A man in his early nineties with reduced general condition, fever and dry cough was admitted to a regional hospital just after the coronavirus pandemic reached Norway. His history did not reveal exposure to infection, and he had not been travelling. He was therefore not isolated on admission. His clinical history shows a serious course in an elderly patient. It also shows the potential for spreading of infection in today's globalised world.
An active man in his early nineties was sent to his general practitioner after falling at a shopping centre and injuring himself. He was accompanied by medical personnel to his GP's office, where he was prioritised for emergency assistance. The onset of symptoms was two days earlier (day 0), when he began to feel lethargic and lost his appetite during a bus trip with other pensioners. The following day (day 1) during choir practice he experienced a pronounced exacerbation with increasing dry cough, reduced general condition, unsteadiness and poor appetite. He later measured fever of up to 38.6oC. Urination and bowel movements were normal.
His GP had known the patient for many years. He had existing hypertension, three-vessel disease and had suffered inferior wall infarction. He had GOLD Stage 2 chronic obstructive pulmonary disease and benign prostatic hyperplasia. Because of a second-degree AV block type II with left bundle branch block, he had had a bicameral pacemaker implanted nine months before the incident in question.
An echocardiogram carried out in this connection showed good left ventricular function without significant valve defects. The patient went to regular check-ups with his GP and took his medication as prescribed. He had stopped smoking forty years previously and had low alcohol consumption. Despite severe chronic disorders, the patient was normally entirely self-sufficient and ambulant and had no need for walking aids. He drove a car and had completed a driver's licence assessment with a good cognitive score. He was active as a volunteer in several spheres of activity.
On day 2 of his illness his GP found him in reduced general condition, with shortness of breath, tachycardia and warm skin. His blood pressure was 120/70 mm Hg, his pulse regular at 100 beats/minute on a beta-blocker and his forehead temperature was 36.5oC. A clinical examination found normal conditions in his mouth, throat, heart, lungs, lymph node areas and skin. The patient had no neurological deficits, neck stiffness or petechiae, and was lucid and orientated with regard to time, place and situation. Blood tests showed haemoglobin 15 g/dl (13.4–17.0), leukocytes 10.8 ∙ 109/l (3.5–10), lymphocytes 3.7 ∙ 109/l (1.1–3.3) and C-reactive protein (CRP) 136 mg/l (0–4).
Because of his respiratory tract symptoms and severely reduced general condition, his GP suspected influenza or bacterial pneumonia.
The patient was transferred by ambulance to the regional hospital. In Acute Admissions he had slightly reduced general condition, a productive cough, fever of 39.4oC in the ear, blood pressure 145/70 mm Hg, regular pulse 82 beats/minute on a beta-blocker and respiratory rate 28 per minute with oxygen saturation 95 % on the pulse oximeter. Auscultation of the heart revealed normal findings. Crackles were heard basally over the lungs bilaterally.
There was no swelling of the lower extremities. Arterial blood gases in atmospheric air showed pH 7.45 (7.35–7.44), pCO2 4.1 kPa (4.7–6.0), pO2 9.4 kPa (10.0–14.0), oxygen saturation 96 % and base excess −3.5 mmol/l (−3–3). His SOFA score was 3 of 24 points (reduced pO2/FiO2 ratio, low platelet count and elevated creatinine), qSOFA score 1 of 3 points (increased respiratory rate) and SIRS score 2 of 4 points (fever and increased respiratory rate).
Blood tests on admission showed haemoglobin 14.6 g/dl (13.4–17.0), platelets 119 ∙ 109/l (145–390), leukocytes 8.2 ∙ 109/l (3.5–10), lymphocytes 0.9 ∙ 109/l (1.1–3.3), erythrocyte sedimentation rate 17 mm (1–12), CRP 159 mg/l (0–4), bilirubin 9 µmol/l (5–25), creatinine 124 µmol/l (60–105), INR 1.2 (0.8–1.2), activated partial thromboplastin time (APTT) 32 s (30–42), fibrinogen 4.3 g/l (2.0–4.0) and D-dimer 1.07 mg/l (age-adjusted < 0.80).
Possible differential diagnoses that were considered were bacterial and viral pneumonia, pulmonary embolism, acute exacerbation of chronic obstructive pulmonary disease, acute heart failure and renal failure. Because of his fever, normal leukocyte count, cough and slightly reduced general condition, influenza was suspected first. His elevated creatinine level was attributed to reduced fluid intake. COVID-19 was not regarded as a probable diagnosis as the patient had not been abroad and had only spent time in the local community. At the time, there were not yet any known cases of infection within Norway.
Table 1
Measurements and blood tests during the course of the illness from day 2 to day 8. No blood tests were taken or vital signs measured on day 9, the day the patient died.
| Reference range |
Day 2 |
Day 3 |
Day 4 |
Day 5 |
Day 6 |
Day 7 |
Day 8 |
|
|---|---|---|---|---|---|---|---|---|
| Max. temperature (°C) |
39.4 |
39.2 |
39.1 |
39.3 |
37.6 |
37.8 |
36.3 |
|
| Minimum-maximum oxygen saturation (%) |
94–99 |
93–95 |
91–95 |
91–95 |
85–90 |
76–88 |
78–88 |
60–78 |
| pO2 (kPa) |
10–14 |
9.4 |
6.8 |
5.5 |
||||
| CRP (mg/l) |
0–4 |
159 |
131 |
124 |
169 |
251 |
||
| Leukocytes (∙ 109/l |
3.5–10 |
8.2 |
4.8 |
5.1 |
4.7 |
7.9 |
||
| Lymphocytes, ∙ 109/l |
1.1–3.3 |
0.9 |
1.0 |
1.1 |
1.1 |
0.7 |
||
| Platelets (∙ 109/l) |
145–390 |
119 |
97 |
92 |
100 |
145 |
||
| Creatinine (µmol/l) |
60–105 |
124 |
108 |
105 |
94 |
89 |
||
| Alanine aminotransferase (U/ml) |
10–70 |
24 |
31 |
74 |
The rapid tests for influenza and RS virus performed in Acute Admissions were negative, and droplet precautions were therefore discontinued. Chest X-ray showed no lung changes. The patient did not show signs of sepsis. On suspicion of bacterial pneumonia, the patient was hospitalised and given empirical treatment with intravenous benzylpenicillin.
Cultured nasopharyngeal specimens showed only growth of normal flora, and PCR for respiratory tract infection agents was negative for influenza, metapneumovirus, RS virus, Chlamydia pneumoniae and Mycoplasma pneumoniae. Repeated blood cultures showed no bacterial growth after five days of incubation.
The patient felt better and wanted to go home on days 3 and 4 of his illness, but he still had a fever. On day 5 he had a temperature of 39.3 °C, respiratory rate 20 per minute with oxygen saturation 90 % in atmospheric air and no biochemical response to antibiotic therapy.
Bacterial pneumonia with penicillin-resistant bacteria was considered as a possible explanation for his failure to respond to empirical treatment. Persistent fever, respiratory tract symptoms and elevated D-dimer could possibly also be explained by an undetected pulmonary embolism or viral pneumonia.
After discussion with the duty microbiologist, extended respiratory tract PCR was ordered on the nasopharyngeal specimen taken at the time of admission (day 2) and pulmonary CT angiography was ordered for further study of the lungs. The CT images showed no signs of pulmonary embolism, but in all lung sections there were areas of ground-glass opacities and reticular changes known as "crazy paving" (Fig. 1).
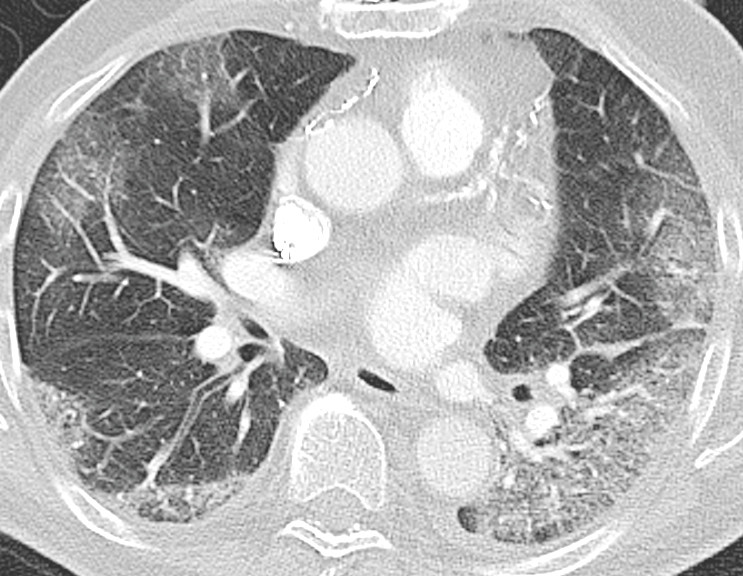
In light of the symptoms and the patient's age, the duty microbiologist elected to assay for SARS-CoV-2, in addition to four types of parainfluenza virus, rhinovirus, enterovirus D68 and adenovirus.
Despite the lack of known exposure to SARS-CoV-2 infection, the patient had symptoms that could be consistent with COVID-19. He was also in the high-risk group with a poorer prognosis if he did have COVID-19 (1).
On day 5 the patient was found to have SARS-CoV-2, and the results were immediately conveyed to clinicians. Infection control measures were initiated immediately, with intensified droplet precautions. To ensure against specimen substitution, the SARS-CoV-2-PCR was repeated, still with positive findings. Benzylpenicillin therapy was continued in view of the possibility of a bacterial superinfection.
On day 6 his creatinine level fell to 94 µmol/l after rehydration, CRP rose to 169 mg/l, with normal leukocytes of 4.7 ∙ 109/l and normalised lymphocytes of 1.1 ∙ 109/l. His oxygen saturation fell to 83 % in atmospheric air. His oxygen requirement increased to 8 l/minute to maintain oxygen saturation of 87 % with a respiratory rate of 30 per minute. Arterial blood gases showed pH 7.46, pCO2 4.0 kPa, pO2 6.8 kPa, base excess −2.9 and oxygen saturation 89 %. In addition he received nebuliser therapy with ipratropium and salbutamol. Antibiotics were changed from benzylpenicillin to cefotaxime as protection against Staphylococcus aureus and gram negative bacteria.
After broad multidisciplinary discussion, and in consultation with his family, it was decided that in view of the patient's advanced age, pronounced comorbidity and very rapid disease progression, ventilator treatment should not be offered.
On day 7 the patient appeared a little confused. He had moderate cough and became short of breath with the slightest exertion. His respiratory rate increased to 36 per minute, and his oxygen saturation fell to 78 % despite an oxygen supply of 15 l/minute with a mask with reservoir bag. Arterial blood tests showed increasing respiratory failure with hypoxia without hypercapnia even with pronounced oxygen support, and he then developed uncompensated heart failure. His kidney function remained normal. His lymphocyte count fell again to 0.7 ∙ 109/l, and his CRP rose to 251 mg/l.
Chest X-rays showed no convincing signs of congestion, but increasing patchy opacities with increasing consolidation which indicated disease progression (Fig. 2). In the course of the evening on day 7 his breathing became steadily more laboured, with a respiratory rate of over 40 per minute and oxygen saturation down towards 60 % despite a high oxygen supply by mask with reservoir bag.
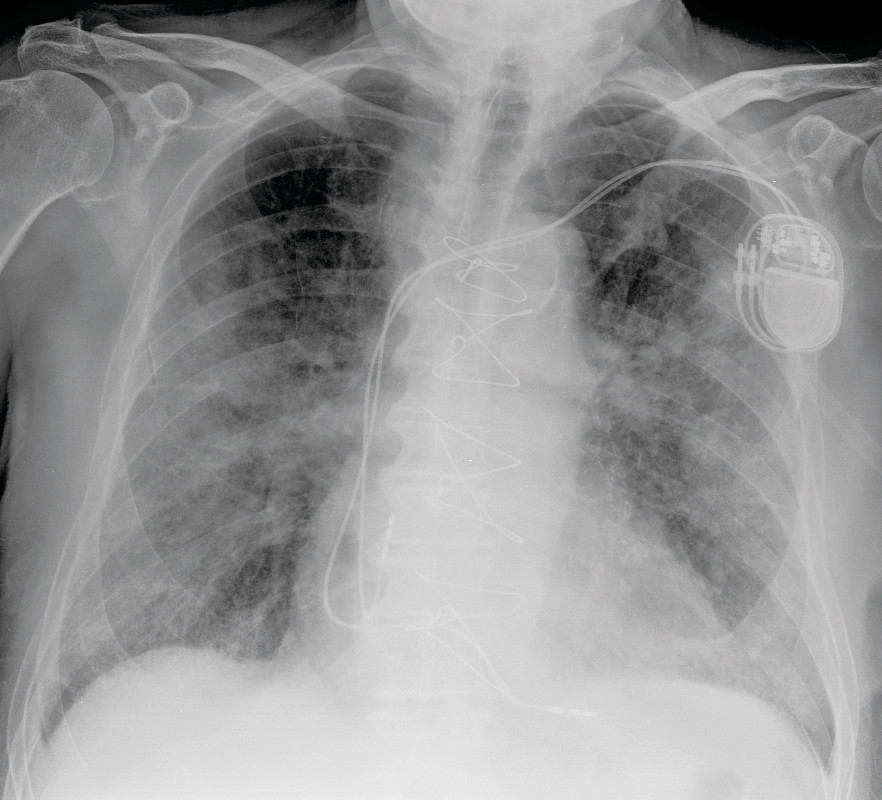
The patient now had increasing radiological pulmonary changes, a SOFA (sepsis-related organ failure assessment) score of 5 points and exacerbated respiratory failure. After a multidisciplinary assessment, the focus was changed from active life-saving treatment to palliative treatment.
Antibiotic therapy was discontinued. The patient was given glycopyrronium bromide and morphine, which were effective in achieving calmer respiration and less distress. On day 8 the patient was in the terminal phase. He died nine days after the onset of symptoms (Fig. 3).
Discussion
The coronaviridae are a family of encapsulated, single-stranded RNA viruses with capsules that are covered in spikes. Electron microscopic images are reminiscent of the corona around the sun. Coronaviruses are found in mammals and birds. As a rule, the infection in humans only causes mild respiratory tract infection and cold symptoms. The exceptions are Middle East respiratory syndrome coronavirus (MERS-CoV), severe acute respiratory syndrome-associated coronavirus (SARS-CoV) and the current SARS-CoV-2, which are human pathogenic coronaviruses which can cause serious pulmonary affection. SARS-CoV-2 causes the disease COVID-19 (corona virus infectious disease 2019), which has a very varied symptomatology (2)−(5). The patient had the most common symptoms of COVID-19: fever, cough, shortness of breath and reduced general condition.
SARS-CoV-2 is primarily detected in respiratory tract specimens, but also in other specimen material, by means of real-time reverse transcription PCR (rRT-PCR). PCR analysis according to the Berlin protocol detects the E-gene of the virus. Sensitivity is 95 %, with a detection limit as low as 5 copies/µl (6). The E-gene is found in beta-coronaviruses, which include SARS-CoV-2, SARS-CoV and other SARS-like viruses in animals. As other SARS-like viruses are not circulating among humans now, it could be concluded that the patient in question must have been infected with SARS-CoV-2.
The most common symptoms of COVID-19 reported in case studies are fever (83–98 %), cough (59–82 %), reduced general condition (38–69 %), shortness of breath (18–55 %) and poor appetite (40 %). It is worth noting that a relatively small proportion had a sore throat (5–17 %) and runny nose (4–5 %) (2–5). There may additionally be symptoms from multiple organ systems, depending on which organs are affected.
The feared complication is acute respiratory distress syndrome, ARDS, which is non-cardiogenic pulmonary oedema and diffuse pulmonary inflammation, reported in 15–29 % of patients in case studies (3–5). Our patient probably developed this condition in the course of a few days, with development of bilateral lung opacities and arterial blood gas analysis yielding a reduced pO2/FiO2 ratio. Respiratory failure dominated the clinical picture throughout the course of his illness, with only a minor degree of other organ affection.
In a retrospective case study with 52 critically ill adult patients there was high mortality, particularly among elderly patients (> 65 years) with comorbidity and acute respiratory distress syndrome (7). After 28 days, 61.5 % of the patients in the study had died, and the median duration from intensive care treatment to death was seven days. Those who died developed acute respiratory distress syndrome (81 %) more frequently than those who survived (45 %). It should be noted that only two of the patients in the study were ≥ 80 years old, and both died. It was regarded as unethical to treat our patient with measures as invasive as respirator treatment. We believe it would have drawn out the dying process rather than improving the prospects for survival.
Laboratory findings reflect organ affection. The most common haematological findings in hospitalised patients with pneumonia are leukopenia, lymphopenia and leukocytosis. Other abnormal findings are neutrophilia, thrombocytopenia and anaemia. The most common abnormal coagulation test results are elevated D-dimer and longer prothrombin time. The patient had moderate thrombocytopenia and lymphopenia, which are described in case studies with a more serious course (8).
In a retrospective cohort study from Wuhan in China, high age, high D-dimer and high SOFA score were particularly associated with an increased probability of a fatal outcome (1). Lymphopenia on admission was also a risk factor for a serious course. Our patient had a high age and high D-dimer, but only a slightly elevated SOFA score on arrival at Acute Admissions. His SOFA score increased from 3 to 5 points from day 2 to day 7.
Elevated alanine aminotransferase (ALT) can be seen in seriously ill patients (5). A letter to The Lancet speculates over whether secondary haemophagocystic lymphohistocytosis (sHLH) may be triggered by COVID-19 (9). The cardinal symptoms of this condition are persistent fever, cytopenia and elevated ferritin. On day 7 the patient had slightly elevated ALT at 74 U/L (10−70), a fall in lymphocytes to 0.7 ∙ 109/l, elevated lactate dehydrogenase at 566 U/L (115−255) and elevated ferritin at 1 175 µg/l (30−400). A retrospective analysis of 150 patients with COVID-19 found elevated ferritin and interleukin-6 (IL-6) to be associated with increased mortality, which may indicate that mortality is due to virus-driven hyperinflammation (10).
It is worth securing respiratory tract specimens and blood cultures in order to exclude other causes of respiratory tract infection prior to the start of empirical antibiotics, particularly in patients with an atypical epidemiological medical history. There was no bacterial growth in respiratory tract specimens and blood cultures from the patient.
While the first X-ray image of the lungs on day 2 showed no opacities, the patient had ground-glass opacities in all sections of the lungs on the pulmonary CT angiogram performed on day 5. Ground-glass opacities can be seen with several conditions and a characteristic feature is that the changes do not obscure the lung structures. Such opacities, with or without consolidation, are described in patients with COVID-19 (11–14). With consolidation, the normal anatomy of the lung is obscured by the opacities. CT must not be used as a diagnostic or screening tool, but may be relevant in cases where there is suspicion of complications that have not been clarified by chest X-ray (15). CT may be more sensitive than rRT-PCR in early stages of the disease and in an epidemic area, but is non-specific, and other interstitial pneumonias may yield similar findings (16–17). For these patients, CT will be a very limited resource, because time-consuming disinfection of the laboratory has to be done after each examination. No new CT was taken during the course of our patient's illness, but on day 7 a chest X-ray was taken that showed patchy opacities with increasing consolidation (Fig. 2). This indicated disease progression.
The patient was among the first to be hospitalised in Norway with COVID-19, and among the first with a detected infection where there was no clear connection with travel abroad or definite contact with an infected person. Since COVID-19 was not initially suspected, he remained in the ward without adequate infection control measures. Recommendations for testing for SARS-CoV-2 have been changing almost daily. At the time of his admission there was not yet any indication for this testing. His GP and two secretaries at the GP's office and some 30 hospital staff were exposed to infection, and were therefore quarantined for 14 days. Shortly after the patient was found to have COVID-19, the GP's office and the hospital changed their procedures for both testing and infection prevention measures.
Infection tracing started immediately, with notification to the hospital's Department of Infectious Disease Control, the patient's family and his GP, and infection control authorities in the borough and municipality. Further extensive infection tracing in the patient's various spheres of activity in the local community was dealt with by the infection control authority in the patient's borough. This patient history is a good illustration of both the need for early appropriate infection control measures and the consequences of late implementation.
The patient's family have consented to the publication of the article.
Our thanks go to Nina Merethe Clausen, chief bioengineer at the Department of Microbiology at Oslo University Hospital, Ullevål. She set up the PCR analysis of SARS-CoV-2 and also performed most of the analyses.
The article has been peer-reviewed.
- 1.
Zhou F, Yu T, Du R et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: S0140-6736(20)30566-3. [PubMed][CrossRef]
- 2.
Guan WJ, Ni ZY, Hu Y et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: NEJMoa2002032. [PubMed][CrossRef]
- 3.
Huang C, Wang Y, Li X et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395: 497–506. [PubMed][CrossRef]
- 4.
Chen N, Zhou M, Dong X et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020; 395: 507–13. [PubMed][CrossRef]
- 5.
Wang D, Hu B, Hu C et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020; 323: 1061–9. [PubMed][CrossRef]
- 6.
Corman VM, Landt O, Kaiser M et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill 2020; 25: 2000045. [PubMed][CrossRef]
- 7.
Yang X, Yu Y, Xu J et al. Clinical course and outcomes of critically ill patients with SARS-CoV2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet 2020; 395. doi: 10.1016/S2213-2600(20)30079-5. [CrossRef]
- 8.
Qin C, Zhou L, Hu Z et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis 2020; 70: ciaa248. [PubMed][CrossRef]
- 9.
Mehta P, McAuley D, Brown M et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395. doi: 10.1016/S0140-6736(20)30628-0. [CrossRef]
- 10.
Ruan Q, Yang K, Wang W et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med 2020; 46. doi: 10.1007/s00134-020-05991-x. [PubMed][CrossRef]
- 11.
Shi H, Han X, Jiang N et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis 2020; 395: S1473-3099(20)30086-4. [PubMed][CrossRef]
- 12.
Bernheim A, Mei X, Huang M et al. Chest CT findings in coronavirus disease-19 (COVID-19): Relationship to duration of infection. Radiology 2020; 295: 200463. [PubMed][CrossRef]
- 13.
Ng M, Lee E, Yang J et al. Imaging Profile of the COVID-19 Infection: Radiologic Findings and Literature Review. Radiology Cardiothoracic Imaging 2020; 2: e200034. [CrossRef]
- 14.
Zhao W, Zhong Z, Xie X et al. Relation between chest CT findings and clinical conditions of coronavirus disease (Covid-19) pneumonia: a multicenter study. AJR Am J Roentgenol 2020; 214: 1–6. [PubMed][CrossRef]
- 15.
ACR recommendations for the use of chest radiography and CT for suspected COVID-19 cases. Imagining Technology News. https://www.itnonline.com/content/acr-recommendations-use-chest-radiography-and-ct-suspected-covid-19-cases Accessed 14.3.2020.
- 16.
Zu ZY, Jiang MD, Xu PP et al. Coronavirus disease 2019 (COVID-19): A perspective from China. Radiology 2020; 295: 200490. [PubMed][CrossRef]
- 17.
Ai T, Yang Z, Hou H et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology 2020; 295: 200642. [PubMed][CrossRef]