MAIN MESSAGE
Most hospitals had local guidelines for treatment of sepsis
Local and national guidelines were largely consistent
Certain aspects of the treatment are frequently questioned and varied locally
Sepsis is a systemic reaction to an infection which affects the patient's general condition and organ dysfunction develops. The condition occurs relatively frequently, with an incidence of approximately 0.5 per 1 000 per year (1). The hospital mortality rate for sepsis is approximately 10 %, but in cases of septic shock it rises to more than 40 % (Box 1) (2). Early detection and treatment are essential to reduce mortality, a fact underscored by the official Norwegian patient safety programme (3). Early initiation of antibiotics treatment is also crucial for the prognosis (4).
Box 1 Sepsis. There is international consensus on the following definitions (2). The Sequential Organ Failure Assessment (SOFA) can be used to detect organ dysfunction1
Sepsis: Life-threatening organ dysfunction1 caused by a dysregulated host response to infection
Septic shock: A vasopressor is required to maintain a mean arterial pressure ≥ 65 mm Hg and serum lactate level ≥ 2 mmol/L in the absence of hypovolemia
There are international (5) as well as national (6, 7) guidelines for treatment of sepsis. The national guidelines are largely consistent with the international ones, although the latter are somewhat more detailed (Table 1) (5–7). In addition, nearly all Norwegian hospitals have prepared local guidelines. Individual therapists may thus need to relate to guidelines issued at various levels when they encounter a seriously ill sepsis patient.
Table 1
Comparison of Norwegian (6, 7) and international (5) guidelines for treatment of sepsis
| Norwegian guidelines | International guidelines | |
|---|---|---|
| Treatment of infection | ||
| Elimination of the focus of infection | + | + |
| Antibiotics | + | + |
| Treatment of organ dysfunction | ||
| Fluid therapy | + | + |
| Vasoactive drugs1 | + | + |
| Inotropic drugs | + | + |
| Glucocorticoids | + | + |
| Blood/blood products | + | + |
| Respiratory support | + | + |
| Glucose control | + | + |
| Bicarbonate for acidosis | + | + |
| Other treatment | ||
| Prevention of infections in ventilator patients | - | + |
| Deep vein thrombosis prophylaxis | - | + |
| Stress ulcer prophylaxis | - | + |
| Dietary recommendations | - | + |
1International guidelines mention adrenaline as an alternative drug, but there is no mention of this in the Norwegian ones.
Since the disease develops quickly and is potentially life-threatening, a rapid and correct diagnosis may be crucial to the outcome. Implementation of and compliance with guidelines has an effect on survival (8). The objective of this study was to assess the extent to which the local guidelines were consistent with the national ones.
Material and method
The national guidelines used in this study included the sepsis document in the Norsk legemiddelhåndbok [Norwegian Medicines Manual] (6) and the Nasjonal faglig retningslinje for bruk av antibiotika i sykehus [National clinical guidelines for the use of antibiotics in hospitals] (7) from the Directorate of Health. There was a large degree of consistency between these, although they emphasise different elements. The sepsis document from the Directorate of Health deals mainly with the use of antibiotics, while the sepsis document in the Norwegian Medicines Manual mainly emphasises organ support.
The local guidelines were collected by sending emails to the senior consultant at the department of infectious diseases (alternatively the medical department) in all Norwegian hospitals that have an emergency internal medicine department (n = 48). Relevant hospitals were identified through a search on the government web page 'Overview of the country's health enterprises' (9).
We grouped the hospitals into regional hospitals (the one appointed to the role of main hospital in each health region), emergency hospitals (those that have at least an emergency internal medicine department, an anaesthetist on duty around the clock and planned surgery) and large emergency hospitals (with a catchment area of at least 60 000–80 000 inhabitants and a broad range of emergency services), cf. the National Health and Hospital Plan (10).
We asked for guidelines for the use of antibiotics as well as for provision of organ support. The first request was sent on 3 February 2016, and the last response came in on 6 September 2016.
The local guidelines were compared to the national ones, especially with regard to their choice of drug types and dosages for antibiotics, fluid therapy and choice of vasoactive and inotropic drugs. Special emphasis was placed on first-line treatment, and all deviations from the national guidelines were noted. For antibiotics treatment we also assessed whether the suspected focus of infection was considered upon initiation.
No requests or assessments concerning matters associated with diagnostics and logistics were made in this study
Results
We received responses from 43 of the 48 hospitals (89.6 %). The type and frequency of local guidelines were distributed as follows (Figure 1): 26 hospitals (six regional hospitals, 13 large emergency hospitals, 7 emergency hospitals) had their own local guidelines. Ten hospitals (two large emergency hospitals, eight emergency hospitals) had local guidelines from larger hospitals. In addition, eight of these reported to use the guidelines for antibiotics use in the specialist health service issued by the Directorate of Health.
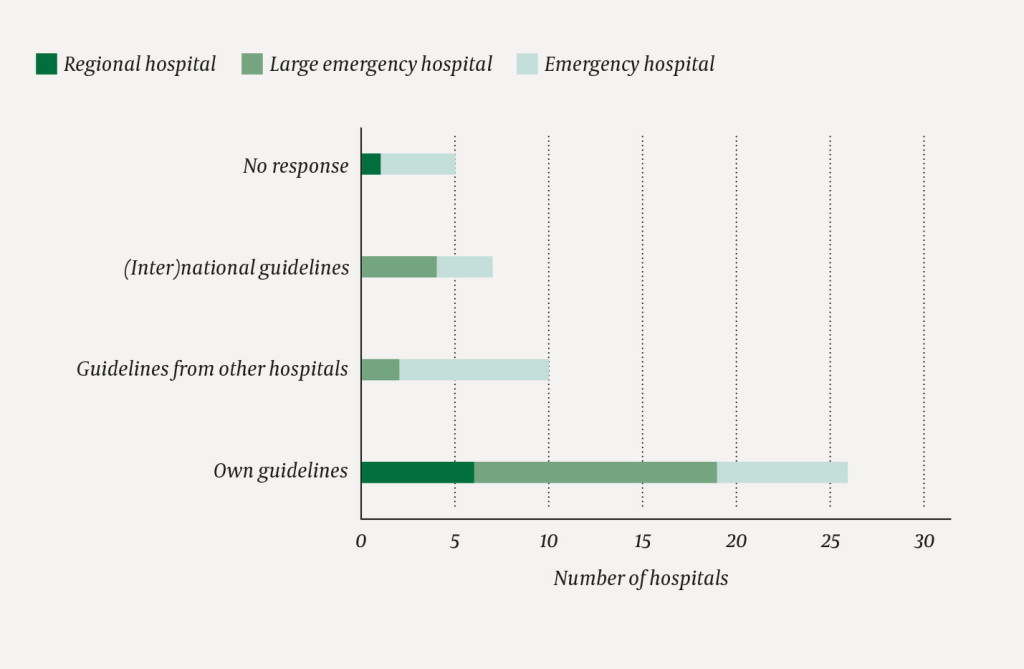
Four hospitals (three large emergency hospitals, one emergency hospital) had their own guidelines, but reported that at the time of the survey they preferred to use national/international guidelines, since their own were either outdated or undergoing revision. Three hospitals (one large emergency hospital, two emergency hospitals) had no local guidelines and used national and international guidelines directly.
The local guidelines were largely consistent with the national ones, but were less detailed. There were also some minor differences between them. For initiation of antibiotics treatment, the guidelines used by three hospitals tended to favour the use of more broad-spectrum drugs. Admittedly, two of these were unspecific, but still recommended 'broad-spectrum antibiotics within one hour after the diagnosis has been made in the intensive care department, ward or accident and emergency department'. The third set of guidelines recommended piperacillin/tazobactam or meropenem as the first choice. The remaining guidelines were consistent with the national ones, which recommend high-dose penicillin in combination with gentamicin.
All the guidelines pointed to concern for the assumed focus of infection or the suspected microbe, but were not as elaborate as the national guidelines. Only a single hospital stated that aminoglycosides were contraindicated in all patients with sepsis and septic shock.
All hospitals had crystalloids as their first choice for fluid therapy, while only 20 referred to colloids as an alternative. The national guidelines list albumin as an option. Three large emergency hospitals referred to hydroxyethyl starch as an alternative, which is clearly advised against in the national guidelines.
There were also some differences concerning the indication for starting fluid therapy, infusion rate and total fluid volume. According to the national guidelines, all sepsis patients should be provided with 30 ml/kg fluid within the first 60–120 minutes, depending on the clinical response. If there is a response (normalisation of blood pressure, reduced heart rate and increased urine production), fluid therapy should continue to the extent necessary to maintain stable circulation.
In intensive care departments, it is recommended to monitor the subsequent fluid therapy with objective measures of cardiac minute volume, pulmonary artery wedge pressure or intrathoracic blood volume. Most local guidelines recommended an initial fluid bolus of 20–35 mg/kg over the first 30–60 minutes. In case of persistent hypotension, initiation of vasoactive drugs was recommended, in combination with continued fluid therapy, with the aim of achieving a central venous pressure equal to 8–12 mmHg. Seven local guidelines noted that fluid volumes up to 10–12 litres are often required during the first 24 hours. Only five guidelines underscored that this treatment should continue only for as long as it produces a clinical response.
For the use of noradrenaline as a vasoactive drug, eleven hospitals had alternatives that are not mentioned in the national guidelines: adrenaline (11 hospitals), levosimendan (four hospitals) and phenylephrine (two hospitals). The national guidelines recommend noradrenaline as the first choice and dopamine and dobutamine as alternatives if an inotropic effect is also needed.
Vasopressin is recommended in patients with refractory shock. Altogether 23 hospitals lacked alternatives that are described in the national guidelines, eight of which did not refer to vasopressin. The greatest range in the use of vasoactive drugs was associated with the use of dopamine. One hospital gave dopamine as its first choice, while 20 hospitals had noradrenaline as their first choice and dopamine as an alternative. Four of these underscored that dopamine should only be used for selected patients. Ten guidelines made no reference to dopamine, while three explicitly stated that this drug should be avoided.
The national guidelines mention glucocorticoids as an alternative if hypotension persists despite treatment with fluids and vasopressors. Of the local guidelines, 28 were consistent with this, while there was no such reference in the remaining ones.
Discussion
Most Norwegian hospitals that have an emergency internal medicine department had local guidelines for treatment of sepsis, either prepared locally or by a larger hospital. All regional hospitals that responded had guidelines they had prepared themselves. Guidelines prepared by other hospitals were primarily used by smaller emergency hospitals.
The local guidelines were generally consistent with the national ones, although at a varying level of detail. Some disagreements and differences were nevertheless observed with regard to the use of aminoglycosides, fluid therapy and the use of vasopressor/inotropic drugs.
Diagnostics
The objective of this study was to examine guidelines for treatment of sepsis. In a number of the hospitals, however, the guidelines for diagnostics and assessment of possible sepsis had been integrated into the guidelines for treatment. The guidelines, both national and local, also included important and appropriate directions regarding logistics, monitoring and specific organ-supporting treatment.
The national guidelines state that the diagnosis of sepsis shall be made if the patient has a clinical infection and organ dysfunction, with recommendations to use the SOFA rating in the examination of organ dysfunction (6). Biochemical tests on blood samples should also be performed (11). Microbiological diagnostics is important, and material should preferably be collected before the start of antimicrobial treatment (unless this will entail a major delay, frequently defined as more than 30–45 minutes). It is recommended to collect two sets of blood cultures, urine for dipstick analysis, microscopy and cultivation, expectorate for microscopy and cultivation, as well as samples from the suspected focus, depending on clinical symptoms and findings (12).
Altogether 29 of the local guidelines were fully consistent with the national ones regarding microbiological diagnostics. Four hospitals had guidelines with minor shortcomings, while ten hospitals made no reference to examinations in their guidelines. This may be due to the fact that some hospitals had split their diagnostic guidelines into a separate document, for which we made no systematic enquiry.
Only six hospitals used the SOFA rating in their definition of sepsis (Box 1). The remaining ones used the old SIRS (systemic inflammatory response syndrome) criteria, which can be explained by the fact that our survey was undertaken at a time when the present definition of sepsis was in the process of being introduced.
Antibiotics
There was a high degree of consistency between national and local guidelines in terms of their choice of antibiotics, although three hospitals tended to use broad-spectrum drugs more frequently. All guidelines noted concerns for the assumed focus of infection and the microbe in question.
The use of aminoglycosides in cases of sepsis is fraught with some controversy. With one exception, the local guidelines were highly consistent with the national ones on this point. Aminoglycosides (and benzylpenicillin) were recommended as the standard regime, although with some advice to consider adjustment of the dosage for patients with reduced renal function. Only the National Hospital recommended refraining from the use of aminoglycosides in all patients with sepsis and septic shock, with reference to the heightened risk of renal failure.
The specialists at the National Hospital argue that aminoglycosides should be avoided in sepsis patients who are generally haemodynamically and/or respiratorily unstable and/or have reduced diuresis (13). This is substantiated by a systematic review by the Cochrane Library (14), later updated (15), showing that the use of aminoglycosides has no significantly better effect than other antibacterial therapies, while increasing the risk of nephrotoxicity.
The opponents of this view in the specialist community refer to other studies and argue that aminoglycosides have a positive effect on survival (16) and that the incidence of acute renal failure in fact can be reduced by administering initial treatment with at least one dose of aminoglycosides to patients with sepsis or septic shock (17). They also point out that the possible renal affection tends to be reversible, and that only multiple-dose regimes increase the risk of nephrotoxicity.
The disagreement over this issue may be regarded in light of the fact that different hospitals have different patient populations and thus may have differing approaches to the first choice of antibiotics. For example, the National Hospital is likely to have many immunosuppressed patients as well as others who need prolonged ventilator treatment, while other hospitals more often receive patients with a smaller previous disease burden.
Fluid therapy
Over the last ten years a change has occurred in intensive care with regard to fluid therapy for sepsis patients. Rather than using this quite liberally as was previously the case, current practice is more restrictive, since it has been shown that excessive fluid therapy may cause increased mortality (18).
A review article from 2016 (19) notes that most likely, less than 40 % of all hypotensive patients with sepsis or septic shock are 'fluid responders' (i.e. patients in whom the stroke volume increases by 10–15 % after a fluid bolus of 250–500 ml or after a 'passive leg raising manoeuvre' PLR), and that the haemodynamic effects of fluid therapy, including in fluid responders, are small, transient and most likely insignificant.
Excessive fluid therapy will be likely to have a negative effect in terms of both mortality and morbidity, for a variety of reasons. A rapid increase in the cardiac fill pressure may cause diastolic heart failure. Furthermore, one can observe destruction of the endothelial glycocalyx with arterial vasodilation and further increased vascular permeability and tissue oedema, in addition to the already increased vascular permeability caused by sepsis. Oedema in vital organs increases the general risk of organ failure.
Recent research recommends that only fluid responders be given fluid therapy, and that the responsiveness should be measured before each bolus (19). Initial fluid therapy should administer boluses no larger than 500 ml up to a maximum of 20 ml/kg. If the initial fluid therapy fails to produce an effect (mean arterial pressure < 65 mmHg), initiating treatment with noradrenaline is recommended (19).
Neither the national, nor the international guidelines recommend that fluid responsiveness be assessed prior to the start of fluid therapy. The recommended fluid volume was also larger than what recent research indicates as being most favourable (19).
The use of central venous pressure as an indicator of fluid balance is hotly debated, nationally as well as internationally. The literature notes that statistical indicators, such as central venous pressure, are of limited value, and that dynamic indicators, such as change in stroke volume when the patient's leg is raised (PLR)/fluid bolus, have proven better for the assessment of fluid responsiveness (5). Echocardiography can be used to determine the kind of haemodynamic disruption present and the kind of treatment indicated, as well as to monitor the response to treatment (20). As yet, this is not widely described in local, national and international guidelines.
The national guidelines recommend crystalloids, normally Ringer acetate, for fluid therapy. Albumin may be considered. Hydroxyethyl starch is contraindicated because of increased risk of renal failure and death. The local guidelines were consistent with this, but three large emergency hospitals listed hydroxyethyl starch as an alternative. This is clearly advised against in both national and international guidelines, as well as in large-scale international studies (21).
Vasoactive and inotropic drugs
The national guidelines recommend vasoactive drugs if circulation failure persists despite adequate fluid therapy. Noradrenaline is noted as the first choice, with dopamine and dobutamine as alternatives if an inotropic effect is also called for. It is underscored that compared to noradrenaline, dopamine is associated with more complications and increased mortality. Vasopressin is mentioned as an alternative for patients with refractory shock.
In the local guidelines there was a certain variation in terms of the drugs that were listed. The recommendations differed most with regard to the use of dopamine, which can be explained by the numerous international studies that urge the use of caution when using this drug.
A meta-analysis from 2012 (22) that included 11 studies (2 786 patients) compared the use of dopamine and noradrenaline in patients with septic shock. The randomised studies (1 408 patients) showed an increased mortality risk when dopamine was used (RR 1.12; 95 % CI 1.01–1.20, p = 0.035). Two of these studies also showed an increased tendency towards arrhythmia when this drug was used (RR 2.34; 95 % CI 1.46–3.77, p = 0.001).
Conclusion
There is reason to question the necessity for local guidelines in hospitals that do not have any special patient populations or special issues associated with resources and/or logistics.
Since good national and international guidelines are available, and preparation of local guidelines is resource-intensive and requires regular updating, it can be argued that such use of resources serves no good purpose.
- 3.
Tidlig oppdagelse av sepsis: Pasientsikkerhetsprogrammet I trygge hender.http://www.pasientsikkerhetsprogrammet.no/om-oss/innsatsomr%C3%A5der/tidlig-oppdagelse-og-behandling-av-sepsis (24.9.2017).
- 6.
Norsk legemiddelhåndbok. T1.10 Sepsis. http://legemiddelhandboka.no/Terapi/s%C3%B8ker/+%2Bsepsis/1780(18.3.2017).
- 7.
Helsedirektoratet. Nasjonal faglig retningslinje for bruk av antibiotika i sykehus. Sepsis. https://helsedirektoratet.no/retningslinjer/antibiotika-i-sykehus/seksjon?Tittel=sepsis-1116 (18.3.2017).
- 9.
Regjeringen. Oversikt over landets helseforetak. http://www.regjeringen.no/no/tema/helse-og-omsorg/sykehus/innsikt/oversikt-over-landets-helseforetak/id485362(18.3.2017).
- 10.
Nasjonal helse- og sykehusplan. https://www.regjeringen.no/no/tema/helse-og-omsorg/sykehus/nasjonal-helse-og-sykehusplan2/id2461509/(15.12.2017).
- 11.
Norsk legemiddelhåndbok. T1 Infeksjonssykdommer. Tabell 8 Aktuelle biokjemiske markører for inflammasjon og organdysfunksjon ved sepsis.http://legemiddelhandboka.no/Terapi/3203 (24.9.2017).
- 12.
Norsk legemiddelhåndbok. T1 Infeksjonssykdommer. Tabell 9 Aktuelle mikrobiologiske prøver ved sepsis.http://legemiddelhandboka.no/Terapi/3240 (24.9.2017).
- 14.
Paul M, Silbiger I, Grozinsky S et al. Beta lactam antibiotic monotherapy versus beta lactam-aminoglycoside antibiotic combination therapy for sepsis. Cochrane Database Syst Rev 2006; 1: CD003344. [PubMed]
- 15.
Paul M, Lador A, Grozinsky-Glasberg S et al. Beta lactam antibiotic monotherapy versus beta lactam-aminoglycoside antibiotic combination therapy for sepsis. Cochrane Database Syst Rev 2014; 1: CD003344. [PubMed]
- 21.
Mutter TC, Ruth CA, Dart AB. Hydroxyethyl starch (HES) versus other fluid therapies: effects on kidney function. Cochrane Database Syst Rev 2013; 7: CD007594. [PubMed]
Erik Refsland Kaspersen, Johan Ræder og Vegard Dahl til orde for at praksisen med å lage lokale retningslinjer for behandling av sepsis bør opphøre til fordel for at innsatsen heller rettes mot utvikling av nasjonale retningslinjer.
I en lederartikkel av professor Per Olav Vandvik får forfatterne støtte i dette synet (1). Samtidig påpeker Vandvik at eksisterende sepsisretningslinjer ved norske sykehus står mye tilbake for hva som er best tilgjengelige empiriske evidens. Han trekker fram tre eksempler som gir grunn til bekymring: væsketerapi, der flere sykehus fortsatt anbefaler bruk av stivelse; vasoaktive medikamenter, der dopamin fortsatt inngår i repertoiret, tross sterk evidens for at noradrenalin er å foretrekke; og aminoglykosider som tillegg til betalaktamer, tross flere gode metaanalyser der det ikke kan påvises gevinst, men heller økt fare for nefrotoksistet og terapisvikt.
Vandvik peker på retningslinjer som utgår fra den nordiske anestesiforeningen (SSAI) som eksempler på anbefalinger som oppfyller kravene til retningslinjer vi kan ha tiltro til. Disse er fritt tilgjengelige på foreningen sin hjemmeside (http://www.ssai.info/guidelines/).
Litteratur
1. Vandvik PO. Har vi troverdige retningslinjer for klinisk praksis i Norge? Tidsskr Nor Legeforen 2018 DOI: 10.4045/tidsskr.18.0102
Vi ser med bekymring på helsemyndighetenes manglende evne til å sørge for oppdaterte retningslinjer.
Erik Refsland Kaspersen og medarbeidere har publisert en studie om bruk av retningslinjer for behandling av sepsis i Norge (1). De konkluderer at man må stille spørsmål ved bruk av lokale retningslinjer. Hovedargumentet var at disse retningslinjene ikke ble oppdatert regelmessig. Videre ble det angitt at man hadde gode nasjonale (og internasjonale) retningslinjer som ble oppdatert. Hva våre nasjonale retningslinjer om antibiotika i sykehus angår er imidlertid dette feil.
I 2013 ble de første nasjonale retningslinjer for antibiotika bruk i sykehus publisert (2). Disse ble til etter et stort dugnadsarbeid blant infeksjonsmedisinere, mikrobiologer og en del andre spesialister med interesse for infeksjoner. Helsedirektoratet var oppdragsgiver, men arbeidet ble utført stort sett på fritiden av de involverte. Etter dette har vi med uro registrert økt forekomst av resistente mikrober, særlig globalt. Regjeringen har laget en ambisiøs plan om å redusere antibiotika bruk i Norge, nettopp for å unngå unødig bruk av resistensdrivende midler (3). For å nå disse målene har man blitt sterkt oppfordret til å bruke den norske veilederen, ettersom den tok mest hensyn til våre epidemiologiske forhold. Bruken av veilederen er økende, men i dagens versjon er det i flere kapitler direkte feil anbefalinger.
Disse retningslinjene er ikke oppdatert siden de ble utgitt i 2013. Det angis imidlertid på Helsedirektoratets nettsider at de sist ble revidert 8. januar 2018 (2). Ingen av de som var med på å skrive disse retningslinjene i 2013 har imidlertid deltatt i revisjonsarbeidet, og vi kan ikke se at det faglige innholdet på noe vis er revidert. Vi er mange i det infeksjonsmedisinske miljøet i Norge i dag som ser med bekymring på helsemyndighetenes manglende evne til å sørge for at det faglige innholdet er oppdatert.
Styret i Norsk forening for infeksjonsmedisin (NFIM) har i vinter etterlyst en plan og tidsramme for ny revisjon og ikke minst for hvordan fortløpende revisjon av retningslinjene skal ivaretas framover. Det skal nå være planlagt et forprosjekt hvor arbeidet skal være ferdig 1.12.18. Styret i foreningen ser fram til fortgang i dette viktige arbeidet. Vi anser oppdaterte retningslinjer som et viktig redskap i arbeidet mot resistens. Men dagens retningslinjer er dessverre ikke det.
For styret i Norsk forening for infeksjonsmedisin
Litteratur
1. Kaspersen ER, Ræder J, Dahl V. Retningslinjer for behandling av sepsis. Tidsskr Nor Legeforen 2018, doi: 10.4045/tidsskr.17.0493
2. Nasjonal faglig retningslinje for bruk av antibiotika i sykehus. Helsedirektoratet.no. 8.1.2018. https://helsedirektoratet.no/retningslinjer/antibiotika-i-sykehus (9.10.2018)
3. Nasjonal strategi mot Antibiotikaresistens 2015-2020. Regjeringen.no. 23.6.2015. https://www.regjeringen.no/no/dokumenter/nasjonal-strategi-mot-antibiotikaresistens-2015-2020/id2424598/ (9.10.2018)