A man in his twenties with fever and severe abdominal pain below the right costal margin
A man in his twenties was admitted to the Department of Surgery after a six-day history of illness with fever and pain that came in waves below the right costal margin. He had known ulcerative colitis, and prednisolone had been gradually reduced and discontinued three weeks previously. The course of his illness was dramatic, and the final diagnosis unexpected.
The patient had had low-grade atopic eczema as a child, and six months previously had been diagnosed as having ulcerative total colitis. Treatment had then been started with mesalazine by mouth followed by budesonide by mouth. As this treatment regime had not led to satisfactory alleviation of symptoms, he had been given prednisolone 40 mg x 1 by mouth, with a schedule for gradual dose reduction and discontinuation over a four-month period. At the time of admission he was not taking regular medication. Prednisolone had been discontinued three weeks previously.
In reception the patient indicated pain below the right costal arch, but was otherwise free of pain. Blood pressure was 147/80 mm Hg, pulse 93/min, temperature 39oC, respiratory rate 20 and peripheral oxygen saturation 99 %.
Apart from tenderness to palpation of the epigastrium and below the right costal arch, the findings of organ palpation were normal. Blood tests on admission showed leukocytes 10.3 · 109/L (reference range 3.9–9.5), CRP 44 mg/L (0–4), alanine aminotransferase (ALT) 80 U/L (10–70), aspartate aminotransferase (AST) 48 U/L (15–45), lactate dehydrogenase (LD) 216 U/L (105–202) and albumin 36 g/L (39–50). Values for haemoglobin, creatinine, alkaline phosphatase, total bilirubin, amylase, lipase and gammaglutamyl transferase (GT) were normal. INR was not measured. Urine dipsticks revealed normal findings and the FOB test was negative.
Hospitalisation of ulcerative colitis patients owing to acute abdominal pain is not unusual. This man's ulcerative colitis was believed to be in remission, and in light of his fever and tenderness to palpation below the right costal arch, acute cholecystitis was considered as a possible cause.
A tentative diagnosis of cholecystitis was made, and intravenous treatment with piperacillin-tazobactam 4 g x 3 intravenously was started while waiting for liver and bile duct ultrasound. Ultrasound showed a reactionless, slightly distended gall bladder, otherwise normal findings. Over the following two days the patient felt better, but had intermittent fever up to 38.6oC. His CRP rose to 60 mg/L (0–4) and leukocyte count to 14.4 · 109/L (3.9–9.5), despite broad antibiotics cover. A CT abdomen was performed on day 3 and revealed an unremarkable gastrointestinal tract. His liver had uneven density, interpreted as an expression of fat infiltration. Tests were ordered for hepatitis A, B and C, HIV, cytomegalovirus (CMV), Epstein-Barr virus (EBV) as well as antinuclear antibodies (ANA) and antineutrophil cytoplasmic antibodies (ANCA).
As the patient had persistent fever and showed no biochemical response to antibiotics, while the imaging provided no grounds for suspecting a condition requiring surgery, the patient was transferred to the Department of Medicine. The clinical picture was now one of liver involvement and fever of unknown origin. The gastroenterologist suspected hepatitis or some other viral infection, possibly an autoimmune disease such as primary sclerosing cholangitis or autoimmune hepatitis.
On transfer to the Department of Medicine (day 3) he had a temperature of 39.5oC, was lethargic and had abdominal pain. He was still slightly tender to palpation below the right costal arch, otherwise normal organ status.
On day 4 after admission, the patient reported pain distally in both lower extremities. Flat, non-blanching exanthema was seen. The rash had not been mentioned in the admission or transfer notes.
Because of the exanthema, the patient was referred the same day to a rheumatologist to determine whether there was an underlying immunological disease. The patient reported to the rheumatologist pain in his legs and problems walking. It now emerged that the rash on the lower legs had been present for 5–6 weeks. In addition he had a slight cough without sputum. He had also had a slight headache and stiff neck in recent weeks, but this was abating.
The rheumatologist conferred with the dermatologist about the rash and considered vasculitis or other rheumatic disease to be unlikely. The recommendations were to supplement with complement factor and cryoglobulin testing, differential counting of leukocytes, as well as urine dipstick testing with microscopy and urine protein/creatinine ratio determination. At about this time negative test results arrived for ANA, myeloperoxidase ANCA, proteinase 3-ANCA and cryoglobulins, and complement factors were normal.
On day 6 the patient had persistent fever, with stable inflammation parameters. His platelet count was observed to have fallen from 150 · 109/L (145–390) on admission to 63 · 109/L (145–390). Piperacillin-tazobactam was now discontinued, as bacterial infection appeared improbable. Hepatitis A, B and C, HIV, EBV and CMV serology were negative. A magnetic resonance cholangiopancreatogram (MRCP) was now performed, which showed large areas of the liver with diffuse oedema, dilated intrahepatic bile ducts with varying calibre and enlarged lymph nodes in the liver hilum. The image raised suspicion of primary sclerosing cholangitis.
The cholestasis parameters alkaline phosphatase, total bilirubin and gammaglutamyl transferase (GT) were still normal and weighed against cholangitis and clinically significant stenosis in intra/extrahepatic bile ducts. Because IgG was normal, autoimmune hepatitis was also considered unlikely. A clinically quiescent primary sclerosing cholangitis could not be excluded, or possibly an overlap between primary sclerosing cholangitis and autoimmune hepatitis. At this point the patient was ambulant and in relatively good general condition and was allowed to take home leave.
The following day (day 7) he returned because of new-onset stabbing thoracic pains and laboured breathing, cough and fever. During the examination he sat bent over and hyperventilated. Vital signs were pulse 114 beats/min, blood pressure 128/83 mm Hg, respiratory rate 32, SpO2 96 % and temperature 39.0 °C. There were normal auscultation findings over the heart and lungs, and the rash on his lower extremities was still present.
Blood gases showed respiratory alkalosis (pH 7.49 (7.35–7.45), pCO2 3.9 kPa (4.7–6.7), pO2 7.9 kPa (10.0–14.0), normal lactate). His leukocyte count had risen to 25.3 · 109/L (3.9–9.5). Differential counting, which was ordered now retrospectively, showed pronounced eosinophilia with eosinophilic granulocytes 14.5 · 109/L (0.0–0.4). The figure for monocytes was 1.4 · 109/L (0.3–0.9), lymphocytes 2.2 · 109/L (1.3–3.4), neutrophil granulocytes 6.6 · 109/L (1.5–5.7) and platelets 37 · 109/L (145–390). CRP had risen to 118 mg/L (0–4), LD to 315 U/L (105–205) and D-dimer was > 4.0 mg/L (0–4). His fibrinogen level was elevated, 4.9 g/L (2.0–4.0), while activated partial thromboplastin time (APTT), antithrombin and INR were normal.
His liver tests were improving. Chest X-ray showed an infiltrate on the left side. CT thorax was performed on suspicion of pulmonary embolism and showed patchy opacities in both lungs with an interstitial character, but no signs of pulmonary embolism.
The clinical exacerbation with eosinophilia, thrombocytopenia and indeterminate infiltration of the liver and lungs raised suspicion of a serious haematological disease. The patient was now moved to the Department of Haematology. The laboratory results revealed no signs of consumption coagulopathy as a cause of the thrombocytopenia.
The blood smear was consistent with the mechanical differential count: 55 % eosinophilic granulocytes and no signs of microangiopathy. A bone marrow biopsy was taken. At the same time a bone marrow smear was made (Figure 1) which showed slightly hypercellular fragments, with 77 % myelopoiesis and predominance of eosinophilic granulocytes, metamyelocytes with rod-shaped nuclei and myelocytes. There was no increase in blasts.
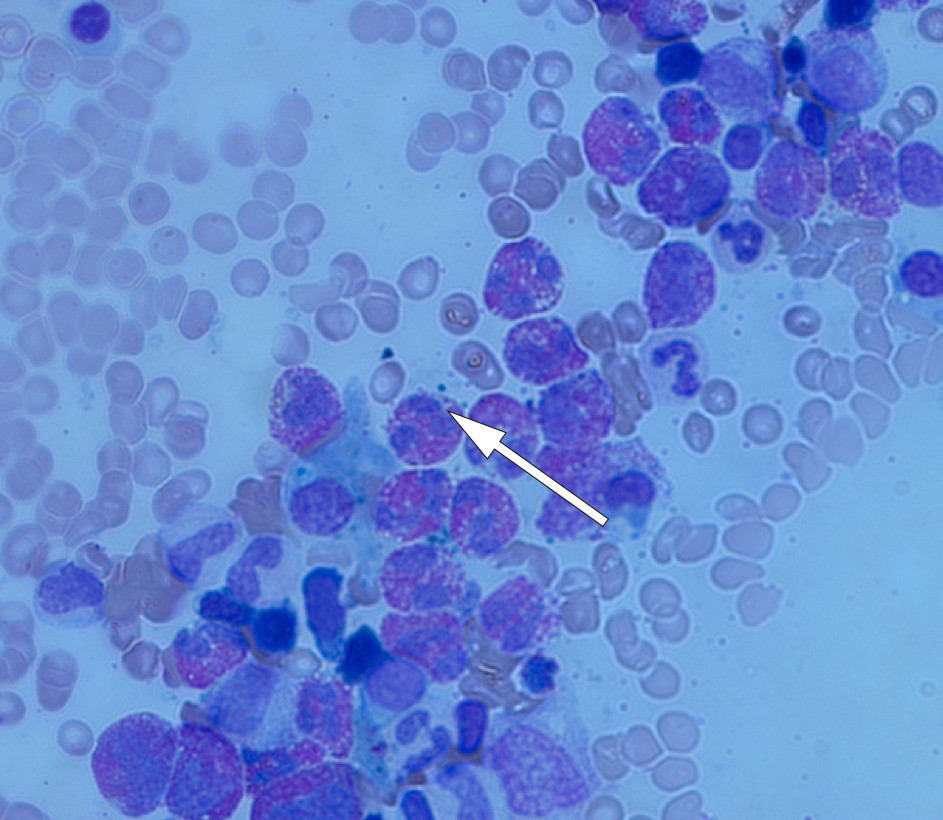
The combination negative serology, CT images without glandular swelling or clear signs of infection and bone marrow without lymphoma infiltration or an increase in blasts led to infection, lymphoma, leukaemia and autoimmunity being regarded as unlikely. A tentative diagnosis was now severe hypereosinophilic syndrome (HES), with multi-organ involvement of lungs, skin and bone marrow.
In light of the tentative diagnosis, treatment was commenced the same evening with high-dose prednisolone 1 mg/kg x 1 daily by mouth. Anticoagulation treatment was not regarded as necessary, as there was no sign of ongoing thrombosing.
A short time later, the bone marrow biopsy confirmed the findings from the bone marrow aspirate smear.
Further haematological assessment was planned with primary and secondary hypereosinophilic syndromes in mind.
The following day (day 8) the patient felt in great form. He had no dyspnoea, SpO2 was 96 % without supplementary O2. Blood tests now showed a fall in eosinophilic granulocytes to 1.8 · 109/L (0.0–0.4), and leukocytes to 13.5 · 109/L (3.9–9.5). The platelet count was unchanged. CRP had risen from 118 to 180 mg/L (0–4). The protein/creatinine ratio in urine was now found, and was 16 mg/mmol (0–14).
On day 9 a skin biopsy of the exanthema was taken from the big toe, with eosinophilic granulomatosis with polyangiitis (EGPA) in mind as a differential diagnosis for hypereosinophilic syndrome. The rashes on the lower extremities were still present, but less pronounced.
In case of ANCA-negative eosinophilic granulomatosis with polyangiitis, which more often affects the heart than the ANCA-positive variant, tests for cardiac markers were ordered. Pro-BNP was 114 ng/L (0–86), and high-sensitivity troponin T was 62–50 ng/L (0–14). ECG findings were normal. Elevated cardiac markers were regarded as part of multi-organ involvement, and no further cardiac assessment was conducted.
On the same day (day 9) the patient again wanted home leave in view of the subjective improvement, and there was a nice fall in CRP to 64 mg/L. Prior to his leaving, he was started on azitromax 500 mg x 1 by mouth for three days as a precaution against possible atypical pneumonia in view of the patchy opacities on the chest X-ray.
After two days (day 11) the patient returned from home leave. He now had a headache, neck pain and numbness, first of the left and then of the right hand. The patient was clearly in pain. His Glasgow Coma Scale (GCS) score was initially 15, but shortly after admission he experienced a generalised tonic-clonic seizure, which was halted with diazepam 5 mg intravenously. When he regained consciousness from the seizure, he was found to have flaccid paresis of the right upper extremity, reduced strength in the right lower extremity and an upward plantar reflex on the right side.
A head CT showed a left frontal cerebral haemorrhage. In the course of a few hours the patient developed increasing symptoms of high intracranial pressure, with an urge to vomit and impaired consciousness. He was intubated and placed on a ventilator, with surveillance, in the Intensive Care Department. He was hypertensive with systolic pressure 160–180 mm Hg. An infusion of labetalol 40 mg/hour was administered intravenously, and he was immediately transferred to the university hospital. A head CT there revealed increased haemorrhagia secondary to infarction-related changes, and venous sinus thrombosis was suspected. This was confirmed with CT venography. It was concluded that he had suffered a haemorrhagic infarction secondary to a clot in the sagittal sinus.
The patient's clinical history and treatment options were discussed with the duty neurosurgeon and haematologist. Anticoagulation treatment was decided against because of existing thrombocytopenia and massive haemorrhaging with breakthrough into the ventricular system. Thrombectomy was considered too risky in view of thrombocytopenia. It was decided to discontinue sedation and allow the patient to regain consciousness.
The following day the patient was in the Intensive Care Department, on a ventilator. He was comatose, with no spontaneous movements. Over a short space of time that afternoon he developed dilated pupils with no reaction to light. A new CT scan revealed massive cerebral oedema and herniation. Thirteen days after hospitalisation the patient was declared brain-dead. Organ donation was not possible because of undetermined underlying disease.
Autopsy revealed infiltration of eosinophilic granulocytes into the wall of the sagittal sinus. Neuropathological examination confirmed haemorrhagic infarction and venous sinus thrombosis. Histology sections taken from several organs revealed vasculitis with eosinophilic granulocytes in the walls of medium-sized arteries in the liver (Figure 2) and in an arterial branch in the meninges. These findings are consistent with eosinophilic granulomatosis with polyangiitis as the cause of the hypereosinophilic syndrome that was detected clinically.
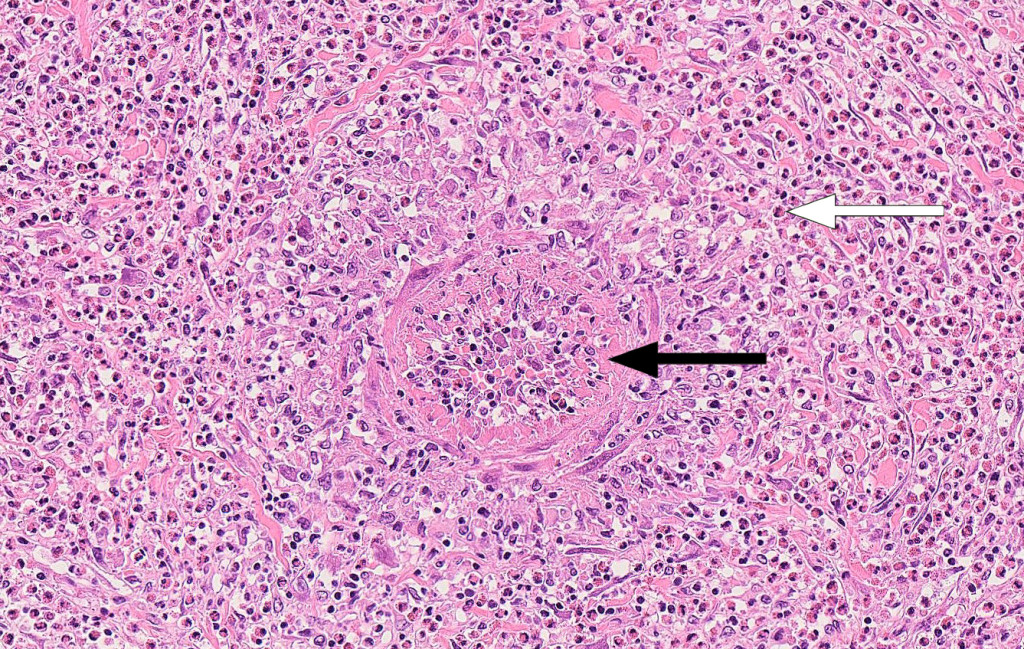
Discussion
Eosinophilic granulomatosis with polyangiitis is a systemic vasculitis that prior to 2012 was called Churg-Strauss syndrome (1). The condition is characterised by allergic rhinitis, asthma and pronounced eosinophilia in peripheral blood (2–4). The definition has been altered several times since the condition was first described in 1951 (2), and today there are more classification criteria Box 1) ((1, 5, 3, 4). The purpose of these changes has been to increase the sensitivity and specificity of the diagnosis and also make faster diagnosis possible, for example by dropping biopsy as a diagnostic requirement.
American College of Rheumatology (1990) (5)
Clinical findings with or without pathological material – diagnosis probable when four of six criteria are present:
Asthma
Eosinophilia > 10 %
Neuropathy, mononeuropathy or polyneuropathy
Pulmonary infiltrates
Paranasal sinus abnormality
Extravascular eosinophil infiltration in biopsy specimens
Chapel Hill Consensus Conference (1994, revised in 2012) (1)
Pathological and clinical findings:
Eosinophil-rich and granulomatous inflammation involving the respiratory tract, and necrotising vasculitis that affects small to medium-sized arteries and is associated with asthma and eosinophilia.
The disease is very rare. The incidence is 0.11–2.66 new cases per 1 million people per year and the prevalence 10.7–14 per 1 million adults (6). Generally speaking, eosinophilic granulomatosis with polyangiitis is considered to be a milder type of systemic vasculitis, with lower mortality than other systemic vasculitides (7). Given appropriate treatment, five-year survival is 70–90 % (8). The involvement of the gastrointestinal tract or the heart is associated with a poor clinical outcome (9). Our patient had an unusual presentation with abdominal pain, fever and general malaise combined with rapidly progressive eosinophilia and signs of multi-organ involvement.
Conventional characteristics of eosinophilic granulomatosis with polyangiitis have been a prodromal phase characterised by asthma and rhinosinusitis, then an eosinophil phase with peripheral eosinophilia and organ involvement and finally a vasculitis phase with clinical manifestations of small vessel vasculitis (10). These phases are not always clearly separated.
In cases of eosinophilic granulomatosis with polyangiitis, there is asthma in the clinical history in 96–100 % of cases (9, 10). According to the literature, asthma occurs later in the vasculitic phase in exceptional cases (9). Our patient had low-grade atopic eczema as a child, but no known eczema, allergy or asthma as an adult. The patient was assessed by a rheumatologist and dermatologist on day 4, without vasculitis being viewed as a probable diagnosis. On hospitalisation his leukocyte count was 10.3 · 109/L (3.9–9.5). This rose rapidly to 25.3 · 109/L a week after admission, but the differential count that showed pronounced eosinophilia was unfortunately only taken on day 7. It was only then that hypereosinophilic syndrome was suspected.
Eosinophilic granulomatosis with polyangiitis has features in common with hypereosinophilic syndrome (Table 1) (6). We interpreted the patient's condition first as hypereosinophilic syndrome, and started treatment for that. In the initial phase, both conditions are treated with high-dose corticosteroids. Although hypereosinophilia is associated in both conditions with an increased risk of thromboembolism, the effect of anticoagulation treatment on these patients is a matter of contention. In the absence of signs of evident thrombosis, it is not recommended as primary prophylaxis (11).
Table 1
Comparison of clinical manifestations of hypereosinophilic syndrome and eosinophilic granulomatosis with polyangiitis (6)
| Clinical manifestations |
Hypereosinophilic syndrome |
Eosinophilic granulomatosis with polyangiitis |
|---|---|---|
| Cardiac involvement |
Endocardial fibrosis, restrictive cardiomyopathy |
Involvement of pericardium and myocardium, small-vessel vasculitis |
| Asthma |
Rarely |
Almost always |
| Sinusitis |
Rarely |
Commonly |
| Involvement of peripheral nerves |
Rarely |
Commonly |
| Skin involvement |
Urticaria |
Purpura, urticaria |
| Increased blood sedimentation rate |
Rarely |
Commonly |
| Increased IgE |
Rarely |
Commonly |
| Vasculitis verified by biopsy |
Absent |
Present (particularly in late phase) |
| Antineutrophil cytoplasmic autoantibodies |
Absent |
Present in about 40 % of patients |
After a while the patient developed lung infiltrates, a very common finding with eosinophilic granulomatosis with polyangiitis. He also had a vasculitis-suspicious rash, and one wonders retrospectively whether his walking problems were an expression of polyneuropathy.
The nervous system is often affected in cases of eosinophilic granulomatosis with polyangiitis, and the American College of Rheumatology has this among its classification criteria – but as peripheral neuropathy Box 1). Central nervous involvement is far less common, and receives little mention in the literature. When it occurs, it is described as cerebral infarction and cerebral haemorrhage ((9, 12, 13).
Anti-neutrophil cytoplasmic antibodies (ANCA) are detected in 40–60 % of patients, and eosinophilic granulomatosis with polyangiitis is classified among the ANCA-associated vasculitides (14, 15). ANCA negativity may be associated with cardiac involvement, gastrointestinal involvement, pulmonary infiltrates, life-threatening incidents and death (16), as was the case with our patient.
Even though he was started on corticosteroids, which is the cornerstone treatment for eosinophilic granulomatosis with polyangiitis (9, 14), the patient's life could not be saved. An earlier differential count of leukocytes could probably have contributed to earlier treatment start-up. The skin biopsy taken on day 9 showed superficial perivascular lymphocytic inflammation, which is a non-specific finding. However, the biopsy was taken two days after the start of steroids, and it is uncertain whether an earlier skin biopsy would have provided an earlier diagnosis.
The patient had several poor prognostic signs, such as involvement of the gastrointestinal tract (liver), myocardium and central nervous system. As the patient showed a god response, both clinical and biochemical, 1–2 days after starting on prednisolone, there were no obvious objections to him going on home leave. Venous sinus thrombosis and haemorrhaging occurred after the eosinophilic granulocyte count had normalised, and it is thus uncertain whether the outcome would have been any different if the diagnosis had been made earlier.
The patient's family have consented to the publication of the article. The article has been peer-reviewed.
- 1.
Jennette JC, Falk RJ, Bacon PA et al. 2012 revised international Chapel Hill consensus conference nomenclature of vasculitedes. Arthritis Rheum 2013; 65: 1–11. [PubMed][CrossRef]
- 2.
Churg J, Strauss L. Allergic granulomatosis, allergic angiitis, and periarteritis nodosa. Am J Pathol 1951; 27: 277–301. [PubMed]
- 3.
Noth I, Strek ME, Leff AR. Churg-Strauss syndrome. Lancet 2003; 361: 587–94. [PubMed][CrossRef]
- 4.
Skoner DP. Allergic rhinitis: definition, epidemiology, pathophysiology, detection, and diagnosis. J Allergy Clin Immunol 2001; 108: S2–8. [PubMed][CrossRef]
- 5.
Masi AT, Hunder GG, Lie JT et al. The American College of Rheumatology 1990 criteria for the classification of Churg-Strauss syndrome (allergic granulomatosis and angiitis). Arthritis Rheum 1990; 33: 1094–100. [PubMed][CrossRef]
- 6.
Baldini C, Talarico R, Della Rossa A et al. Clinical manifestations and treatment of Churg-Strauss syndrome. Rheum Dis Clin North Am 2010; 36: 527–43. [PubMed][CrossRef]
- 7.
Mukhtyar C, Flossmann O, Hellmich B et al. Outcomes from studies of antineutrophil cytoplasm antibody associated vasculitis: a systematic review by the European League Against Rheumatism systemic vasculitis task force. Ann Rheum Dis 2008; 67: 1004–10. [PubMed][CrossRef]
- 8.
Saku A, Furuta S, Hiraguri M et al. Longterm outcomes of 188 Japanese Patients with eosinophilic granulomatosis with polyangiitis. J Rheumatol 2018; 45: 1159–66. [PubMed][CrossRef]
- 9.
Guillevin L, Cohen P, Gayraud M et al. Churg-Strauss syndrome. Clinical study and long-term follow-up of 96 patients. Medicine (Baltimore) 1999; 78: 26–37. [PubMed][CrossRef]
- 10.
Lanham JG, Elkon KB, Pusey CD et al. Systemic vasculitis with asthma and eosinophilia: a clinical approach to the Churg-Strauss syndrome. Medicine (Baltimore) 1984; 63: 65–81. [PubMed][CrossRef]
- 11.
Ogbogu PU, Rosing DR, Horne MK. Cardiovascular manifestations of hypereosinophilic syndromes. Immunol Allergy Clin North Am 2007; 27: 457–75. [PubMed][CrossRef]
- 12.
André R, Cottin V, Saraux JL et al. Central nervous system involvement in eosinophilic granulomatosis with polyangiitis (Churg-Strauss): Report of 26 patients and review of the literature. Autoimmun Rev 2017; 16: 963–9. [PubMed][CrossRef]
- 13.
Sabio JM, Rivero-Rodriguez M, Vargas-Hitos JA. Demographic and clinical characteristics associated with central nervous system hemorrhage in patients with eosinophilic granulomatosis with polyangiitis: A case report and review of the literature. J Rheumatol 2017; 44: 1413–5. [PubMed][CrossRef]
- 14.
Conron M, Beynon HL. Churg-Strauss syndrome. Thorax 2000; 55: 870–7. [PubMed][CrossRef]
- 15.
Pagnoux C, Guillevin L. Churg-Strauss syndrome: evidence for disease subtypes? Curr Opin Rheumatol 2010; 22: 21–8. [PubMed][CrossRef]
- 16.
Healy B, Bibby S, Steele R et al. Antineutrophil cytoplasmic autoantibodies and myeloperoxidase autoantibodies in clinical expression of Churg-Strauss syndrome. J Allergy Clin Immunol 2013; 131: 571–6.e1, 6. [PubMed][CrossRef]