MAIN POINTS
Our results confirm that catheter-based left atrial appendage closure is feasible in most patients
Catheter-based left atrial appendage closure provides complete or near complete closure of the left atrial appendage in most patients
Catheter-based left atrial appendage closure entails a risk of serious complications
Atrial fibrillation is a common condition, with a prevalence of 3.2 % in those over 20 years of age (1). The condition is associated with increased morbidity and mortality, and the risk of stroke is 4–5 times higher with atrial fibrillation compared to sinus rhythm (2). After hypertension, atrial fibrillation is the second most common cause of stroke, responsible for 20–30 % of all cases (3). Stroke caused by atrial fibrillation is associated with high morbidity and mortality (4).
Atrial fibrillation increases the risk of thrombosis owing to stasis and turbulence in the atria, endothelial dysfunction and hypercoagulability; that is, all three criteria in Virchow's triad are fulfilled (5). The likelihood of stroke in cases of atrial fibrillation depends on various risk factors that can be summarised in the patient's CHA2DS2-VASc score (6). Likewise, the risk of serious bleeding depends on risk factors that can be summarised in the patient's HAS-BLED score (7) (Table 1).
Table 1
The two scoring instruments CHA2DS2-VASc (6) and HAS-BLED (7). TIA = transient ischaemic attack, TE = thromboembolism, INR = international normalised ratio, NSAID = non-steroidal anti-inflammatory agent
| CHA2DS2-VASc-score |
||
|---|---|---|
| Risk factor |
Score |
|
| Congestive heart failure |
1 |
|
| Hypertension |
Blood pressure > 140/90 mmHg or current hypertension treatment |
1 |
| Age ≥ 75 years |
2 |
|
| Diabetes |
1 |
|
| Stroke / TIA / TE |
Stroke/ TIA / thromboembolism |
2 |
| Vascular disease |
Vascular disease (myocardial infarction, peripheral atherosclerosis, aortic plaque) |
1 |
| Age 65–74 years |
Age 65–74 years |
1 |
| Sex (i.e. female gender) |
Female |
1 |
| Total |
||
| HAS-BLED score | ||
| Risk factor |
Score |
|
| Hypertension |
Uncontrolled hypertension, > 160 mmHg systolic |
1 |
| Abnormal renal and liver function |
Impaired renal and liver function |
1 or 2 |
| Stroke |
Prior history of stroke |
1 |
| Bleeding |
Bleeding history or predisposition |
1 |
| Labile INR |
INR in therapeutic range < 60 % |
1 |
| Elderly |
Age > 65 years |
1 |
| Drugs or alcohol |
Antiplatelet therapy or NSAIDs |
1 or 2 |
| Total |
||
In the event of a CHA2DS2-VASc score ≥ 2 for men and ≥ 3 for women, European guidelines strongly recommend initiating anticoagulant therapy with a vitamin K antagonist (warfarin) or a non-vitamin K-dependent oral anticoagulant (NOAC) (dabigatran, rivaroxaban, apixaban or edoxaban) (3). In the event of a CHA2DS2-VASc score ≥ 1 for men and ≥ 2 for women, the recommendation is somewhat weaker (3).
Warfarin therapy reduces the risk of stroke by 64 % compared to placebo (8). NOACs have been shown to be at least as efficacious as warfarin in preventing thromboembolism in cases of atrial fibrillation, with reduced risk of intracranial haemorrhage (9).
Anticoagulant therapy is contraindicated in more than one in ten patients with atrial fibrillation (10). However, in clinical practice, only about 60 % of those with an indication for anticoagulant therapy are found to receive this treatment (11). Common reasons for withholding treatment are previous bleeding events and a perceived high risk of bleeding, frequent falls, and frailty (3).
Data from sources including an autopsy study show that as many as 90 % of cardiac thrombi in cases of atrial fibrillation form in the left atrial appendage (12, 13). Surgical excision of the appendage has been performed in conjunction with other cardiovascular surgery for many years, but complete closure of the appendage has been difficult to achieve (14). As an alternative to anticoagulant therapy, mechanical plugs have been developed in recent years for catheter-based left atrial appendage closure (15). In Norway, this procedure is offered at Oslo University Hospital, Haukeland University Hospital and St Olavs Hospital (15).
Here we present procedure outcomes, as well as complications and clinical events during a one-year follow-up after attempted catheter-based left atrial appendage closure at Oslo University Hospital during the period September 2014–April 2016.
Material and method
All patients in this series were treated on the basis of clinical indication. At Oslo University Hospital, this refers predominantly to patients with non-valvular atrial fibrillation (paroxysmal, persistent or chronic) with CHA2DS2-VASc score ≥ 1 for men and ≥ 2 for women and in whom anticoagulant therapy is considered contraindicated.
Transthoracic echocardiography was performed beforehand to exclude, for example, valvular dysfunction requiring surgery. Patients were informed verbally and in writing about the indication for the procedure and its execution, and about the possibility of complications, and gave written consent to participate in the study. They received check-ups at approximately 50 days and one year after the procedure. The incidence of new cerebrovascular events, bleeding and complications related to the treatment was recorded.
The Regional Ethics Committee declined to process our application because the study was perceived to be quality assurance and not research. The study was subsequently approved by the Data Protection Officer at Oslo University Hospital. The number of patients is small and data are presented as median (range) or absolute value.
Catheter-based left atrial appendage closure was performed in fasting patients under general anaesthesia with intubation, under visual guidance with transoesophageal echocardiography and X-ray fluoroscopy. Before the procedure (preferably the previous day), a CT scan of the heart is performed to map the anatomy of the left atrial appendage and to exclude thrombus formation in the left atrial appendage or atrium, which would be considered a contraindication. Patients who are not receiving antiplatelet therapy are pretreated with acetylsalicyclic acid (aspirin) 300 mg orally, or clopidogrel 300 mg orally in cases of acetylsalicyclic acid allergy.
Venous access is established through the femoral vein in the groin, and transseptal puncture is then performed guided by transoesophageal echocardiography and fluoroscopy. Following transseptal puncture, heparin is administered intravenously to achieve an activated clotting time > 250 s.
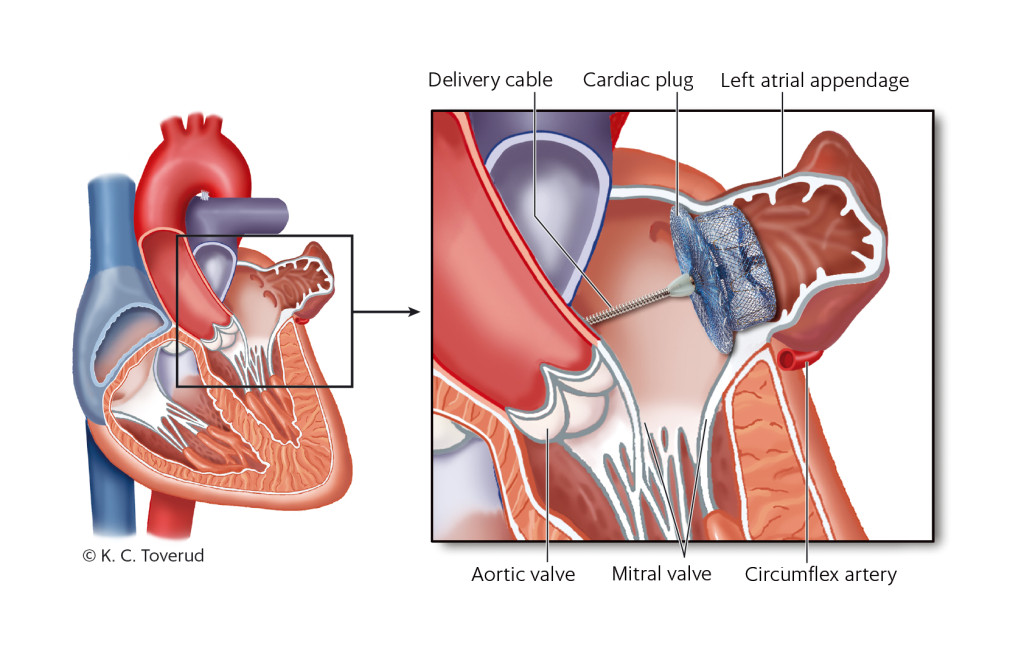
The size of the cardiac plug is decided on the basis of the CT scan, using measurements of the left atrial appendage ostium and the landing zone in the 'neck' of the appendage. At Oslo University Hospital, we have so far used only Amplatzer cardiac plugs (St. Jude Medical, St. Paul, MN). These are available in eight different sizes from 16 mm to 34 mm, and the delivery system has a diameter of 4–4.5 mm.
The Amplatzer plug has a 'lobe' to secure it within the 'neck' of the left atrial appendage, and a 'disc' to cover the ostium (Figure 1). The lobe is unfolded inside the left atrial appendage, followed by the disc. When an acceptable position is achieved according to established guidelines, the plug is released; if necessary the plug can be repositioned or another size used instead (15). Antibiotic prophylaxis is provided in association with the procedure in the form of cefalotin, or clindamycin in cases of penicillin allergy. The delivery system is removed, the access site in the groin compressed and the patient woken.
Patients are monitored for 24 hours in hospital after the procedure. Before returning home, they undergo transthoracic echocardiography and a chest X-ray.
The cardiac plug will undergo endothelialisation over the course of 3–6 months. Patients must receive antiplatelet therapy throughout this period to prevent thrombus formation on the plug. Patients are discharged with acetylsalicylic acid 75 mg × 1, or clopidogrel 75 mg × 1, as appropriate.
After approximately 50 days, patients attended an outpatient check-up with transoesophageal echocardiography. In the absence of thrombus formation on the plug or significant leakage (> 3 mm between the disc and left atrial appendage ostium), the recommendation is for acetylsalicylic acid to be discontinued after six months. If there is any other indication for platelet inhibition, such as a coronary stent, antiplatelet therapy should be continued indefinitely. Patients should receive endocarditis prophylaxis during dental appointments and other high risk procedures for the first six months after implantation. A final outpatient check-up is performed after one year, again with transoesophageal echocardiography.
Results
A total of 27 patients were included in the study – 13 were treated at Rikshospitalet and 14 at Ullevål. At each site, two interventional cardiologists performed all procedures (KL and AH at Rikshospitalet, AAA and AO at Ullevål).
Patient characteristics are shown in Table 2. The median CHA2DS2-VASc score was 4 (range 0–8) and the median HAS-BLED score was 2 (range 0–3). The indication in 26 of the patients was a previous bleeding event with or without anticoagulant therapy. One patient had undergone ablation therapy for arrhythmia; because complete electrical isolation of the left atrial appendage had been performed, the electrophysiologist recommended catheter-based left atrial appendage closure in addition to anticoagulant therapy, despite CHA2DS2-VASc and HAS-BLED scores of 0. In another patient, implantation of a cardiac plug was not possible for anatomical reasons; implantation was performed in the remaining 26 patients.
Table 2
Patient characteristics, comorbid conditions, indication for the procedure and medication after the procedure
| Total number of patients |
27 |
|---|---|
| Median age (years) (range) |
72 (56–86) |
| Age < 65 |
2 |
| Age 65–74 |
14 |
| Age ≥ 75 |
11 |
| Women |
7 |
| Other conditions |
|
| Permanent atrial fibrillation |
17 |
| Paroxysmal/persistent atrial fibrillation |
10 |
| Heart failure |
6 |
| Hypertension |
22 |
| Diabetes mellitus |
5 |
| Previous stroke or TIA |
7 |
| Vascular disease |
15 |
| Indication for percutaneous left atrial appendage closure |
|
| Previous intracerebral haemorrhage |
19 |
| Gastrointestinal haemorrhage/urinary tract haemorrhage |
7 |
| Other indication |
1 |
| Blood-thinning medication on discharge |
|
| Acetylsalicyclic acid monotherapy |
15 |
| Clopidogrel monotherapy |
1 |
| Acetylsalicyclic acid and clopidogrel |
9 |
| Anticoagulant therapy (apixaban) |
1 |
| Blood-thinning medication at one-year follow-up |
|
| Acetylsalicyclic acid monotherapy |
11 |
| Clopidogrel monotherapy |
1 |
| Acetylsalicyclic acid and clopidogrel |
0 |
| Anticoagulant therapy (1 × apixaban, 1 × warfarin) |
2 |
In 21 patients, the correct sized plug was chosen at the first attempt, in five patients the plug was replaced by one of a different size. The median size of the cardiac plug was 26 mm (range 18–34 mm). The median procedure duration, measured from establishment of venous access in the groin to removal of the delivery sheath, was 69 minutes (range 42–175 min). The median contrast medium consumption was 100 ml (range 40–200 ml).
Complications
Procedure-related complications. One patient suffered a stroke during the procedure. Subsequent MRI of the brain revealed fresh emboli, which gave rise to mild cognitive impairment. One patient experienced a groin bleed and received a transfusion with three units of erythrocytes.
Complications related to catheter-based left atrial appendage closure and thromboembolic events from time of discharge to one year later. The patient in whom a cardiac plug could not be successfully implanted died 12 months after the procedure as a result of stroke. Another patient was admitted five months after the procedure with circulatory shock with cardiac tamponade owing to haemopericardium, and underwent percutaneous pericardial effusion drainage. Three patients experienced a TIA several months after the procedure. There were no cases of intracranial or gastrointestinal haemorrhage.
Echocardiography
Of the 26 patients who had a cardiac plug implanted, 25 attended a follow-up appointment with transoesophageal echocardiography after a median of 50 days (range 20–188 days). Sixteen patients attended a one-year follow-up appointment.
Complete left atrial appendage closure was observed in 15 patients in their most recent transoesophageal echocardiogram. Trivial leakage between the disc and the left atrial appendage ostium (≤ 3 mm) was seen in seven patients, and significant leakage (> 3 mm) in three.
Of the three patients who experienced a TIA in the follow-up period, one had trivial leakage, while the other two had complete left atrial appendage closure. No patient had thrombus formation on the cardiac plug.
Discussion
Catheter-based left atrial appendage closure is a new method for preventing thromboembolism in cases of atrial fibrillation. In a randomised trial with 707 patients, closure with the Watchman plug (Boston Scientific, St. Paul, MN) was non-inferior to warfarin therapy after 18 months (16), and produced significantly better results than warfarin therapy after an average follow-up of 3.8 years (17).
A composite endpoint comprising stroke, systemic embolism and cardiovascular mortality was used to evaluate treatment efficacy. Only patients who tolerated warfarin were included in the study; in the group that underwent catheter-based left atrial appendage closure, warfarin therapy was mandatory for 45 days after implantation.
Owing to a procedure-related complication rate of 8.7 % in the first seven days following the procedure (4.0 % pericardial effusion requiring drainage, 1.1 % ischaemic stroke, 0.4 % embolisation of the cardiac plug and 3.2 % vascular complications) (18), the US Food and Drug Administration (FDA) ruled that further evidence would be required before the product could be approved.
This formed the basis for a new study, in which 407 patients with atrial fibrillation were randomised to catheter-based left atrial appendage closure or to anticoagulant therapy with warfarin alone (18). At least 25 % of the procedures were required to be performed at institutions without previous experience with the method. In this study, left atrial appendage closure did not prove as efficacious as anticoagulant therapy with respect to the composite endpoint of stroke, systemic embolism and cardiovascular mortality after 18 months of follow-up. The incidence of procedure-related complications was reduced to 4.5 %.
A similar incidence of complications (4.2 %) was reported in a registry of 566 patients who had undergone catheter-based left atrial appendage closure (18). In 2015, on the basis of the available evidence, the FDA approved closure with the Watchman plug as an alternative to anticoagulant therapy. The procedure has been shown to be cost-effective compared to pharmaceutical treatment alone (19).
In our dataset, clinical events occurred in six out of 27 patients. The most serious complication (death due to stroke) occurred in the patient in whom closure could not be successfully performed. Of the 26 patients who underwent catheter-based left atrial appendage closure, two experienced procedure-related complications, which is in line with the numbers in the Watchman studies (16, 18) and a registry study with Amplatzer cardiac plugs (6.5 %) (20). One patient has had life-threatening complications, which we believe to be the result of late atrial appendage perforation. This is a rare complication following catheter-based closure that has been described previously (21). What constitutes an acceptable incidence of complications must be judged in relation to the anticipated efficacy in terms of stroke prevention.
Our dataset is small, and in the absence of a control group it is difficult to draw conclusions about treatment efficacy. The median CHA2DS2-VASc score in our dataset is 4, which corresponds to an expected incidence of stroke/TIA of approximately 4 % per year (22). During our one-year follow-up, four patients experienced stroke or TIA, a number that might appear on the high side.
One of the cases of TIA was probably attributable to embolisation as a result of carotid artery stenosis, against which catheter-based left atrial appendage closure of course offers no protection. It is therefore important to investigate other causes of stroke also in patients with atrial fibrillation. In one of the patients with TIA, brain MRI revealed a lacunar infarct, which is usually attributable to small vessel disease and not cardiac embolism.
It is worth noting that none of the patients experienced intracranial or gastrointestinal haemorrhage during the period from discharge to one-year follow-up. The median HAS-BLED score was 2, which corresponds to an estimated risk of severe bleeding of 1.9 % per year (22). A low incidence of bleeding after catheter-based left atrial appendage closure is consistent with findings in other studies (16, 20).
In the United States, catheter-based left atrial appendage closure is approved as an alternative to anticoagulant therapy. In Europe, most catheter-based closures are performed in patients in whom anticoagulant therapy is considered contraindicated. This is a paradox, since the randomised trials of the closure procedure included patients who tolerated warfarin (16, 18). The efficacy of anticoagulant therapy in preventing stroke in patients with atrial fibrillation is very well documented (8), and in the absence of contraindications, anticoagulant therapy is the clear first-line treatment.
Intracranial haemorrhage during ongoing anticoagulant therapy has been the indication for catheter-based left atrial appendage closure in the majority of patients at Oslo University Hospital. Those with a history of intracerebral haemorrhage usually do not receive thromboembolism prophylaxis.
Acetylsalicylic acid has been used as an alternative to anticoagulant therapy, but offers poor protection against thromboembolism in cases of atrial fibrillation (22 % stroke reduction with acetylsalicylic acid versus 64 % with warfarin) (8). The absolute reduction in haemorrhage risk with acetylsalicylic acid is modest compared to the bleeding risk with anticoagulant therapy (8). European guidelines on atrial fibrillation include only a weak recommendation for catheter-based left atrial appendage closure in patients for whom anticoagulant therapy is contraindicated or who are at high risk of bleeding (3).
Various approaches are adopted with respect to blood-thinning after left atrial appendage closure. There remains a risk of thrombosis until the cardiac plug undergoes endothelialisation after 3–6 months. In the randomised trials with the Watchman plug, the standard protocol specified anticoagulant therapy for the first 45 days after closure, followed by dual antiplatelet therapy with acetylsalicylic acid and clopidogrel for six months, and acetylsalicylic acid monotherapy thereafter (16, 17).
Dual antiplatelet therapy for six months after catheter-based left atrial appendage closure, followed by acetylsalicylic acid monotherapy, has been shown to be an equally valid alternative (23) and most institutions recommend such a regime (15).
However, good results have also been obtained with acetylsalicylic acid monotherapy following the procedure (24), and dual antiplatelet therapy entails an increased risk of bleeding compared to acetylsalicylic acid monotherapy (25). On this basis, we at Oslo University Hospital now recommend acetylsalicylic acid monotherapy.
If the 45-day follow-up reveals satisfactory closure and the absence of thrombus formation on the cardiac plug, then discontinuation of acetylsalicylic acid after six months is recommended if there is no other indication for use. If another indication is present, then acetylsalicylic acid should be continued indefinitely. The reason that many patients in our dataset nevertheless received dual antiplatelet therapy was that they had undergone percutaneous coronary intervention or percutaneous aortic valve replacement close to catheter-based left atrial appendage closure.
Patients with atrial fibrillation who require these procedures and who have an indication for anticoagulant therapy represent a major challenge for the clinician. Anticoagulant therapy offers poor protection against stent thrombosis, dual antiplatelet therapy offers poor protection against thromboembolism, and the combination of anticoagulant and antiplatelet therapy results in a high risk of bleeding (26). Catheter-based left atrial appendage closure in combination with antiplatelet therapy has therefore been proposed for such patients (15).
Several cardiac plugs have been approved for use in Europe (27). Only the Watchman plug has been used in randomised trials (16, 17), and this is the only cardiac plug approved in the United States. In spite of this, Amplatzer cardiac plugs are the most frequently used in Europe, and we have also opted for these at Oslo University Hospital. Registry data from more than 1 000 patients have shown that use of Amplatzer cardiac plugs results in fewer cases of stroke and bleeding than would be expected from CHA2DS2-VASc and HAS-BLED scores, and the incidence of procedure-related complications is at the same level as for the Watchman plug (20). Nevertheless, we acknowledge that the lack of randomised trials of Amplatzer cardiac plugs is a weakness.
More evidence is required in general before extensive use of catheter-based left atrial appendage closure can be recommended. We believe this treatment should initially be reserved for patients at high risk of stroke and with a contraindication for anticoagulant therapy, as assessed on a case-by-case basis.
To date, no randomised studies have compared percutaneous catheter-based left atrial appendage closure with NOAC therapy, with antiplatelet therapy alone, or with no treatment. In a forthcoming Scandinavian study, 750 patients with atrial fibrillation and intracerebral haemorrhage within the last six months will be randomised to either catheter-based left atrial appendage closure with an Amplatzer cardiac plug or pharmaceutical treatment. The results of this study are awaited with great interest (28). We hope that Norwegian neurologists will actively recruit patients to the study.
All authors have participated in product training for Amplatzer cardiac plugs arranged and funded by St. Jude Medical.
- 10.
O'Brien EC, Holmes DN, Ansell JE et al. Physician practices regarding contraindications to oral anticoagulation in atrial fibrillation: findings from the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF) registry. Am Heart J 2014; 167: 601 - 609.e1. [PubMed][CrossRef]
- 22.
CHA2DS2-VASc / HAS-BLED / EHRA atrial fibrillation risk score calculator. https://chadsvasc.org/ (19.12.2017).
- 23.
Reddy VY, Möbius-Winkler S, Miller MA et al. Left atrial appendage closure with the Watchman device in patients with a contraindication for oral anticoagulation: the ASAP study (ASA Plavix Feasibility Study With Watchman Left Atrial Appendage Closure Technology). J Am Coll Cardiol 2013; 61: 2551 - 6. [PubMed][CrossRef]
- 26.
Lip GY, Windecker S, Huber K et al. Management of antithrombotic therapy in atrial fibrillation patients presenting with acute coronary syndrome and/or undergoing percutaneous coronary or valve interventions: a joint consensus document of the European Society of Cardiology Working Group on Thrombosis, European Heart Rhythm Association (EHRA), European Association of Percutaneous Cardiovascular Interventions (EAPCI) and European Association of Acute Cardiac Care (ACCA) endorsed by the Heart Rhythm Society (HRS) and Asia-Pacific Heart Rhythm Society (APHRS). Eur Heart J 2014; 35: 3155 - 79. [PubMed][CrossRef]
- 28.
Prevention of Stroke by Left Atrial Appendage Closure in Atrial Fibrillation Patients After Intracerebral Hemorrhage. 2017. Prevention of Stroke by Left Atrial Appendage Closure in Atrial Fibrillation Patients After Intracerebral Hemorrhage. https://clinicaltrials.gov/ct2/show/NCT02830152 (19.12.2017).