Main Findings
When the response to the COVID-19 pandemic had intensified in Norway, we saw a general reduction in the number of registered cases of other notifiable diseases.
The reduction was observed in all included disease groups.
On 30 January 2020 (week 5), the World Health Organization declared the novel coronavirus outbreak a public health emergency of international concern (PHEIC). On the next day, the Norwegian Ministry of Health and Care Services added COVID-19 to the list of notifiable diseases, irrespective of clinical symptoms.
On 26 February (week 9), the first case of COVID-19 was detected in Norway. On 12 March (week 11), Norwegian authorities imposed strict control measures to limit contacts between persons, and on 14 March (week 11) travel restrictions were introduced with quarantine for people who entered Norway. During weeks 10–14, laboratories increased their capacity to investigate suspected COVID-19 cases. The number of suspected cases tested increased from 400 tests per day during week 10, to 6 000 tests per day during week 12, and has since remained at a high level (1).
During the course of intensified COVID-19 response and increased incidence of COVID-19 cases, we observed a reduction in the number of notifications of other infectious diseases to the Norwegian Surveillance System for Communicable Diseases (MSIS). Since 9 March, the number of COVID-19 cases reported to MSIS has exceeded the cumulative number of all other notifications (Figure 1).
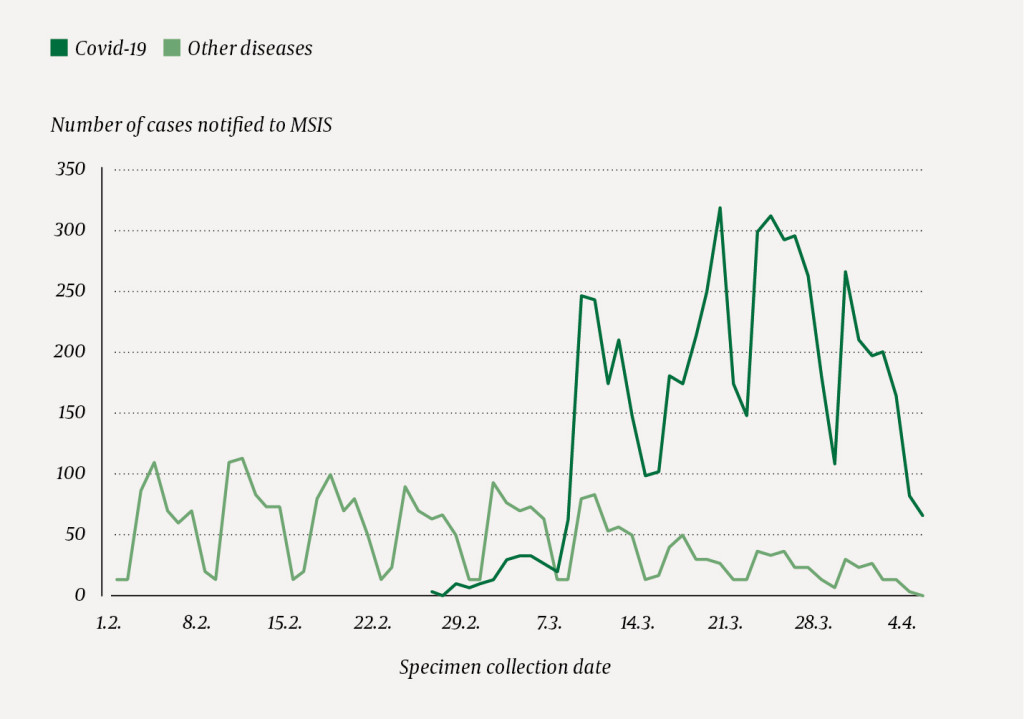
In this study, we wanted to compare reporting of notifiable diseases in weeks 6–14 of 2020 with the previous three years to assess whether intensified COVID-19 response may have affected reporting to MSIS of infectious diseases other than COVID-19.
Material and method
Data were retrieved from MSIS and consisted of the number of notifiable disease cases reported during weeks 6–14 in four consecutive years (2017–2020). We included only notifiable diseases that have been subject to notification throughout the period (Box 1, Appendix 1).
Included:
-
Campylobacteriosis, cryptosporidiosis, E. coli gastroenteritis, giardiasis, hepatitis A, listeriosis, paratyphoid fever, salmonellosis, shigellosis, typhoid fever and yersiniosis (classified as food and waterborne diseases)
-
Gonorrhoea, acute hepatitis B, HIV infection and syphilis (classified as sexually transmitted and blood borne infections)
-
Invasive Hib disease, invasive meningococcal disease, invasive pneumococcal disease, measles, mumps, pertussis and rubella (classified as vaccine preventable diseases)
-
Methicillin resistant S. aureus (MRSA), penicillin resistant pneumococci (PRP), vancomycin resistant enterococci (VRE) and carbapenemase producing organisms (CPO) (classified as antibiotic resistant infections)
-
Brucellosis, dengue fever, echinococcosis, legionellosis, Lyme borreliosis, malaria, nephropathia epidemica, prion diseases, Q fever, invasive strep A disease, invasive strep B disease, tularemia and viral infections of the central nervous system (classified as other diseases and syndromes)
Excluded:
-
Clostridium difficile infection, lymphogranuloma venereum, rotavirus infections, Zika fever, West Nile fever, Shewanella and Vibrio infections (notifiable since 2019)
-
Anthrax, botulism, cholera, diphtheria, haemorrhagic fevers, influenza with epidemic potential, leprosy, plague, poliomyelitis, rabies, relapsing fever, SARS, smallpox, spotted fever rickettsioses, tetanus, trichinosis and yellow fever (no cases reported during evaluated period).
-
AIDS, chronic hepatitis B, hepatitis C and tuberculosis because of the long incubation.
To assess the association between the control measures against COVID-19 and the reporting of other disease groups, we summarised the number of cases reported per week during weeks 6–14. We then compared the number of cases of each disease group reported during a given week in 2020 with the median of cases reported in the respective week during the three previous years. We describe a percentage change from 2017–2019 to 2020.
We applied for and received aggregated anonymous data from MSIS. These are publicly available data, as the number of cases of each disease reported to MSIS is published daily on www.msis.no.
Results
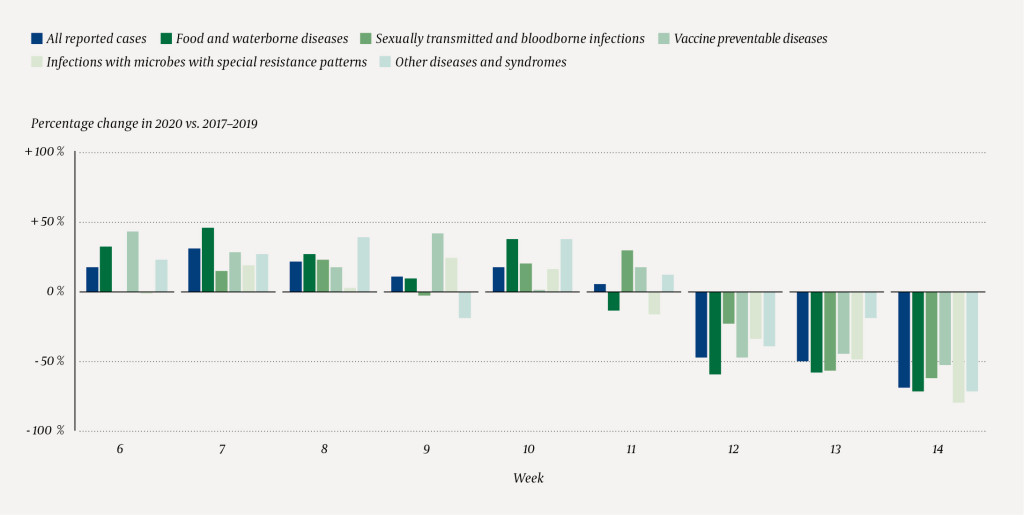
During weeks 6–14 in 2020, 2 250 cases of included infectious diseases were registered, which corresponded with reported frequency in previous years: 2 632 in 2019, 2 250 in 2018 and 2 283 in 2017. Although the total number of cases in the evaluated period was similar to previous years, we saw a noticeable difference in the number of cases reported in 2020 before and after week 11. During weeks 6–11, there were more cases reported in 2020 than during the corresponding weeks in 2017–2019 (Figure 2). Compared to the median number of cases reported in corresponding weeks in 2017–2019, physicians and laboratories reported 47 % fewer cases (159 vs. 301) in week 12, 50 % fewer cases (131 vs. 261) in week 13 and 69 % fewer cases (77 vs. 252) in week 14. During week 14, we observed marked reductions in all disease groups compared with the previous three years: antibiotic resistant infections (–80 %, 11 vs. 55), food and waterborne diseases (–71 %, 24 vs. 83), other diseases and syndromes (–71 %, 6 vs. 21), sexually transmitted and bloodborne infections (–62 %, 14 vs. 37), and vaccine preventable diseases (–53 %, 22 vs. 47) (Figure 2).
Discussion
The apparent reduction in the number of reported infectious diseases other than COVID-19 may reflect a generally reduced risk of communicable diseases. Increased social distancing results in reduced contacts between people, and thus a decreased likelihood of transmission and incidence of infectious diseases. This will be particularly relevant for infectious diseases transmitted via droplets or aerosols (e.g. whooping cough or pneumococcal infections), or where infection occurs mainly through direct and indirect contact (e.g. MRSA).
Furthermore, closure of restaurants and increased dissemination of hand hygiene advice may have reduced the risk of food contamination, both in connection with catering services and home cooking, which may have resulted in lower risk of foodborne infections. Higher awareness of infection prevention measures among health professionals may have contributed to a better prevention of nosocomial infections. The travel restrictions, with fewer travellers and decreased trade in live animals and food, have reduced the likelihood of infectious disease spread across borders.
There is no sufficient evidence to determine an association between public health measures that reduce contact between people and reduced incidence of infectious diseases, with the exception of the effect that school closures can have on decreased transmission of influenza (2). SARS-CoV-2 is a new virus and most countries have implemented comprehensive measures to deal with COVID-19. However, it is difficult to assess the impact of the various measures so early in the epidemic. Social distancing measures have not previously been used to the extent to which they are currently implemented. Thus, there is no strong evidence on the effect of such measures on reducing the incidence of infectious diseases. Systematic reviews have been published assessing the effect of school closures on COVID-19 infection risk (3, 4). These knowledge summaries and models have focused on the effect of person-to-person airborne transmission, but have not considered how the measures can affect other transmission routes that can contribute to the spread of various infectious diseases.
The correlation between control measures against COVID-19 and reporting of other disease groups may also indicate a reduced sensitivity of surveillance of infectious diseases other than COVID-19. First, it is likely that most people working in communicable disease surveillance, both locally and nationally, have had to re-prioritise ongoing activities during February–April 2020 in order to use the resources to deal with the COVID-19 epidemic. The working days have been filled with finding and following up new COVID-19 cases and their contacts, providing guidance and protecting health professionals, and communicating necessary information to the general population. Second, clinical microbiological laboratories were under great pressure on their human resources and materials use, since the volume of weekly specimens has been well above what they routinely processed before the epidemic. Third, the recommendation on social distancing and fear of infection may have led many to avoid seeking health care, especially if symptoms were perceived as mild and not requiring immediate medical attention.
We cannot disentangle to what extent the reduction of reporting of particular notifiable diseases is the result of competing priorities and increased pressure on public health personnel and laboratories, or a real reduction in the spread and prevalence of communicable diseases. The number of negative specimens could give us some answers, but unfortunately, we do not have access to this data at national level.
Our main goal with this overview is to increase awareness among health professionals and local and national health authorities that such potential effect of the efforts to control COVID-19 can lead to decreased attention and prevention of other infectious diseases. With reduced support for reporting systems, our ability to detect signals on potential threats is hampered. This can lead to long-term negative consequences that can be difficult to address and resolve when resources are no longer needed for COVID-19. We therefore recommend identifying priority conditions for public health, and ensuring maintaining of necessary resources for their monitoring, prevention and control.
The Norwegian Institute of Public Health has placed special emphasis on maintaining the childhood vaccination programme (5) and will use the national vaccination register (SYSVAK) to regularly monitor and respond quickly to signs of reduced vaccination coverage (6). Due to uncertainty in global markets and trade restrictions in many parts of the world, we can also expect increasing food security challenges. This can be very difficult to counter globally, but more importantly can be challenging to monitor nationally (7).
Conclusion
We have documented a temporal association between response to COVID-19 and a noticeable reduction in reported cases of other infectious diseases to MSIS. Although reduced support to the surveillance system may be a possible cause, this is likely to be a temporary effect and resolve after the decline of the COVID-19 pandemic. Nevertheless, the large redistribution of resources can continue for months before reporting systems restore the required sensitivity. We therefore recommend maintaining of reporting and monitoring of other infectious diseases. We also recommend a continued prioritisation of critical public health resources to prevent that efforts aimed at COVID-19 response lead to adverse effects of other infectious diseases on population health.
The article has been peer-reviewed.
- 1.
The Norwegian Institute of Public Health. Daily and weekly reports on COVID-19 in Norway (in Norwegian). https://www.fhi.no/sv/smittsomme-sykdommer/corona/dags--og-ukerapporter/dags--og-ukerapporter-om-koronavirus/ Accessed 12.5.2020.
- 2.
Jackson C, Mangtani P, Hawker J et al. The effects of school closures on influenza outbreaks and pandemics: systematic review of simulation studies. PLoS One 2014; 9. doi: 10.1371/journal.pone.0097297. [PubMed][CrossRef]
- 3.
Herrera-Diestra JL, Meyers LA. Local risk perception enhances epidemic control. PLoS One 2019; 14. doi: 10.1371/journal.pone.0225576. [PubMed][CrossRef]
- 4.
Viner RM, Russell SJ, Croker H et al. School closure and management practices during coronavirus outbreaks including COVID-19: a rapid systematic review. Lancet Child Adolesc Health 2020; 4: 397–404. [PubMed][CrossRef]
- 5.
World Health Organization. Guidance on routine immunization services during COVID-19 pandemic in the WHO European Region. Copenhagen; WHO, March 2020. http://www.euro.who.int/en/health-topics/communicable-diseases/hepatitis/publications/2020/guidance-on-routine-immunization-services-during-COVID-19-pandemic-in-the-who-european-region-2020 Accessed 12.5.2020.
- 6.
The Norwegian Institute of Public Health. High coverage for the Childhood Immunisation Programme in Norway. https://www.fhi.no/en/news/2020/high-coverage-for-the-childhood-immunisation-programme-in-norway/ Accessed 12.5.2020.
- 7.
World Food Programme. COVID-19 and the five major threats it poses to global food security. https://insight.wfp.org/COVID-19-and-the-5-major-threats-it-poses-to-global-food-security-1c4da2ff6657 Accessed 3.4.2020.