Contact with jellyfish can cause local skin discomfort and at worst serious reactions. It is estimated that more than 150 million people worldwide come into contact with jellyfish that cause symptoms (1) and that at least 200 die as a result each year (2).
Anything from moderate local reactions such as irritation and exanthema to serious, life-threatening conditions such as anaphylactic shock with circulatory and respiratory collapse may result from direct skin contact with jellyfish. The severity of reactions depends on the jellyfish species, the amount of skin that is exposed to the jellyfish’s tentacles, and whether the person is predisposed to serious reactions. Most people who come to hospital after contact with jellyfish are children and adolescents (3).
Jellyfish that are dangerous to humans vary in size from a couple of millimetres to species up to two metres in diameter and with tentacles that may be tens of metres long. Common to all of these (and other) cnidarians are tentacles with stinging cells (cnidocytes) that are activated by chemical substances and/or touch. When the cnidocyte is activated, a nematocyst is fired that transfers toxins into the prey (4). It is reasonable to suppose that if a sunscreen cream containing jellyfish sting inhibitor blocks cnidocytes and the transfer of toxin from one type of cnidocyte, there are good chances that it will also provide protection against cnidocytes and toxins from other related jellyfish with the same type of cnidocytes.
Both the lion’s mane jellyfish (Cyanea capillata) and the blue jellyfish (Cyanea lamarckii) are common along the coast of Norway at certain times of the year. They are therefore also the species that most often cause discomfort, although as a rule the discomfort is transient and moderate in severity. In tropical and subtropical waters there is a risk of encountering jellyfish that occur in large colonies, including the Portuguese man-of-war (in the Mediterranean) and box jellyfish such as the sea wasp and Irukandji jellyfish (around Australia, Indonesia and Thailand). Both Portuguese man-of-war and most species of box jellyfish can cause injuries with life-threatening symptoms.
We have previously published articles in Tidsskriftet on the development of symptoms and findings relating to contact with jellyfish, the mechanisms behind the effects and the prevention and treatment of jellyfish stings (5, 6). Posting jellyfish warnings in jellyfish season at places where they are particularly prevalent is an important preventive measure. It is also possible to protect the skin with a wetsuit or special suit that prevents exposure of the skin to jellyfish. However, wetsuits and other suits providing full body cover (stinging suits) are used by very few bathers in Norway. A two-piece bathing suit (top and bottom) is not very suitable for protection against jellyfish tentacles, even if the stitch density satisfies the criteria for stinging suits, because jellyfish tentacles may become squeezed between suit and body and be mechanically activated. Reports have demonstrated that this mechanism has caused deaths in contacts with Irukandji jellyfish, which do not occur in Norwegian waters, however (7).
In recent years sunscreen lotions have been developed that inhibit stings from certain species of jellyfish (1, 8). However, few scientific studies have been performed of these, and none have tested the effectiveness of these protective creams against the blue and lion’s mane jellyfish. The lion’s mane jellyfish has several types of cnidocytes, including all those found in the blue jellyfish (9). This means that any inhibition achieved against lion’s mane tentacles can be expected to be equally effective against blue jellyfish tentacles.
The purpose of this trial was to compare the effect of a commercially available jellyfish sting-inhibiting sunscreen lotion with the effect of a conventional water-resistant sunscreen lotion and of no prophylaxis against discomfort caused by lion’s mane jellyfish.
Material and method
This is a randomised trial conducted in the period September 2008 to December 2009 at the Marine Biological Station in Drøbak, which is associated with the University of Oslo. On the basis of previous studies a magnitude calculation was performed which showed that at least 24 persons had to be included (magnitude 80 %, level of significance 5 %) to demonstrate a clinically relevant difference in pain registration on a VAS scale of 10 mm and assumed standard deviation of the difference of 7 mm. The trial has been registered at clinicaltrials.gov (registration number 2008-001519-39/NCT00693641) and has been approved by the regional ethics committee (registration number 6.2008.524).
Test subjects
A notice at the Marine Biological Station in Drøbak and announcements in the local media led to the recruitment of 38 volunteers (21 men). The median age was 26 (range 20 – 48). Exclusion criteria were an age of under 18, chronic illness, use of antihistamines, systemic or topical steroids or other allergy medicines, use of topical medical or cosmetic products on the forearms during the previous 48 hours, and pregnancy. Skin pigmentation was graded according to Fitzpatrick’s classification into types 1 – 4 (10).
Method
Lion’s mane jellyfish were collected from Drøbak Sound in the Oslo Fjord on the day before or the same day as testing. Collection took place cautiously with the aid of underpressure into a bucket so as not to activate the cnidocytes or harm the jellyfish in any way. The jellyfish were placed in large water tanks with automatic through-flow of saltwater.
A small round watch glass with a diameter of 3.5 cm was prepared containing 0.6 g of severed tentacles (wet) for each skin area to be tested. A standardised area of 16 × 5 cm (80 cm²) was marked on both forearms of all test persons.
Block randomisation then took place. The subjects received two different treatments, one on each arm, from the following three possibilities:
creaming with 0.16 g sunscreen lotion containing jellyfish sting inhibitor (SPF 15, AC-SunCare institute, Norway, based on US Patent 7081247, (SafeSea) from Nidaria Technology, Israel)
creaming with 0.16 g conventional sunscreen lotion (Nivea children’s sun lotion, SPF 15)
no treatment
The choice of prophylaxis was unknown to the person (first author) who was to record the reactions of the trial subjects, who were blinded with respect to use of sunscreen lotion with or without jellyfish sting inhibitor.
The skin on the forearms was exposed to the severed tentacles in the watch glass by placing the watch glass upside down on the forearm and fixing it with tape. Both forearms were tested simultaneously. The trial subjects were instructed to move and rotate their arms cautiously so that there was good contact between tentacles and skin. The watch glass and contents were then removed when the trial subject felt pain/discomfort, after a maximum period of four minutes.
The following were recorded:
time from exposure to tentacles to registration of pain/discomfort
skin manifestations after four minutes
forearm pain/discomfort after ten minutes, registered on a visual analogue scale (VAS) from 0 – 100.
Registrations of time until pain/discomfort developed were divided into three categories: early reaction (within 45 seconds), late reaction (45 seconds – 4 minutes) and no pain/discomfort by the end of four minutes, when the exposure was terminated.
Skin manifestations after four minutes were classified as no skin manifestations, slight colour changes, pronounced colour changes, structural changes or vesicles.
A doctor was present during the test and able to provide treatment in the event of serious allergic reactions.
Statistical analysis
Demographic data are presented as median (range) and number of persons (%). Descriptive comparisons of the time to pain/discomfort for the different types of treatment are presented using Kaplan-Meier curves. Since the observations were paired, no log rank tests were performed.
With three randomised prophylactic treatments and within-subject paired data, the effects of two treatments for each subject – one for each arm – were recorded. The data were analysed using generalised estimating equations (GEE) for ordinal outcomes, assuming interchangeable covariance between the arms of each person.
Paired comparisons were made between the three treatments. The reported p-values were subjected to Bonferroni adjustment. The records of pain/discomfort on the VAS scale were analysed using a linear mixed model with a random intercept to take account of the within-subject relationship. Statistical significance was defined as a p-value of < 0.05. The statistical analyses were performed using SPSS, version 18 (SPSS Inc., Chicago, IL, USA).
Results
Demographic data for the persons in the various groups are shown in Table 1. Of those who had received the jellyfish sting inhibitor, 13 of 25 persons did not register any pain/discomfort after 4 minutes’ exposure to tentacles, compared with two of 25 and two of 26 who had received prophylaxis with conventional sunscreen lotion (p = 0.32) and no prophylaxis (p < 0.001), respectively. Fig. 1 provides a visual representation of the time to pain/discomfort with and without prophylaxis.
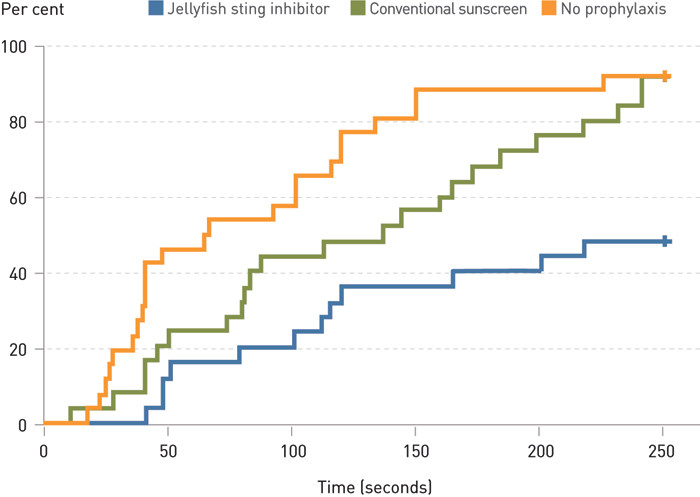
Table 1
Demographic data by treatment group expressed as median (range) or number (%)
|
|
Jellyfish sting inhibitor and conventional sunscreen (n = 12) |
Jellyfish sting inhibitor and no cream (n = 13) |
Conventional sunscreen and no cream (n = 13) |
| Age, median (range) |
34 (22 – 47) |
26 (20 – 48) |
23 (20 – 38) |
| Gender, no. of men (%) |
5 (42) |
8 (62) |
8 (62) |
| Skin type (Fitzpatrick), n (%)¹ |
|||
| Type 1 |
1 (9) |
0 (0) |
0 (0) |
| Type 2 |
1 (9) |
5 (38) |
5 (38) |
| Type 3 |
7 (64) |
7 (54) |
5 (38) |
| Type 4 |
2 (18) |
1 (8) |
3 (23) |
| [i] | |||
[i] ¹ Skin type information was lacking for one person
Table 2
Time between exposure to jellyfish tentacles and registration of pain/discomfort. Number of forearms in each time group. All p-values stated after Bonferroni correction
| No pain/discomfort¹ |
Pain/discomfort |
Total (n) |
||
| Early |
Late |
|||
| Jellyfish sting inhibitor² |
13 |
1 |
11 |
25 |
| Ordinary sunscreen³ |
2 |
5 |
18 |
25 |
| No prophylaxis |
2 |
11 |
13 |
26 |
| [i] | ||||
[i] ¹ These persons also did not register pain after termination of exposure (> 240 s)
² Jellyfish sting inhibitor compared with conventional sunscreen (p = 0.32) and no prophylaxis (p < 0.001)
³ Conventional sunscreen lotion compared with no prophylaxis (p = 0.55)
There were differences in visible clinical reactions between skin on which jellyfish sting inhibitor lotion had been used, on the one hand, and conventional sunscreen (p = 0.03) and no prophylaxis on the other (p = 0.006) (Table 3). No difference was found between conventional sunscreen and no prophylaxis. On average, subjects who had received jellyfish sting inhibitor lotion registered lower VAS scores for pain/discomfort after ten minutes than those with both conventional sunscreen (difference 106, 95 % CI 3.1 – 17.9) and no prophylaxis (difference 14.2; 95 % CI 6.9 – 21.5) (Table 4). No difference was found between conventional sunscreen and no prophylaxis (p = 0.65).
Table 3
Degree of skin inflammation four minutes after exposure to jellyfish tentacles. All p-values reported after Bonferroni adjustment
| Skin manifestations |
||||||
| No skin manifestation |
Slight colour change |
Pronounced colour change |
Structural change |
Vesicles |
Total (n) |
|
| Jellyfish sting inhibitor¹ |
19 |
4 |
2 |
0 |
0 |
25 |
| Conventional sunscreen³ |
14 |
5 |
5 |
1 |
0 |
25 |
| No prophylaxis |
14 |
5 |
7 |
0 |
0 |
26 |
| [i] | ||||||
[i] ¹ Jellyfish sting inhibitor compared with conventional sunscreen (p = 0.03) and no prophylaxis (p = 0.006)
² Conventional sunscreen lotion compared with no prophylaxis (p = 1.00)
No severe topical or systemic reactions were registered during or after testing.
Table 4
Registration of pain/discomfort on visual analogue scale (VAS) ten minutes after exposure to jellyfish tentacles. Average VAS score estimated from the linear mixed model
| n |
Average (95 % CI) |
|
| Jellyfish sting inhibitor¹ |
25 |
6.3 (1.6 – 11.1) |
| Conventional sunscreen² |
25 |
16.9 (12.1 – 21.7) |
| No protection |
26 |
20.5 (15.8 – 25.2) |
| [i] | ||
[i] ¹ Jellyfish sting inhibitor compared with conventional sunscreen (p = 0.003) and no prophylaxis (p < 0.001)
² Conventional sunscreen lotion compared with no prophylaxis (p = 0.65)
Discussion
In this trial, we tested the effectiveness of jellyfish sting inhibitor cream compared with conventional sunscreen lotion and no prophylaxis before exposure to jellyfish tentacles. We found that the use of jellyfish sting inhibitor reduced the number of subjects who suffered discomfort, and the number of objective findings. The discomfort was also less severe.
These results are consistent with the few published studies of the use of jellyfish sting inhibitor cream, although the studies have somewhat different methodology and other types of jellyfish were used. In a field trial from the Gulf of Mexico the protection provided by jellyfish sting inhibitor lotion (Safe Sea) compared with conventional sunscreen was tested on ten voluntary snorkellers (1). One of the two lotions was applied to the right and one to the left half of the body. The subjects snorkelled a total of 82 times in seawater for around 30 minutes. Two symptom-generating contacts with jellyfish were registered on the skin areas that had received jellyfish protection, compared with 11 on the areas to which conventional sunscreen had been applied (p = 0.02). In another trial with a method more like our trial, 23 subjects were tested with inhibitor lotion (Safe Sea) or conventional sunscreen lotion (Coppertone) on the forearm (8). All of the 12 trial subjects who were exposed to the jellyfish Chrysaora fuscescens developed erythema and experienced pain on the areas of the skin to which conventional sunscreen had been applied, while erythema was found and pain registered only in two trial subjects whose skin had been creamed with jellyfish sting inhibitor (p < 0.01). Twelve subjects were tested against the box jellyfish Chiropsalmus quadrumanus, and erythema and pain were registered in ten and nine, respectively, of these who had received conventional sunscreen, compared with one and three of those who had received jellyfish sting inhibitor (p < 0.01).
The advantage of our trial compared with previous studies is a higher number of trial subjects and that the jellyfish sting inhibitor cream was also tested against no prophylaxis. We also made greater efforts in the trial to measure the time before pain/discomfort occurred and to compare the effect of the jellyfish sting inhibitor cream with conventional sunscreen or no cream. The jellyfish that are most prevalent in Norwegian seawater were used. We therefore believe that this trial is appropriate for Northern European conditions.
The weakness of our model is that we investigated the effect of three different treatments, of which each subject only received two. However, we have taken this into account in our statistical analyses. The reason for our comparison of the jellyfish sting inhibitor cream with both conventional sunscreen and no treatment was that electron microscope studies have shown that conventional water-repellent sunscreen can have a protective effect against cnidocytes compared with no prophylaxis (11). We found greater differences between the jellyfish inhibitor lotion and no prophylaxis than between the inhibitor lotion and conventional sunscreen, but no statistically significant difference between conventional sunscreen and no prophylaxis. It is possible that by increasing the number of trial subjects we could have demonstrated such a difference, but it is uncertain whether it would have had any clinically significant relevance. It ought also to be noted that we had no negative control, and that the trial was only blinded with respect to prophylaxis with jellyfish inhibitor and conventional sunscreen. This blinding was not possible for subjects who received no prophylaxis, which could influence the registrations of the trial subjects.
As most sea-bathers have experienced, pain/discomfort commences virtually immediately after exposure to jellyfish tentacles, corresponding to the time when the cnidocytes fire the toxins into the skin. This normally occurs at the first touch. In our trial, we registered pain/discomfort at different times in the course of the four minutes the subjects were exposed to tentacles, even though many of those who did not receive prophylaxis registered pain/discomfort within 150 seconds. This may be because we tested the subjects with a relatively small quantity of jellyfish tentacles in a relatively small area. Some of the cnidocytes had also already been activated while being moved from the sea to the trial premises and thus were no longer potentially harmful. We chose a relatively long exposure time in order to be sure that some of the cnidocytes were activated against the trial subjects’ skin and thus could provoke pain/discomfort. We did not investigate the effect of an exposure time longer than four minutes, but we believe this time is satisfactory viewed in relation to expected exposure time in ordinary seawater. None of those who did not experience pain/discomfort during exposure developed pain/discomfort after the tentacles had been removed. This may indicate that the risk of developing symptoms in the time after exposure is limited.
The jellyfish sting inhibitor effect may be attributed to a number of factors (1). The cream is very hydrophobic, and therefore reduces the number of tentacles that come into contact with the skin. The cream contains glycosaminoglycans similar to those in the jellyfish, and receptor antagonists that trigger the cnidocyte receptors on activation. The cream also contains substances that block calcium and magnesium exchange in the cnidocytes’ cnidocysts and therefore reduce the osmotic pressure for transferring cnidocyte toxin. Although this can reduce the quantity of toxins a subject is exposed to, the cream will not neutralise the effect of any toxin that enters the skin.
The treatment strategy described earlier in Tidsskriftet (5, 6) therefore remains relevant for those persons who develop topical or systemic symptoms. After exposure, tentacles should be removed carefully with tweezers and/or rinsed off with seawater. A medium for deactivating cnidocytes should not be used unless one is sure of the mechanism by which the medium affects that specific jellyfish. At worst it could exacerbate the situation, for example the use of vinegar against the lion’s mane jellyfish. After all tentacles have been removed, topical reactions can be dampened with lidocaine gel or corticosteroid cream. Peroral medicines such as paracetamol, non-steroid anti-inflammatories, antihistamines and steroids may also be considered. In the case of systemic reactions the patient should be observed or hospitalised.
Tabell
| Main points |
|
- 2.
Fenner PJ, Williamson JA. Worldwide deaths and severe envenomation from jellyfish stings. Med J Aust 1996; 165: 658 – 61. [PubMed]
- 4.
Kass-Simon G, Scappaticci AA Jr. The behavioral and developmental physiology of nematocysts. Can J Zool 2002; 80: 1772 – 94. [CrossRef]
- 5.
Tønseth KA. Helseskade ved kontakt med brennmaneter. Tidsskr Nor Lægeforen 2007; 127: 1777-8. [PubMed]
- 6.
Tønseth KA, Andersen TS, Karlsen HE. Brennmanetskader. Tidsskr Nor Legeforen 2009; 129: 1350.
- 7.
Gershwin L, Dabinett K. Comparison of eight types of protective clothing against Irukandji jellyfish stings. J Coast Res 2009; 25: 117 – 30. [CrossRef]
- 8.
Kimball AB, Arambula KZ, Stauffer AR et al. Efficacy of a jellyfish sting inhibitor in preventing jellyfish stings in normal volunteers. Wilderness Environ Med 2004; 15: 102 – 8. [PubMed]
- 9.
Östman C, Hydman J. Nematocyst analysis of Cyanea capillata and Cyanea lamarckii. Sci Mar 1997; 61: 313 – 44.
- 11.
Heeger T, Möller H, Mrowietz U. Protection of human skin against jellyfish (Cyanea capillata) stings. Mar Biol 1992; 113: 669 – 78. [CrossRef]
Jeg ser at en av forfatterne er styreleder og daglig leder for firmaet som markedsfører den substansen som (naturlig nok) kommer best ut i testen.
Vedommende har oppgitt interessekonflikten.
Men det er likevel påfallende at en slik "reklame" publiseres i tidsskriftet idet badesesongen begynner.
Hva har man i tidsskriftets redaksjon tenkt om dette?
Takk til Olav Sandstad for en kommentar om den kanskje største utfordringen for seriøse medisinske tidsskrifter for tiden: Kommersielle interessekonflikter. Det er knapt noen originalartikler om behandlingsmetoder som publiseres - her hjemme eller i andre land - som ikke har minst én forfatter med potensielle interessekonflikter. Interessekonflikten kan være kommersiell - som her - eller det kan dreie seg om andre typer interessekonflikter som f.eks. at ett fagmljø ønsker å sentralisere en metode til sitt sykehus.
Å la være å publisere artikler der det er potensielle interessekonflikter er ikke hensiktsmessig, men det helt selvsagt avgjørende er at slike interessekonflikter synliggjøres både for tidsskriftene, fagvurdererne og selvsagt leserne - slik det er gjort her.
Sandstad spør hva vi i redaksjonen har tenkt om dette når vi har vurdert artikkelen. Uten å gå altfor mye i detalj, kan jeg si at vi alltid er ekstra på vakt når det kan være potensielle interessekonflikter, men forøvrig gjennomgår artiklene nøyaktig samme grundige granskning som alle andre artikler: altså ekstern fagvurdering, egen redaksjonell vurdering og evt supplerende vurdering av statistiker.
Vi legger også vekt på at det her er tre medforfattere uten noen interessekonflikter som går god for metodene og resultatene.
Når vi da etter grundig faglig og redaksjonell vurdering, kom frem til at denne artikkelens konklusjoner var holdbare, syntes vi det var naturlig å publisere artikkelen nettopp i sommernummeret.