It has long been known that some cancer changes exhibit great heterogeneity with respect to the progress from the start of cell changes until the disease manifests itself. Cell changes may also show total or partial regression (1, 2). The problem is that with current methods it is not always possible to distinguish between these different growth patterns at an early stage in the individual patient. When an increasing proportion of benign tumours are detected, disease-specific survival will increase without this representing an improved treatment outcome. The disease-specific mortality of the disease is a more valid measure of the treatment outcome.
Prostate cancer is the second most frequent cause of cancer death and the most commonly occurring form of cancer among men in Norway, accounting for 29 % of all cancer cases (3) – (5). Systematic examinations of prostate tissue from patients with other diseases and among men who died of trauma have shown that close to one in ten have detectable prostate cancer in their twenties already, and this rises to three of four men in their 70s (6, 7).
Since testing for the prostate cancer marker prostate-specific antigen (PSA) in the blood became available, its use has become widespread, and in the USA 30 million men are now tested annually (8). In Norway there has been broad consensus on warning against uncritical use of PSA testing for screening, and the health authorities communicated this agreement through an information campaign targeting general practitioners and urologists in 2001. The Norwegian Directorate of Health has maintained a restrictive view of the use of PSA testing in guidelines adopted in 2012 (9), although testing is allowed in cases of a family history or hereditary predisposition for cancer after the patient has been fully informed of the advantages and drawbacks.
Although a large European randomised study showed that PSA screening led to a 20 % decline in prostate cancer mortality, this involved a great deal of overdiagnosis (10). Other large studies failed to find a similar effect (11, 12). It is therefore generally accepted that a large number have to be treated, with a significant risk of side effects, in order to improve the outlook for a minority (5, 13). Studies of the amount of PSA testing have also been carried out earlier, in 1996 and 1999 (14), and Kvåle et al. found a significant increase in the number of PSA tests performed in the period 1996 – 2005 (15).
We accordingly wanted to look at available registry data and investigate the increase in the number of PSA tests in Norwegian counties from 1999 and up to 2011, and to relate this to cancer incidence per county and the rates of cancer-related prostate surgery. In addition, the situation in Sogn and Fjordane was subjected to special examination. This county has a low general cancer mortality compared with the national average, but at the same time the highest incidence of prostate cancer, adjusted for age (139/100 000 compared with the national average of 103/100 000) (4). We also investigated the attitudes and practice related to PSA testing prevailing among the primary doctors in the county.
Material and method
In collaboration with two organisations that administer external quality control for Norwegian laboratories (Norwegian Quality Improvement of non-Hospital Laboratory Activities and the Norwegian Clinical Chemistry External Quality Assurance), a national survey was conducted of all public and private laboratories in Norway regarding the number of analysed PSA tests. 47 responses were received from 49 laboratories (two small laboratories were lacking). The same questionnaire was used in an earlier study in 1999 (14). The laboratories were asked to state the distribution by county of the analyses they carried out. If the laboratories lacked records by county of the test requisitioners (25 of 47 laboratories), the analyses were registered on the county where most of the requisitioners were domiciled.
County prostate removal rates in the period 2008 – 2011 were obtained from the Norwegian Patient Register and include the following procedure codes: KEC00, KEC10, KEC20 and KEC01. Data for age-adjusted cancer incidence and mortality (world standard population) were obtained from the Cancer Registry of Norway and the Norwegian Cause of Death Registry.
In order to reveal the practice and attitudes of primary doctors in Sogn og Fjordane associated with PSA testing, a brief electronic questionnaire was sent out in collaboration with the ICT Department of the Western Norway Regional Health Authority and local practice consultants in the second half of June 2012 with the aid of the Surveyor tool. The form was distributed to 100 (of a total of 114) primary doctors whose email addresses we had available. Responses came from half of them after a reminder. 56 % of those who responded were specialists in general medicine.
Statistics
The data are presented descriptively. We used Pearson's correlation analysis to investigate relationships between the frequency of PSA testing and the incidence of prostate cancer and associated surgical procedures. We used one-tailed testing because we have few observations (19 counties) and no good a priori documentation to indicate that there may be inverse correlation between test frequency and cancer incidence. The analytical tool used was SPSS 20.0.
Ethics
Personally identifiable data were not used in the study. The study was approved by the local Data Protection Officer.
Results
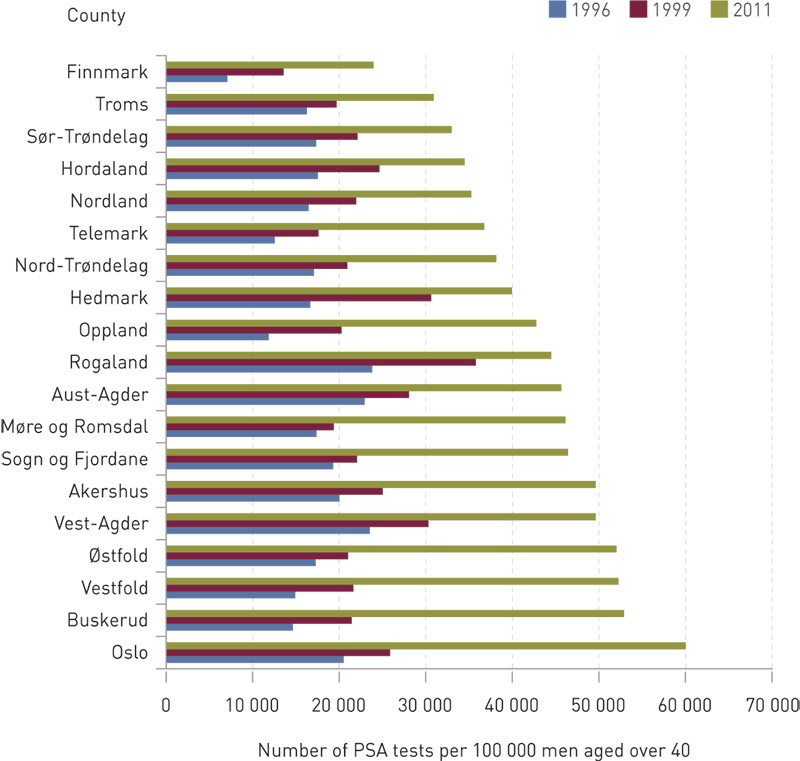
Figure 1 shows the results of the national study in 2011 compared with the studies in 1996 and 1999. A total of 524 959 PSA analyses were carried out in 2011, and the bulk of them, 87 %, were ordered by the primary health service (variation across counties 80 – 94 %). The male population aged over 40 was about 1.17 million that same year. The bulk of these PSA tests were probably used for screening/diagnostic purposes. The increase in the period 1999 – 2011 was 120 %, and was largest for Oslo. The four northernmost counties and Hordaland County occupied the lower part of the scale, and Finnmark County was at the bottom. At the opposite end were the counties under the South-Eastern Norway Regional Health Authority, with Oslo at the top. Sogn og Fjordane occupied seventh place.
The correlation between the number of PSA tests in 2011 and the incidence of prostate cancer in the period 2006 – 10 for all 19 counties was 0.41 (Pearson) (p = 0.41, one-tailed test). For the 13 counties with a test rate of < 50 000/100 000, this correlation was 0.83 (p > 0.001).
The corresponding Pearson correlation between incidence rates in the period 2006 – 2010 and average prostate surgery rates (2008 – 11) was 0.66 (p = 0.001). Figure 2 illustrates that a rising number of PSA tests is accompanied by an increase in the incidence rates of both prostate cancer and prostate surgery.
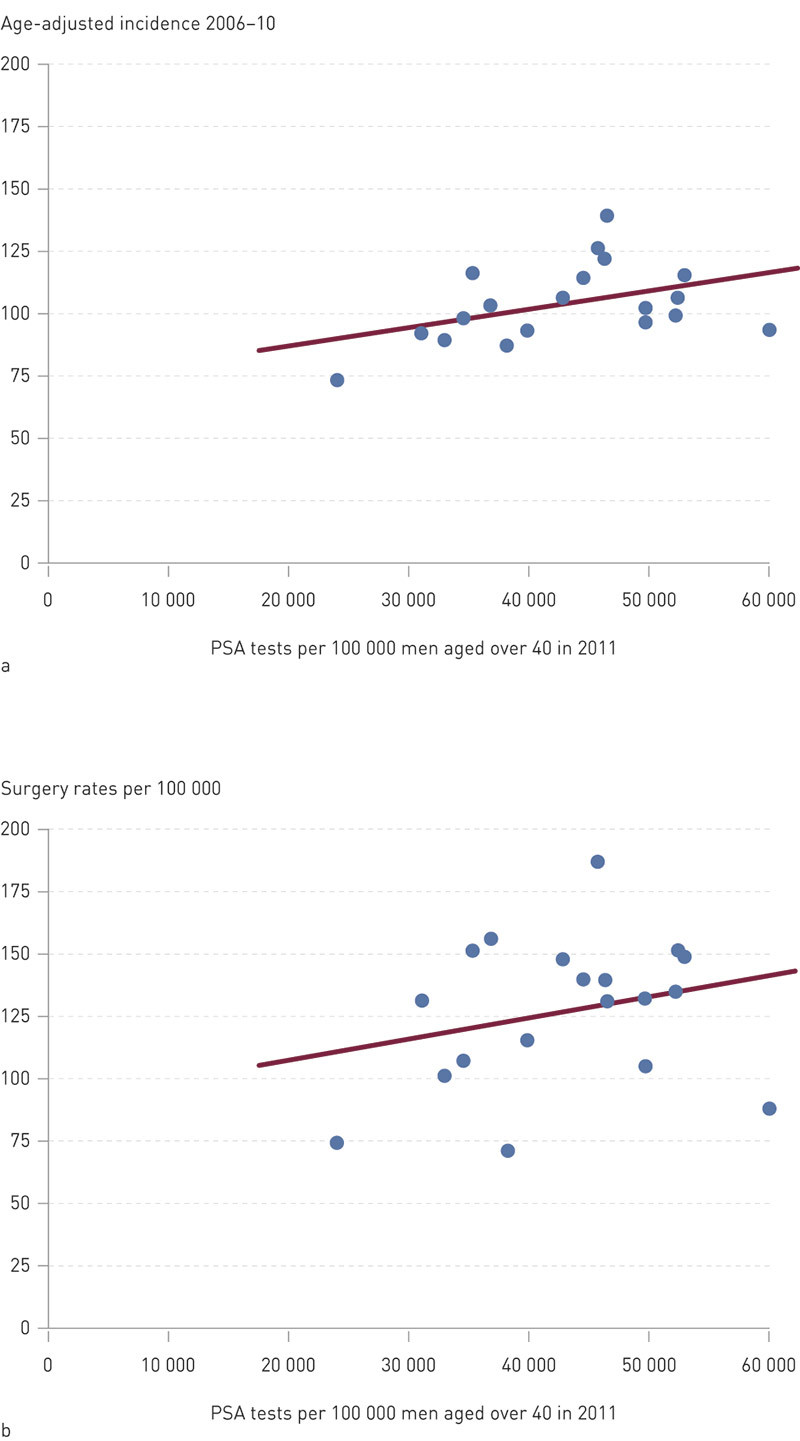
Figure 3 shows developments in the prostate cancer incidence rate, five-year survival rate and mortality over time in Sogn og Fjordane compared with the average for Norway. The curves for incidence rate and disease-specific survival have a pronounced kink followed by a stronger rise than the national average. The incidence of prostate cancer in Sogn og Fjordane almost tripled from the early 1990s to 2010 (50.9 and 139.5) compared with an approximate doubling (55.0 and 102.3) for the country as a whole. The difference between Finnmark, with the lowest incidence, and Sogn og Fjordane is 90 %. Prostate cancer mortality in Sogn og Fjordane has declined since the 1990s from 41.6 to 32.5 (national average from 40.5 to 33.7).
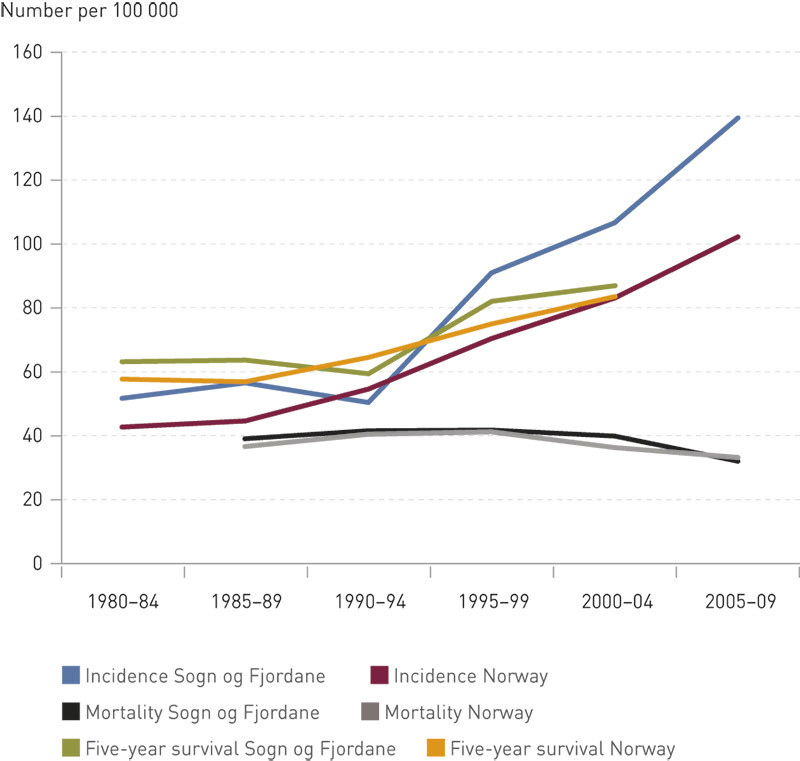
The proportion of those diagnosed who undergo surgical interventions is about a third, and this also applies to Sogn og Fjordane, with 34 % (national average 36 %). Five-year survival rose in the county from 59.5 % to 87 % (period 2000 – 2004), while the national average was 84 %.
The results of the questionnaire survey among primary doctors in Sogn og Fjordane concerning their practice and attitude to PSA testing are shown in Table 1. Only a minority of those asked allow the test to be included routinely in check-ups of men aged over 50. At the same time, it is clear that most of them carry out the test at the request of the patient, and that many also have difficulty in not referring patients for further examination if the PSA value is higher than a stipulated threshold value.
Table 1
Results of the questionnaire on practice and attitudes to PSA testing among primary doctors in Sogn og Fjordane
| Own practice/claim |
Number of valid responses |
Often/almost always (versus seldom/never) Per cent |
Completely/Partly agree (versus partly/completely disagree) Per cent |
| I order PSA tests: as part of a routine medical examination of men aged over 50: |
49 |
22 |
|
| if the patient requests it |
50 |
70 |
|
| if the patient has problems passing water |
50 |
94 |
|
| in the event of abnormal findings on exploration |
50 |
100 |
|
| as part of monitoring of prostate cancer |
49 |
99 |
|
| PSA testing should always be carried out if the patient requests it |
50 |
32 |
|
| Patients normally follow my advice on PSA testing or not |
50 |
78 |
|
| The patient should take a PSA test if a number of close relatives had prostate cancer detected |
50 |
82 |
|
| All patients with a positive biopsy test for cancer should have further treatment (surgery, radiotherapy, medical treatment etc.) |
49 |
30 |
|
| The side effects and complications of cancer treatment are outweighed by the benefit. |
50 |
26 |
|
| It is difficult not to refer a patient with PSA levels higher than the age-adjusted reference value to a urologist |
49 |
72 |
|
| In screening situations, an information circular about PSA testing should always be distributed before a test is ordered |
49 |
91 |
Discussion
There are pronounced differences in the registered occurrence of prostate cancer in Norwegian counties, and there has been a considerable increase in the number of PSA tests carried out in recent years. We find a county-wise correlation between increased PSA testing and increased diagnosis of prostate cancer and surgical treatment of the condition, while mortality has only been moderately reduced since about 1995. Sogn og Fjordane, with the highest registered incidence of this type of cancer, exhibits a typical pattern – incidence that started rising rapidly at the time when the PSA test was introduced, in 1991. Survival shows the same rising pattern, while mortality continues to lie near the national average. This suggests overdiagnosis and overtreatment of the condition. The general practitioners in the county are aware of the authorities’ restrictive attitude to PSA screening, but often comply with their patients’ wish for a screening test nonetheless.
There are sources of error associated with the survey. Incidence and surgery rates date from the period before PSA registration in 2011. This may be one reason that we find a distinctly lower correlation with the number of PSA tests than Norderhaug et al. found in 1999 (Pearson’s r = 0.73) (14). We have not received data from all laboratories that perform the test, but this source of error is minimal, since it is a matter of two small laboratories out of a total of 49. In some cases the laboratories had difficulty in identifying the county to which some of the requisitioners belonged, and placed them in the county from which the majority of requisitioners came. This makes the analyses of the correlation between the number of PSA tests and the incidence of cancer and treatment rates somewhat uncertain.
PSA testing is used both for assessing possible prostate cancer and for monitoring patients known to have the disease. In all counties, most analyses were carried out in response to a requisition from a general practitioner, consistent with the bulk of the testing having been performed as screening and early diagnosis of cancer disease. A very large number of analyses were carried out in Oslo, but 84 % of these had been ordered by general practitioners, as in other counties. The Oslo laboratories were also able to assign the requisitioners to the counties they belonged to. There are therefore no definite indications that the high test rate in Oslo is due to referral and treatment of patients from other counties. There is a large private laboratory in Norway (the Fürst Medical Laboratory) which accepts samples from general practitioners all over Norway. This laboratory was able to account for the counties to which its requisitioners belonged.
The percentage of primary doctors from Sogn og Fjordane who responded was low (only 50 %), whereas the gender distribution and number of specialists in general medicine indicate that the respondents were representative. Experience indicates that a higher percentage of responses to questionnaires regarding the use of laboratory tests among general practitioners cannot be expected (16). However, it is possible that those who did not answer may have attitudes and practice that differ from those who responded.
Finnmark has the least PSA testing and the lowest incidence of cancer; Sogn og Fjordane has the highest incidence and also a high test rate. Oslo, however, distinguishes itself by a very large number of PSA tests that nonetheless do not take the county to the top of the cancer statistics. This may indicate that mechanisms other than PSA testing also help to determine the registered incidence. One possibility may be steadily increasing testing of «the worried well», and that this population will make up a growing number, with increasing testing frequency. Another possibility is that the PSA test is carried out on indications other than cancer screening to a greater extent here than in other counties, so that many low-risk cases are tested (17). However, our study does not give answers to these questions.
The risk of overdiagnosis followed by overtreatment has been put on the agenda as a result of screening surveys (2). Health measures that have no value in themselves will moreover entail the same risk of error and complications as when treatment is indicated, and thus may constitute a double source of potential harm for patients (2, 3). In addition, health service resources are being used that could have benefited other patient groups (4). We support efforts to increase patient safety, but they should also encompass overdiagnosis and overtreatment (18). Different forms of treatment for prostate cancer are associated with significant side effects, in addition to concerns associated with having a malignant disease diagnosed (13, 19). The primary doctors’ responses also indicate that they are aware of the risk of side effects.
The last couple of years have seen a slight decline in the registered incidence of prostate cancer in Norway. According to our data, it is doubtful whether this is due to reduced use of PSA tests. An alternative explanation may be temporary «exhaustion» of the reservoir in the population (20).
Our study does not reveal lower mortality in Sogn og Fjordane, despite the increase in treatment. This is consistent with the recently published PIVOT study with 12 years of follow-up, which also did not find reduced mortality, either all-cause or specifically for prostate cancer with radical prostatectomy, as compared with observation (21, 22). However, the Scandinavian SPCG study showed reduced mortality and metastasis following early stage radical prostatectomy (22).
Consistent with the results of our study, a recently published study shows a large increase in the number of PSA tests in the Stockholm area. It also shows that the probability of retesting was high irrespective of the original PSA level (23). Other studies from Nordic countries show a similar tendency (15).
The results of our questionnaire survey show that PSA testing is seldom initiated by routines at the primary doctors’ offices, but that it is difficult to resist patients’ expectations and fail to start the procedures that lead to the diagnosis and treatment of located tumours. A survey conducted in 2002 also showed that general practitioners agreed and complied more than urologists with the health authorities’ guidelines (14).
Specialists often act as opinion leaders both in public debate and vis-à-vis patients. As recently as in 2009, a number of urological cancer researchers stated in Tidsskriftet that we must comply with patients’ wishes with respect to PSA testing (24). This may explain why general practitioners – despite knowing that a restrictive practice is recommended – often elect to order PSA tests on lack of indication, and why there is then a tendency for «wild» screening (and a subsequent risk of overtreatment). Carlsen et al. have shown that general practitioners are vulnerable in their function of gatekeeper, and align their activities towards meeting their «customers» expectations (25).
Our findings indicate that primary doctors need help from the health authorities and the opinion leaders, for example urologists, to curb the expectations of patients and their families. Health legislation in recent decades has increasingly been characterised by requirements of informed consent and sharing in decisions concerning assessment and treatment. Methods have been developed for holding dialogues on such decisions, and IT tools have also been developed to assist doctors and patients in revealing their preferences and the dilemmas associated with medical and surgical procedures (shared decision-making) (26).
Since we do not have reliable methods at present for making a prognosis for the individual patient with detected prostate cancer, the following three approaches could potentially be used to reduce overdiagnosis and overtreatment:
Greater compliance with national guidelines for PSA testing and balanced information to relevant patient groups (shared decision-making)
A generally more wait-and-see attitude to active treatment (watchful waiting), particularly in relation to the oldest group of patients (27) and use of higher PSA threshold values (28).
Data from the Cancer Registry of Norway have been used in this article/study. The interpretation and reporting of these data are the sole responsibility of the authors, and have not been subject to approval by the Cancer Registry. We should like to thank Johanne Gulbrandsen, special consultant at the Cancer Registry’s Data Delivery Unit, Inger Helen Berge of the Western Norway Regional Health Authority’s ICT unit, Jarle Øen at the Førde Hospital Trust for electronic surveying of primary doctors in Sogn og Fjordane and trainee consultant Normund Svoen for active participation in the formulation and conducting of the survey. We should also like to thank Karl Ove Hufthammer, statistician at the Department of Gynaecology and Obstetrics, Bergen Hospital Trust, for assistance with Fig. 2.
MAIN POINTS
The amount of PSA testing has increased substantially in recent years, but there are large differences across counties.
The incidence of prostate cancer has risen sharply and is correlated with the amount of PSA testing, while mortality due to the disease has changed relatively little over time.
Primary doctors appear to be having difficulty in not complying with their patients’ desire to be tested and find it difficult not to refer cases of elevated values to specialists.
Greater discretion is recommended with respect to both PSA testing and treatment.
- 3.
Nasjonalt handlingsprogram med retningslinjer for diagnostikk, behandling og oppfølging av prostatakreft. Oslo: Helsedirektoratet, 2009.
- 4.
Cancer in Norway 2010 – Cancer incidence, mortality, survival and prevalence in Norway. Oslo: Kreftregisteret, 2012.
- 5.
Welch HG. Overdiagnosed. Boston, MA: Beacon Press, 2011.
- 6.
Montie JE, Wood DP Jr, Pontes JE et al. Adenocarcinoma of the prostate in cystoprostatectomy specimens removed for bladder cancer. Cancer 1989; 63: 381 – 5. [PubMed]
- 7.
Sakr WA, Grignon DJ, Haas GP et al. Age and racial distribution of prostatic intraepithelial neoplasia. Eur Urol 1996; 30: 138 – 44. [PubMed]
- 8.
Albin RJ. «The Great Prostata Mistake». New York Times 9.3.2010.
- 9.
Nasjonalt handlingsprogram med retningslinjer for diagnostikk, behandling og oppfølging av prostatakreft. Oslo: Nasjonalt kunnskapssenter for helsetjenesten, 2012.
- 11.
Sandblom G, Varenhorst E, Rosell J et al. Randomised prostate cancer screening trial: 20 year follow-up. BMJ 2011; 342: d1539.
- 14.
Norderhaug IN, Wisløff T, Fosså S et al. Formidling av kunnskapsbasert informasjon om PSA-test og prostatakreft til leger. Tidsskr Nor Lægeforen 2004; 124: 2893 – 5. [PubMed]
- 24.
Fosså SD, Egey A, Haukaas SA et al. Feilaktig om hormonbehandling ved prostatakreft. Tidsskr Nor Legeforen 2009; 129: 429. [PubMed]
Breidablik og medarbeideres artikkel (1) er betimelig nå som professor Richard Albin, mannen som oppdaget PSA-testen, har skrevet bok og erklært at PSA ikke er en god indikator for cancer prostata, og i mange tilfeller ikke burde vært brukt for dette formål(2). Debatten er ikke ny. I 1978 kjørte magasinet Der Spiegel en serie artikler om kreft med tittelen «Ingen angst for kreft» signert professor Julius Hackenthal (3). Her tas troverdigheten av kreftdiagnostikk opp, herunder bruken av mikroskop ved analyse av celler fra antatt prostatakreft. Konklusjonen er nedslående: Selv med elektron-mikroskopi var ikke diagnostikken av kreftceller hos dyr og mennesker entydig. Hva angår prostata var opptil 20% av protstakjertlene infiserte, og muligens var det da infeksjonsendringer man mistolket som kreft.
Debatten om korrekt diagnostikk er absolutt betimelig og er parallell til debatten om Borreliose som verserer for tiden. I denne sammenheng har jeg som lege og forfatter drevet et toårig journalistisk arbeid som har resultert i en bok om flåttbårne sykdommer, der jeg har forsøkt å bringe klarhet i hvor troverdige diagnoser er (4). Man har her en helt parallell situasjon til prostatakreft: Usikre serologiske tester og konflikter med mikroskopi. Som tidligere fysikkstudent ser jeg også parallellsituasjonen til fysikk: I teknologiens gråsone der våre identifaktorer for gjenkjennelse ikke lenger er entydige, inntrer Werner Heizenbergs usikkerhetsprinsipp, som sier at den som undersøker med sin metode vil påvirke det man undersøker så mye at entydighet i konklusjon om erkjennelse forsvinner. I atomfysikk lever man med dette. Kan vi leger leve med det? Og hva med pasientene? Og når situasjonen er usikker, er det vår angst for å skulle overse og ikke behandle kreft på et tidlig stadium som får oss til å ”se” kreft der det ikke finnes med påfølgende feilbehandling? Er tiden moden for økt erkjennelsesteoretisk kunnskap og mer inngående selvransakelse (5)?
Litteratur
1. Breidablik HJ, Meland E, Aakre KM et al. PSA-måling og prostatakreft - overdiagnostisering og overbehandling? Tidsskr Nor Legeforen 2013; 133:1711 – 6.
2. Albin Richard: The great prostate Hoax; How Big Medicine Hijacked PSA test and caused a Public Health Disaster Palgrave McMillian, NewYork , 2014
3.Hackenthal J: Kein Angst für Krebs, Der Spiegel, 43/1978 s: 208-218
4. Øverbye BJ . Flåttbårne sykdommer, Veiviseren forlag, Fevik, 2014
5. Dilts Robert, Hallbom Tim ,Smith Suzi. Beliefs. Metamorphous Press, Portland, 1990