuberculous mycobacteria in a respiratory specimen do not necessarily imply clinically relevant infection. There is a strong risk of contamination of sputum or bronchoscopy samples (e.g. from the oral cavity), and a positive finding may also represent bronchial colonisation without this being the cause of the patient's complaint. Species identification is very important, as some species are more pathogenic than others (9, 25) (Fig. 2). The occurrence of a clinically relevant pulmonary infection with the various species in an population will also depend on the geographical distribution of the bacterium in the environment and the prevalence of risk factors in the population (1, 25). Growth in two or more sputum specimens taken at different times is a required microbiological criterion for NTM pulmonary infection. Alternatively, growth in only one specimen extracted via bronchoscopy (bronchoalveolar lavage, BAL), is accepted, or a culture positive lung biopsy (11). At the Departments of Pulmonary and Infectious Diseases at Oslo University Hospital we always want sputum samples to be taken some weeks apart to confirm persistent infection. There has been debate about these criteria, particularly that only one bronchoscopy specimen is sufficient, as contamination is a risk here too. In any case, the criteria must be used with discretion, with emphasis on which microbes are found, and whether other findings and clinical presentation are convincing.
In addition to identifying the bacteria, it is important to perform relevant susceptibility testing in order to be able to choose the correct treatment regimen. For the fast-growing mycobacteria, it is recommended that resistance to a number of antibiotics be tested, while testing for fewer drugs is recommended for the slow-growing mycobacteria. Unfortunately, the connection between in vitro susceptibility determination and in vivo efficacy is uncertain for several of the drugs in question. In the case of infection with the Mycobacterium avium complex (MAC), for example, only macrolides and amikacin have similar in vitro and in vivo effect (26–28).
Macrolide sensitivity has proved to be an important prognostic indicator in the treatment of NTM infections (5, 29). In addition to acquired resistance due to mutations in the 23S rRNA gene (the rrl gene), inducible macrolide resistance has been observed in some fast-growing, non-tuberculous mycobacteria in recent years. These may initially be susceptible to macrolides, but resistance can appear and is detected in vitro after exposure to macrolides for up to 14 days. This is associated with a functional erm gene in some species and subspecies (30). It is therefore important to take this into account when susceptibility testing is done and when choosing antibiotics regimens. The most important risk factor for the development of macrolide resistance is macrolide monotherapy (31). In light of this, and because macrolides play such a central part in the treatment of these infections, questions have been raised about long-term macrolide therapy for patients with pulmonary disease, for example patients with cystic fibrosis (32, 33).
Treatment
A few randomised, controlled studies have been conducted to evaluate the therapeutic efficacy of different regimens (34–38). However, these studies have several weaknesses, and do not provide unambiguous answers. Recommended treatment regimens are therefore based largely on experience and expert statements. We provide a brief account below, but for a more comprehensive review, we refer to the American and British guidelines (11, 13). Unless otherwise specified, the treatments recommended in the article are taken from these guidelines.
Treatment of NTM pulmonary infections is long-term, requires concurrent use of several antimicrobial drugs, and has varying effectiveness. Successful treatment is defined as negative sputum tests for 12 months during ongoing treatment. Treatment is generally recommended for at least 18 months. The exception is treatment of hypersensitivity pneumonitis, where treatment with antimicrobial agents has not been seen to have any definite effect. Recommended therapy for this is systemic glucocorticoids and avoidance of further exposure.
The treatment regimens are resource-intensive and often cause adverse reactions and drug interactions. At the same time, there is great variation in the natural course of these infections – from subclinical to rapidly progressing disease. It is therefore very important to make a thorough assessment of the patient with respect to whether initiating treatment would be appropriate. At the same time, it is important to optimise other treatment, such as mucous mobilisation and pulmonary physiotherapy, and to exclude other causes of disease. Monitoring of therapeutic effectiveness by means of sputum samples taken at 1–2 month intervals is recommended. If sputum is still culture-positive after six months of treatment, the treatment has probably failed. New samples should be taken for susceptibility testing before the antibiotic regimen is revised. Although the object of the treatment is usually cure, in some cases this will be unrealistic. The goal of the treatment may then be suppression of the infection.
Relevant antibiotics
Macrolides have an important place in the treatment of non-tuberculous mycobacteria. No definite difference in efficacy has been found between clarithromycin and azithromycin, and the primary advantage of the latter is fewer drug interactions. The risk of drug interactions should also be borne in mind when rifampicin is used. The usual dosage of rifampicin is 600 mg for weights over 50 kg, 450 mg for lower weights. The aminoglycosides recommended in the international guidelines are streptomycin or amikacin, except for treatment of M. chelonae, where tobramycin has proved most efficacious. Determining the serum concentration of streptomycin is not easy, and in consequence amikacin is preferred of these two. If patients are to be treated with aminoglycosides, renal function must be assessed and serum concentration measured, and patients must be monitored for ototoxicity. If isoniazid is used, a daily supplement of 40 mg x 1 pyridoxine is recommended as prophylaxis for polyneuropathy, and when ethambutol is used, visual acuity and colour vision should be tested before commencement and in case of symptoms. ECG is recommended before commencement and after two weeks on therapy with drugs that may result in a prolonged QT interval, such as macrolides and fluoroquinolones (13).
Table 1 sets out our recommendations for treatment of the most common pulmonary diseases due to non-tuberculous mycobacteria, based on the American and British guidelines (11, 13).
Table 1
The authors' recommendations for treatment of the pulmonary diseases due to the most common non-tuberculous mycobacteria, based on American and British guidelines (11, 13)
| Drug regimen | Duration | |
|---|---|---|
| Mycobacterium avium complex | ||
| Nodular/bronchiectatic disease | Three times a week | 12 months after negative sputum culture |
| Cavitary lung disease | Daily | 12 months after negative sputum culture |
| M. kansasii | Daily | 12 months after negative sputum culture |
| M. malmoense | Daily | 12 months after negative sputum culture |
| M. abscessus complex | Intensive phase: | 12 months after negative sputum culture |
Slow-growing non-tuberculous mycobacteria
Macrolides are the cornerstone of treatment of most infections with slow-growing non-tuberculous mycobacteria (Table 1). There are only guidelines for susceptibility testing of some of these species. It is initially recommended that species in the Mycobacterium avium complex be tested only for resistance to macrolides and amikacin. If macrolides cannot be used, moxifloxacin and linezolid should also be tested. For M. kansasii, M. malmoense and M. xenopi, initial testing for resistance to rifampicin is recommended, and expanded testing if resistance to rifampicin is found.
The Mycobacterium avium complex (MAC)
The M. avium complex includes the species M. avium, M. intracellulare and M. chimaera. The recommended treatment regimen is macrolide, rifampicin and ethambutol for 12 months after the last negative culture, typically 18–24 months. For patients without cavitary lesions, an intermittent regimen with dosing three times a week has shown results equally as good as daily treatment (39, 40). Recent studies have also shown less severe adverse reactions with intermittent therapy (41, 42). The results of intermittent therapy are poorer for cavitary disease, and it is therefore not recommended. In cases of severe illness, it is recommended that an aminoglycoside be added for the first two to three months of the therapy. If there is a need for long-term aminoglycoside therapy, amikacin inhalation may be an option (43). In case of treatment failure, addition of moxifloxacin may increase the success rate (44, 45). Clofazimine may be used as an effective alternative to rifampicin, or for refractory MAC disease (46, 47).
The treatment results for MAC pulmonary disease are still relatively poor. Termination of treatment because of adverse reactions is common (10–30 %), and there is only 40–60 % total treatment success (29, 48). The outcomes are better (about 70–85 %) in patients without cavitary disease, but recurrence of the disease after successful treatment is common (30–50 %). This is usually due to reinfection with a new strain of the complex (42, 49). Resistance to macrolides substantially reduces therapeutic success and complicates treatment (31).
Mycobacterium kansasii
This is generally the easiest species of all non-tuberculous mycobacteria to treat.
Studies have shown treatment success of up to 95 % (50). In contrast to other non-tuberculous mycobacteria, there is relatively good correlation between in vitro determination of resistance and clinical efficacy for rifampicin, macrolides and fluoroquinolones (51). The standard regimen has been daily treatment with rifampicin, ethambutol and isoniazid (11). However, there is uncertainty concerning the efficacy of isoniazid in the treatment, and a beneficial effect has been reported for macrolides and fluoroquinolones. Many therefore recommend replacing isoniazid with a macrolide or fluoroquinolone (52) (Table 1).
Mycobacterium malmoense
There is no consensus regarding the optimal therapeutic regimen for this mycobacterium, and studies with different regimens have produced divergent outcomes (34, 53). A retrospective study from the Netherlands reported relatively high therapeutic success (70 %) (54). A regimen of isoniazid, rifampicin and ethambutol used to be recommended.
However, this has been changed in more recent guidelines, which replace isoniazid with a macrolide (13). In cases of serious illness, supplementary aminoglycoside therapy can be considered (13). A fluoroquinolone may also be added.
Fast-growing non-tuberculous mycobacteria
Although the correspondence between clinical efficacy and susceptibility testing is uncertain, it is recommended that this should govern the choice of antibiotics. In the current guidelines for fast-growing mycobacteria, testing is recommended for amikacin, cefoxitin, ciprofloxacin, moxifloxacin, clarithromycin, doxycycline, imipenem, linezolid, trimethoprim- sulfamethoxazole and tobramycin (11), even though not all drugs are relevant for all species. M. chelonae is the only species for which tobramycin is preferred to amikacin as aminoglycoside.
M. fortuitum is generally regarded as the easiest to treat, and is usually sensitive to a number of drugs (55), while infection with the M. abscessus complex, which is by far the most frequent cause of pulmonary infections, is particularly difficult to treat.
The effect of different treatment regimens on M. abscessus has been investigated in only two major studies. In 2009, a retrospective Korean study was published in which 65 patients who had received standardised treatment against M. abscessus were evaluated (56). They all received clarithromycin, ciprofloxacin and doxycycline by mouth. For four weeks initially, they also received intravenous therapy with amikacin and either cefoxitin or imipenem. Negative sputum culture tests for more than one year were achieved for 58 % of patients. Symptom improvement was experienced by 83 %, and radiological improvement by 74 % of patients. Twenty-two percent had undergone surgery. Only 63 % completed the therapy according to plan. It is noteworthy that macrolide resistance was strongly associated with a poor therapeutic outcome. Only 17 % in this group achieved a cure.
In 2011, a retrospective study was published by National Jewish Health in Colorado (57). Sixty-nine patients were followed for 34 months on average. In this study, the antibiotic regimen was customised on the basis of in vitro resistance determination and patient tolerance. The average treatment time was 52 months, six of them with intravenous treatment. Sputum samples from 48 % of the patients remained culture negative. It is noted here that substantially better results in terms of culture negativity for 12 months were achieved by those who had surgery – comprising 33 % of those in this study.
It is not possible on the basis of the literature to give an exact recommendation for which drugs should be selected to treat the M. abscessus complex. In addition to the resistance pattern, the side effect profile has to be taken into account. An intensive phase of 1–6 months with at least two parenteral drugs and a macrolide is recommended, followed by a maintenance phase with oral drugs for 12 months following negative sputum tests (Table 1). The most effective parenteral drugs include amikacin, cefoxitin, imipenem and tigecycline. After the intensive phase of treatment has been completed, treatment with macrolides is recommended and in addition one or two other oral antibiotics such as fluoroquinolones, linezolid or clofazimine. Amikacin inhalation is also an option. All patients infected with the M. abscessus complex should be considered for surgery if there is focal disease, as the success rate is higher for patients who have combined medical and surgical treatment (57–61).
Summary
The diagnosis and treatment of pulmonary infections with non-tuberculous mycobacteria are based on clinical and radiological findings and on microbiological test results. Identification of species and, if relevant, subspecies is very important, and susceptibility testing is often also important for choosing a medication regimen. Treatment consists of a combination of several drugs for a long period, sometimes has limited efficacy and can cause considerable adverse reactions and drug interactions. It is therefore important to decide whether the benefit of the treatment is expected to outweigh the disadvantages it can entail. If a decision is made to treat, patients should be followed up regularly with sputum cultures and monitoring of adverse reactions. We recommend that these patients be treated at hospitals with specialists in infectious diseases or pulmonary medicine (preferably working together), microbiology, radiology and thoracic surgery. We have established a multidisciplinary group of this kind at Oslo University Hospital, Ullevål, which discusses all patients who may be candidates for treatment.
Main points
We found that diagnosing and treating pulmonary infections with non-tuberculous mycobacteria requires a thorough evaluation of clinical, radiological and microbiological findings.
The treatment is long-term and is conducted with a combination of antibiotics, and its efficacy varies.
There is a high incidence of adverse reactions and drug interactions
- 1.
Prevots DR, Marras TK. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: a review. Clin Chest Med 2015; 36: 13 - 34. [PubMed][CrossRef]
- 2.
Kendall BA, Winthrop KL. Update on the epidemiology of pulmonary nontuberculous mycobacterial infections. Semin Respir Crit Care Med 2013; 34: 87 - 94. [PubMed][CrossRef]
- 3.
Tortoli E. Microbiological features and clinical relevance of new species of the genus Mycobacterium. Clin Microbiol Rev 2014; 27: 727 - 52. [PubMed][CrossRef]
- 4.
List of prakaryotic names with standing in nomenclature. Genus Mycobacterium. www.bacterio.net/mycobacterium.html (27.8.2018).
- 5.
Koh WJ, Jeon K, Lee NY et al. Clinical significance of differentiation of Mycobacterium massiliense from Mycobacterium abscessus. Am J Respir Crit Care Med 2011; 183: 405 - 10. [PubMed][CrossRef]
- 6.
Bryant JM, Grogono DM, Greaves D et al. Whole-genome sequencing to identify transmission of Mycobacterium abscessus between patients with cystic fibrosis: a retrospective cohort study. Lancet 2013; 381: 1551 - 60. [PubMed][CrossRef]
- 7.
Aitken ML, Limaye A, Pottinger P et al. Respiratory outbreak of Mycobacterium abscessus subspecies massiliense in a lung transplant and cystic fibrosis center. Am J Respir Crit Care Med 2012; 185: 231 - 2. [PubMed][CrossRef]
- 8.
Khan K, Wang J, Marras TK. Nontuberculous mycobacterial sensitization in the United States: national trends over three decades. Am J Respir Crit Care Med 2007; 176: 306 - 13. [PubMed][CrossRef]
- 9.
Stout JE, Koh WJ, Yew WW. Update on pulmonary disease due to non-tuberculous mycobacteria. Int J Infect Dis 2016; 45: 123 - 34. [PubMed][CrossRef]
- 10.
Winthrop KL. Pulmonary disease due to nontuberculous mycobacteria: an epidemiologist's view. Future Microbiol 2010; 5: 343 - 5. [PubMed][CrossRef]
- 11.
Infectious Disease Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med 2007; 175: 367 - 416. [PubMed][CrossRef]
- 12.
Floto RA, Olivier KN, Saiman L et al. US Cystic Fibrosis Foundation and European Cystic Fibrosis Society consensus recommendations for the management of non-tuberculous mycobacteria in individuals with cystic fibrosis: executive summary. Thorax 2016; 71: 88 - 90. [PubMed][CrossRef]
- 13.
Haworth CS, Banks J, Capstick T et al. British Thoracic Society guidelines for the management of non-tuberculous mycobacterial pulmonary disease (NTM-PD). Thorax 2017; 72 (Suppl 2): ii1 - 64. [PubMed][CrossRef]
- 14.
Koh WJ, Yu CM, Suh GY et al. Pulmonary TB and NTM lung disease: comparison of characteristics in patients with AFB smear-positive sputum. Int J Tuberc Lung Dis 2006; 10: 1001 - 7. [PubMed]
- 15.
Kim YK, Hahn S, Uh Y et al. Comparable characteristics of tuberculous and non-tuberculous mycobacterial cavitary lung diseases. Int J Tuberc Lung Dis 2014; 18: 725 - 9. [PubMed][CrossRef]
- 16.
Chu H, Zhao L, Xiao H et al. Prevalence of nontuberculous mycobacteria in patients with bronchiectasis: a meta-analysis. Arch Med Sci 2014; 10: 661 - 8. [PubMed][CrossRef]
- 17.
Lee AR, Lee J, Choi SM et al. Phenotypic, immunologic, and clinical characteristics of patients with nontuberculous mycobacterial lung disease in Korea. BMC Infect Dis 2013; 13: 558. [PubMed][CrossRef]
- 18.
Kartalija M, Ovrutsky AR, Bryan CL et al. Patients with nontuberculous mycobacterial lung disease exhibit unique body and immune phenotypes. Am J Respir Crit Care Med 2013; 187: 197 - 205. [PubMed][CrossRef]
- 19.
Kim RD, Greenberg DE, Ehrmantraut ME et al. Pulmonary nontuberculous mycobacterial disease: prospective study of a distinct preexisting syndrome. Am J Respir Crit Care Med 2008; 178: 1066 - 74. [PubMed][CrossRef]
- 20.
Ziedalski TM, Kao PN, Henig NR et al. Prospective analysis of cystic fibrosis transmembrane regulator mutations in adults with bronchiectasis or pulmonary nontuberculous mycobacterial infection. Chest 2006; 130: 995 - 1002. [PubMed][CrossRef]
- 21.
Hanak V, Kalra S, Aksamit TR et al. Hot tub lung: presenting features and clinical course of 21 patients. Respir Med 2006; 100: 610 - 5. [PubMed][CrossRef]
- 22.
Marras TK, Wallace RJ, Koth LL et al. Hypersensitivity pneumonitis reaction to Mycobacterium avium in household water. Chest 2005; 127: 664 - 71. [PubMed][CrossRef]
- 23.
Jeong YJ, Lee KS, Koh WJ et al. Nontuberculous mycobacterial pulmonary infection in immunocompetent patients: comparison of thin-section CT and histopathologic findings. Radiology 2004; 231: 880 - 6. [PubMed][CrossRef]
- 24.
Chung MJ, Lee KS, Koh WJ et al. Thin-section CT findings of nontuberculous mycobacterial pulmonary diseases: comparison between Mycobacterium avium-intracellulare complex and Mycobacterium abscessus infection. J Korean Med Sci 2005; 20: 777 - 83. [PubMed][CrossRef]
- 25.
van Ingen J. Microbiological diagnosis of nontuberculous mycobacterial pulmonary disease. Clin Chest Med 2015; 36: 43 - 54. [PubMed][CrossRef]
- 26.
Brown-Elliott BA, Nash KA, Wallace RJ. Antimicrobial susceptibility testing, drug resistance mechanisms, and therapy of infections with nontuberculous mycobacteria. Clin Microbiol Rev 2012; 25: 545 - 82. [PubMed][CrossRef]
- 27.
van Ingen J, Boeree MJ, van Soolingen D et al. Resistance mechanisms and drug susceptibility testing of nontuberculous mycobacteria. Drug Resist Updat 2012; 15: 149 - 61. [PubMed][CrossRef]
- 28.
Brown-Elliott BA, Iakhiaeva E, Griffith DE et al. In vitro activity of amikacin against isolates of Mycobacterium avium complex with proposed MIC breakpoints and finding of a 16S rRNA gene mutation in treated isolates. J Clin Microbiol 2013; 51: 3389 - 94. [PubMed][CrossRef]
- 29.
Xu HB, Jiang RH, Li L. Treatment outcomes for Mycobacterium avium complex: a systematic review and meta-analysis. Eur J Clin Microbiol Infect Dis 2014; 33: 347 - 58. [PubMed][CrossRef]
- 30.
Nash KA, Brown-Elliott BA, Wallace RJ. A novel gene, erm(41), confers inducible macrolide resistance to clinical isolates of Mycobacterium abscessus but is absent from Mycobacterium chelonae. Antimicrob Agents Chemother 2009; 53: 1367 - 76. [PubMed][CrossRef]
- 31.
Griffith DE, Brown-Elliott BA, Langsjoen B et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med 2006; 174: 928 - 34. [PubMed][CrossRef]
- 32.
Serisier DJ, Martin ML, McGuckin MA et al. Effect of long-term, low-dose erythromycin on pulmonary exacerbations among patients with non-cystic fibrosis bronchiectasis: the BLESS randomized controlled trial. JAMA 2013; 309: 1260 - 7. [PubMed][CrossRef]
- 33.
Altenburg J, de Graaff CS, Stienstra Y et al. Effect of azithromycin maintenance treatment on infectious exacerbations among patients with non-cystic fibrosis bronchiectasis: the BAT randomized controlled trial. JAMA 2013; 309: 1251 - 9. [PubMed][CrossRef]
- 34.
Jenkins PA, Campbell IA, Banks J et al. Clarithromycin vs ciprofloxacin as adjuncts to rifampicin and ethambutol in treating opportunist mycobacterial lung diseases and an assessment of Mycobacterium vaccae immunotherapy. Thorax 2008; 63: 627 - 34. [PubMed][CrossRef]
- 35.
Research Committee of the British Thoracic Society. First randomised trial of treatments for pulmonary disease caused by M avium intracellulare, M malmoense, and M xenopi in HIV negative patients: rifampicin, ethambutol and isoniazid versus rifampicin and ethambutol. Thorax 2001; 56: 167 - 72. [PubMed][CrossRef]
- 36.
Kobashi Y, Matsushima T, Oka M. A double-blind randomized study of aminoglycoside infusion with combined therapy for pulmonary Mycobacterium avium complex disease. Respir Med 2007; 101: 130 - 8. [PubMed][CrossRef]
- 37.
Fujita M, Kajiki A, Tao Y et al. The clinical efficacy and safety of a fluoroquinolone-containing regimen for pulmonary MAC disease. J Infect Chemother 2012; 18: 146 - 51. [PubMed][CrossRef]
- 38.
Miwa S, Shirai M, Toyoshima M et al. Efficacy of clarithromycin and ethambutol for Mycobacterium avium complex pulmonary disease. A preliminary study. Ann Am Thorac Soc 2014; 11: 23 - 9. [PubMed][CrossRef]
- 39.
Griffith DE, Brown BA, Murphy DT et al. Initial (6-month) results of three-times-weekly azithromycin in treatment regimens for Mycobacterium avium complex lung disease in human immunodeficiency virus-negative patients. J Infect Dis 1998; 178: 121 - 6. [PubMed][CrossRef]
- 40.
Griffith DE, Brown BA, Cegielski P et al. Early results (at 6 months) with intermittent clarithromycin-including regimens for lung disease due to Mycobacterium avium complex. Clin Infect Dis 2000; 30: 288 - 92. [PubMed][CrossRef]
- 41.
Jeong BH, Jeon K, Park HY et al. Intermittent antibiotic therapy for nodular bronchiectatic Mycobacterium avium complex lung disease. Am J Respir Crit Care Med 2015; 191: 96 - 103. [PubMed][CrossRef]
- 42.
Wallace RJ, Brown-Elliott BA, McNulty S et al. Macrolide/Azalide therapy for nodular/bronchiectatic mycobacterium avium complex lung disease. Chest 2014; 146: 276 - 82. [PubMed][CrossRef]
- 43.
Olivier KN, Shaw PA, Glaser TS et al. Inhaled amikacin for treatment of refractory pulmonary nontuberculous mycobacterial disease. Ann Am Thorac Soc 2014; 11: 30 - 5. [PubMed][CrossRef]
- 44.
Jo KW, Kim S, Lee JY et al. Treatment outcomes of refractory MAC pulmonary disease treated with drugs with unclear efficacy. J Infect Chemother 2014; 20: 602 - 6. [PubMed][CrossRef]
- 45.
Koh WJ, Hong G, Kim SY et al. Treatment of refractory Mycobacterium avium complex lung disease with a moxifloxacin-containing regimen. Antimicrob Agents Chemother 2013; 57: 2281 - 5. [PubMed][CrossRef]
- 46.
Jarand J, Davis JP, Cowie RL et al. Long-term follow-up of Mycobacterium avium complex lung disease in patients treated with regimens including clofazimine and/or rifampin. Chest 2016; 149: 1285 - 93. [PubMed][CrossRef]
- 47.
Ferro BE, Meletiadis J, Wattenberg M et al. Clofazimine prevents the regrowth of Mycobacterium abscessus and Mycobacterium avium type strains exposed to amikacin and clarithromycin. Antimicrob Agents Chemother 2015; 60: 1097 - 105. [PubMed][CrossRef]
- 48.
Field SK, Fisher D, Cowie RL. Mycobacterium avium complex pulmonary disease in patients without HIV infection. Chest 2004; 126: 566 - 81. [PubMed][CrossRef]
- 49.
Lee BY, Kim S, Hong Y et al. Risk factors for recurrence after successful treatment of Mycobacterium avium complex lung disease. Antimicrob Agents Chemother 2015; 59: 2972 - 7. [PubMed][CrossRef]
- 50.
Park HK, Koh WJ, Shim TS et al. Clinical characteristics and treatment outcomes of Mycobacterium kansasii lung disease in Korea. Yonsei Med J 2010; 51: 552 - 6. [PubMed][CrossRef]
- 51.
Griffith DE. Management of disease due to Mycobacterium kansasii. Clin Chest Med 2002; 23: 613 - 21, vi. [PubMed][CrossRef]
- 52.
Philley JV, Griffith DE. Treatment of slowly growing mycobacteria. Clin Chest Med 2015; 36: 79 - 90. [PubMed][CrossRef]
- 53.
Pulmonary disease caused by M. malmoense in HIV negative patients: 5-yr follow-up of patients receiving standardised treatment. Eur Respir J 2003; 21: 478 - 82. [PubMed][CrossRef]
- 54.
Hoefsloot W, van Ingen J, de Lange WC et al. Clinical relevance of Mycobacterium malmoense isolation in The Netherlands. Eur Respir J 2009; 34: 926 - 31. [PubMed][CrossRef]
- 55.
Brown-Elliott BA, Philley JV. Rapidly growing Mycobacteria. Microbiol Spectr 2017; 5: 5. [PubMed][CrossRef]
- 56.
Jeon K, Kwon OJ, Lee NY et al. Antibiotic treatment of Mycobacterium abscessus lung disease: a retrospective analysis of 65 patients. Am J Respir Crit Care Med 2009; 180: 896 - 902. [PubMed][CrossRef]
- 57.
Jarand J, Levin A, Zhang L et al. Clinical and microbiologic outcomes in patients receiving treatment for Mycobacterium abscessus pulmonary disease. Clin Infect Dis 2011; 52: 565 - 71. [PubMed][CrossRef]
- 58.
Griffith DE, Girard WM, Wallace RJ. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis 1993; 147: 1271 - 8. [PubMed][CrossRef]
- 59.
Sugino K, Kobayashi M, Iwata M et al. Successful treatment with pneumonectomy for pulmonary Mycobacterium abscessus infection. Intern Med 2009; 48: 459 - 63. [PubMed][CrossRef]
- 60.
Koh WJ, Kim YH, Kwon OJ et al. Surgical treatment of pulmonary diseases due to nontuberculous mycobacteria. J Korean Med Sci 2008; 23: 397 - 401. [PubMed][CrossRef]
- 61.
Mitchell JD. Surgical approach to pulmonary nontuberculous mycobacterial infections. Clin Chest Med 2015; 36: 117 - 22. [PubMed][CrossRef]