A woman in her sixties with COVID-19-related acute respiratory distress syndrome was critically ill with no improvement after three weeks of intensive care. On her nineteenth day on a ventilator she was treated with corticosteroids. Clinical improvement was rapid, and she could be weaned from ventilatory support. Her improvement may have been fortuitous or a result of other factors, but we believe this case study contributes to the discussion concerning the use of corticosteroid treatment in severe COVID-19.
A woman in her sixties was admitted to a local hospital in mid-March after ten days with a cold, joint pains, headache and fever. She had been curatively treated for breast cancer some years earlier, had ceased smoking the same year and used no regular medicines. On her fifth day of symptoms, the patient had contacted her GP. C-reactive protein (CRP) was then 20 mg/l (reference range < 5) and ferritin 550 µg/l (15–350). The following days she had reduced appetite, laboured breathing even from slight activity and passed watery stools several times a day. When the patient again contacted her GP on the ninth day, her CRP level was 177 mg/l, peripheral oxygen saturation (SpO2) without additional oxygen 94 % and her respiratory rate 22 per minute. Her GP started penicillin therapy for assumed pneumonia. After one day of antimicrobial therapy the patient was still in poor condition, with dyspnoea and a fever of 39.5 °C. She was then admitted to the local hospital.
SpO2 on admission was 88 %. This rose to 93 % with 4 l O2 by nasal catheter. The patient herself did not experience her breathing as laboured; her respiratory rate was 22 per minute. Other vital signs were normal: blood pressure 127/61 mm Hg, pulse rate 74 per minute and rectal temperature 36.8 °C. Crackles were heard basally over the right lung on auscultation, otherwise clinical findings were normal. Blood gases without supplementary oxygen revealed pH 7.41 (7.35–7.45), pO2 6.0 kPa (> 9.3), pCO2 4.3 kPa (4.3–6), bicarbonate (HCO3−) 21 mmol/l (22–27), base excess (BE) −4 mmol/l (−2–+3), lactate 1 mmol/l (0.4–0.8) and oxygen saturation (SaO2) 84 % (> 94 %). CRP was 172 mg/l, leukocytes 7.6 · 109/l (3.5–11), lactate dehydrogenase (LD) 325 U/l (105–205), d-dimer 1.0 (< 0.5), creatinine 105 µmol/l (45–90), alanine aminotransferase (ALT) 51 U/l (< 45), aspartate aminotransferase (AST) 58 U/l (< 35), alkaline phosphatase (ALP) 100 U/l (< 105) and gamma glutamyl transferase (GGT) 211 U/l (< 75).
A nasopharyngeal polymerase chain reaction (PCR) test was conducted for pulmonary pathogenic bacteria and viruses, including SARS-CoV-2, and blood and urine cultures were obtained. Intravenous therapy for possible bacterial pneumonia was started (penicillin 1.2 mg x 4 and erythromycin 500 mg x 4), and the patient was put into isolation. A chest X-ray taken the following day showed diffuse small opacities in both lungs and basal atelectasis on the right side.
The night after admission, she was given non-invasive ventilatory treatment with continuous positive airway pressure (NIV-CPAP). This was initially tolerated, but after 12 hours of treatment saturation fell to 85 %. She was given 7 l/min O2 by mask instead, and saturation settled at 89 %. During her first 24 hours in hospital she passed watery stools 10–12 times and desaturated significantly (down to SpO2 64 %) in connection with visits to the toilet.
Two days after admission she was reported to be positive for SARS-CoV-2. She was judged to be in imminent need of a ventilator, and was therefore transported urgently to the central hospital in an Epishuttle incubator with 9 l/min O2 by mask.
Blood gases on arrival at the central hospital (with 9 l/min O2 by mask) showed pH 7.48, pO2 6.2, pCO2 3.9, HCO3− 22, BE −2, lactate 1.7 and Sa02 84 %. The patient met all of the Berlin criteria (1) for severe acute respiratory distress syndrome (ARDS) (pO2/Fi O2 < 13.3 kPa/< 100 mm Hg). Intubation was uncomplicated and ventilator treatment commenced under deep sedation on day 3 after admission to the local hospital. Pressure-controlled ventilation was established: inspiratory pressure 15 cm H2O, positive end-expiratory pressure (PEEP) 10 cm H2O and respiratory frequency 20 per minute.
A short time after intubation the oxygen concentration in the inhaled air (FiO2) could be reduced from 80 % to 65 %, and a percutaneous dilatational tracheostomy was established. Antimicrobial therapy was changed to intravenous piperacillin-tazobactam 4 g x 4 and azithromycin 500 mg x 1, according to a locally developed procedure. Antimycotic therapy consisting of intravenous caspofungin 50 mg x 1 was added. After a week, azithromycin and caspofungin were discontinued. Azithromycin was terminated because she had intermittent AV block, and the indication for continued treatment was regarded as weak. Caspofungin was terminated because fungal infection was regarded as clinically unlikely.
Five days after admission to the local hospital the patient had a CRP value of 380 mg/l and still required a substantial amount of oxygen. With the patient in the prone position, FiO2 could be reduced from 0.8 to 0.5. In order to avoid high airway pressure (> 30 cm H2O) we allowed high pCO2 and a subsequent pH of around 7.20 (permissive hypercapnia). In light of promising reports from France and China (2, 3), we decided six days after admission to initiate hydroxychloroquine 600 mg × 2 in a saturation dose by gastric tube, followed by 200 mg × 3 for ten days (equivalent to the dose in the planned WHO study).
The patient appeared to be improving 11 days after the initial hospitalisation (Figure 1). She was easier to oxygenate in the supine position, and her CRP value had fallen to 128 mg/l. Subsequently, her condition deteriorated considerably, and she became more difficult to ventilate. Three days later, her CRP had risen to 343 mg/l, Fi O2 had to be increased and we again had to place her in a prone position. Despite negative findings from bacteriological cultures (five blood cultures, seven expectorate cultures and three urine cultures), we chose to switch antibiotics to meropenem 2 g x 3.
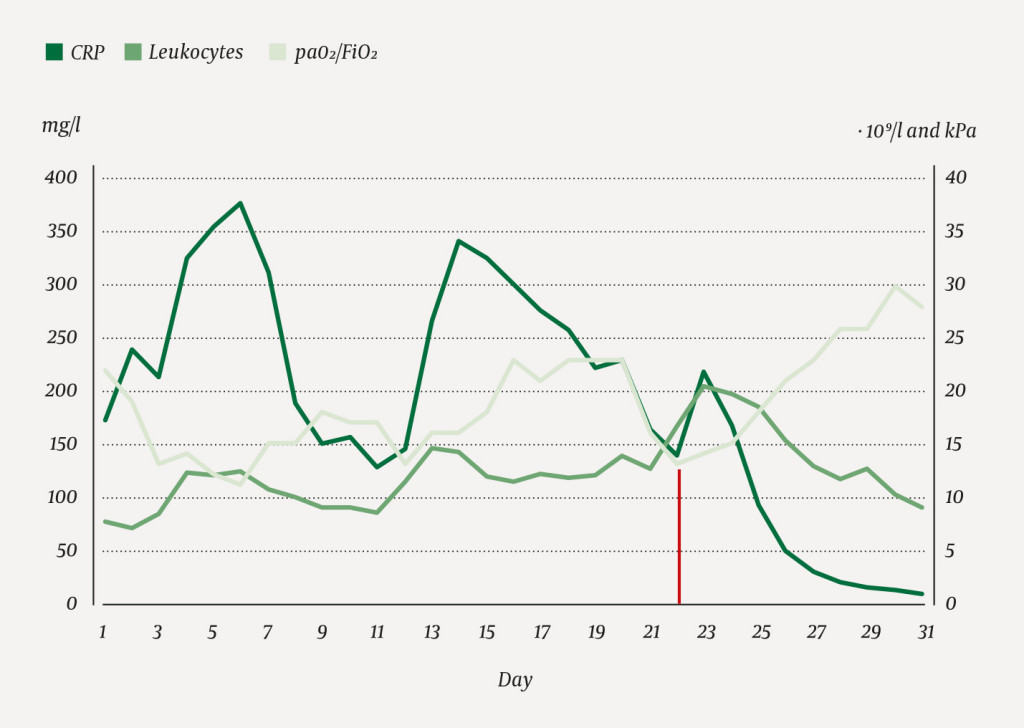
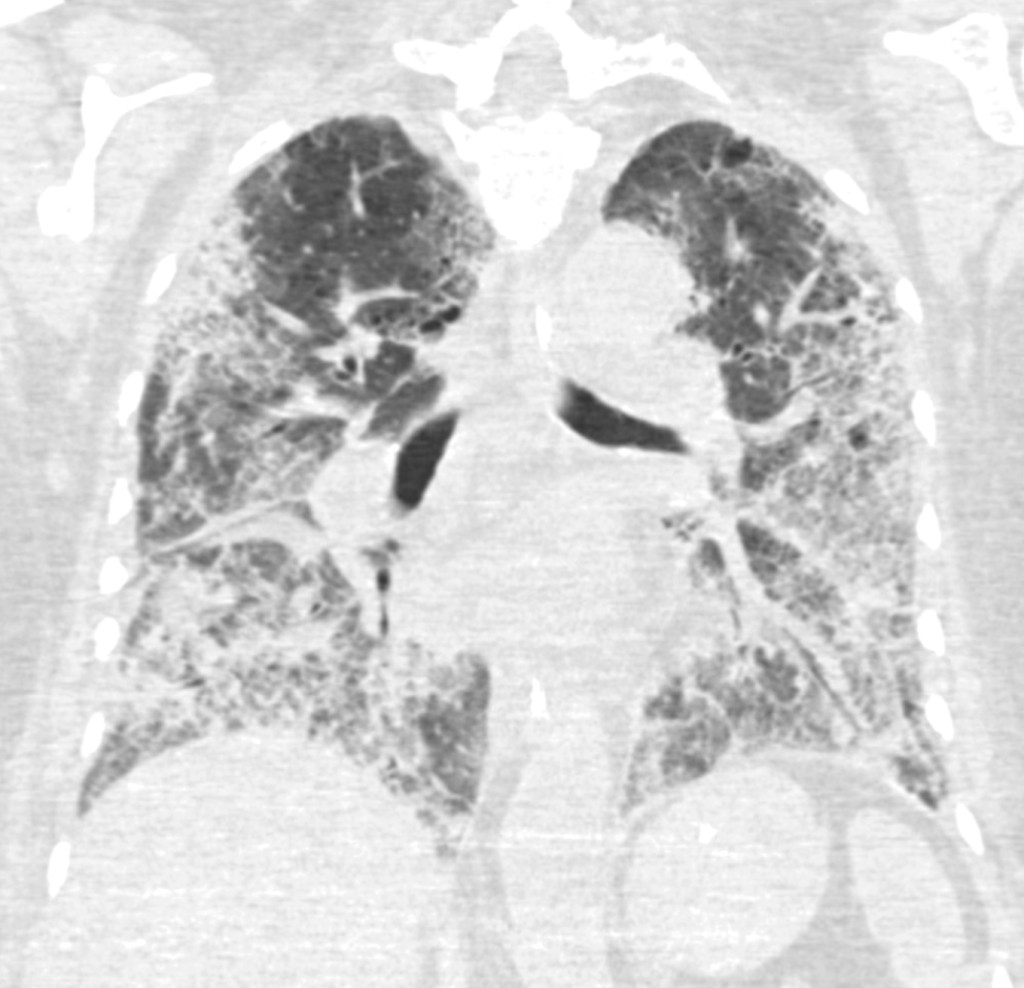
Eighteen days after hospitalisation, a CT scan (Figure 2) showed bilateral pulmonary opacities and peripheral pulmonary embolisms. The prophylactic dose of anticoagulant that the patient had been given since she was placed on a ventilator (dalteparin 5 000 U x 1 subcutaneously), was therefore increased to a weight-adjusted therapeutic dose for pulmonary embolisms (12 500 U x 1 subcutaneously). Her CRP value remained high despite broad-spectrum antimicrobial therapy, and for the third time the patient required extensive O2 support, with unchanged ventilatory input.
As therapists, we felt that we had our backs to the wall. We examined the literature in search of evidence for potentially effective treatment, and found weak recommendations for trying corticosteroids on patients with severe virus-induced ARDS (4–6). After interdisciplinary discussion, we chose on day 22 after hospitalisation (day 19 on ventilation) to administer corticosteroid therapy in the form of Solu-Medrol 80 mg (equipotent to dexamethasone 20 mg) x 1 intravenously for five days, then 40 mg x 1 for five days (5).
Two days after the start of the corticosteroid therapy, the patient was markedly easier to oxygenate, had a rising PaO2/FiO22 ratio and rapidly falling CRP and leukocyte values, after the latter had initially risen sharply the day after administration of corticosteroids (Figure 1). The patient could then be gradually weaned from the ventilator, treatment that by the time of extubation had lasted for a total of 28 days.
Thirty-eight days after admission, the patient was transferred from the Intensive Care Unit to a ward. She had lost 15 kilos. SpO2 was then 92 % with 1 l O2 by nasal catheter, with desaturation to 87–88 % on mobilisation. She was discharged to a specialised rehabilitation centre 56 days after her admission to the local hospital.
Discussion
Our patient with severe COVID-19 had typical onset symptoms, increasing saturation problems and developed severe acute respiratory distress syndrome (ARDS) with a need for prolonged ventilatory support. She had a triple-peaked course, and negative findings from bacterial cultures and lack of response to antimicrobial therapy, indicated that her severe need for ventilator support was probably due to a systemic inflammatory response, and not a secondary bacterial infection. Twenty-two days after hospitalisation the prognosis was sombre.
During the course of the patient's illness, recommendations for treatment were constantly being updated. Several of the clinical decisions made along the way are open to debate: the use of a transport incubator, pressure-controlled ventilatory support, ventilation in the prone position and early treatment with hydroxychloroquine and broad-spectrum antibiotics. The improvement following treatment with corticosteroids may have been fortuitous or due to other factors.
The World Health Organization's March recommendations advised in principle against treating patients with COVID-19 and acute respiratory distress syndrome with corticosteroids (7). The Surviving Sepsis Campaign arrived at a weak recommendation in favour of treating such patients with corticosteroids in low doses and for a short time (4). In its May update, also the WHO included the possibility of considering corticosteroid therapy for severe acute respiratory distress syndrome (8).
Five days after the present article was first submitted, Nature published a press release on the main findings of the RECOVERY trial (9), and a month later the preliminary results – which in the meantime were publicly available (10) – were published in the New England Journal of Medicine (11). The study showed significantly lower mortality (29.3 % versus 41.4 %) for patients with severe COVID-19 on ventilatory support who received dexamethasone 6 mg daily for ten days. This was given to patients ten days (on average) after symptom onset, corresponding to the first serious exacerbation for our patient (hospitalisation day 11 after symptom onset). In the study, the effect of dexamethasone was most pronounced among patients who had been on ventilatory support for more than seven days, for whom inflammatory lung changes probably play a greater part.
We treated our patient in accordance with the evidence-based recommendations at the time, when knowledge about COVID-19 was limited. At the time, UpToDate recommended that if corticosteroids were to be used to treat acute respiratory distress syndrome, dexamethasone 20 mg x 1 for five days followed by 10 mg x 1 for five days should be used (5). This is the dose administered to our patient. UpToDate's recommendation has subsequently been changed specifically for COVID-19 in light of the findings in the RECOVERY trial, i.e. 6 mg x 1 for 10 days (6). Thus, our patient first received corticosteroid therapy at the time of the third exacerbation, three times as long a time from symptom onset as the average in the RECOVERY trial, and with three times as high a dose of an equipotent corticosteroid.
The RECOVERY trial may come to change practice for patients with severe COVID-19. In retrospect, one could question whether our patient should have received the treatment earlier. We believe this case study contributes to shedding light on the use of corticosteroids in patients with COVID-19.
We wish to thank all staff who were involved in treating the patient.
The patient has consented to the publication of the article.
The article has been peer-reviewed.
- 1.
Ranieri VM, Rubenfeld GD, Thompson BT et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012; 307: 2526–33. [PubMed]
- 2.
Gautret P, Lagier JC, Parola P et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 2020; 56: 105949. [PubMed][CrossRef]
- 3.
Chen J, Liu D, Liu L et al. A pilot study of hydroxychloroquine in treatment of patients with moderate COVID-19. Zhejiang Da Xue Xue Bao Yi Xue Ban 2020; 49: 215–9. [PubMed]
- 4.
Alhazzani W, Møller MH, Arabi YM et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med 2020; 46: 854–87. [PubMed][CrossRef]
- 5.
Uptodate. Acute Respiratory Distress Syndrome: Supportive Care and Oxygenation in Adults. https://www.uptodate.com/contents/acute-respiratory-distress-syndrome-supportive-care-and-oxygenation-in-adults?sectionName=Glucocorticoids&search=covid%252&topicRef=127419&anchor=H1087880222&source=see_link#H1087880222 Accessed 6.4.2020.
- 6.
Uptodate. Coronavirus disease 2019 (COVID-19): Critical care and airway management issues. https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-critical-care-and-airway-management-issues?sectionName=Glucocorticoids&search=covid%252#H4152848011 Accessed 6.4.2020 og 30.7.2020.
- 7.
WHO. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected. Interim guidance 13 March 2020. https://www.who.int/docs/default-source/coronaviruse/clinical-management-of-novel-cov.pdf Accessed 6.4.2020.
- 8.
WHO. Clinical management of severe acute respiratory infection (SARI) when COVID-19 disease is suspected. Interim guidance 27 May 2020. https://apps.who.int/iris/bitstream/handle/10665/332196/WHO-2019-nCoV-clinical-2020.5-eng.pdf?sequence=1&isAllowed=y Accessed 6.6.2020.
- 9.
Ledford H. Corona virus breakthrough: dexamethasone is first drug shown to save lives. Nature 16.6.2020. https://www.nature.com/articles/d41586-020-01824-5 Accessed 16.6.2020.
- 10.
Horby P, Lim WS, Emberson J et al. Effect of Dexamethasone in hospitalized patients with Covid-19: Preliminary Report. MedRXiv 2020.
- 11.
Horby P, Lim WS, Emberson JR et al. Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report. N Engl J Med 2020; 383: NEJM2021436. [PubMed]