Main findings
All patients who completed one year in the standardised care pathway for patients with gout achieved the treatment goal in the form of serum urate below 300 μmol/l for tophaceous gout or below 360 μmol/l for non-tophaceous gout.
The probability of achieving the treatment goal at six months was 83 per cent.
Allopurinol was well tolerated and had good urate-lowering effect, with a fall in serum urate concentration from an average of 446 µmol/l to within the target range.
Gout is a chronic, inflammatory disease of the joints that occurs when serum urate crystals are deposited in joints and soft tissue after a preliminary period of elevated serum levels of urate. The most common presentation is an acute, self-limiting inflammatory arthritis flare, usually monoarthritis of the foot or ankle of one to two weeks' duration. The condition can recur if untreated, affect more joints, and in some cases develop into a chronic form of arthritis with tophi, joint destruction and reduced function (1, 2).
Epidemiological studies have shown an increasing prevalence of gout in Europe, North America, China and Southeast Asia (3). In all countries, the prevalence is relatively higher among men, with a ratio of 3–4: 1 (2). The prevalence figure in the Nordic countries is estimated at around 1 % for men, and far lower for women, with an upward trend in the last 25 years (4). In the USA, hospitalisations due to gout doubled in the period 1993–2011, with an increase in annual incidence from 4.4 to 8.8 per 100 000 adults (5).
Drug treatment for gout consists of two main components: treatment of acute flares and maintenance treatment. The fundamental principle of long-term treatment is to reduce the serum urate concentration to below the threshold for crystallisation by inhibiting the enzyme xanthine oxidase with allopurinol or febuxostat. Serum urate below 360 µmol/l will normally lead to reduced frequency of flares and urate crystal precipitation (1), but in tophaceous gout (characterised by solid masses of urate (tophi) in joints and soft tissues, and progressive joint damage), it will be necessary to lower the concentration below 300 µmol/l (6, 7).
A UK study showed that only one-third of patients with gout had been prescribed urate-lowering therapy in the period 1997–2012 (8). Compliance with urate-lowering therapy has also proven to be low, from 10–46 % in different datasets based on electronic prescribing (9). Systematic follow-up of gout has previously been tested in England, where good initial patient education and clear treatment goals gave satisfactory therapeutic results and medication compliance assessed after five years (10). Many years of experience with suboptimal maintenance treatment for gout formed the background for this project, the purpose of which was to ensure adequate serum urate-lowering therapy through structured follow-up.
Material and method
At Haukeland University Hospital we have established a standardised care pathway for patients with gout. Since March 2017, all patients at the Department of Rheumatology with certain or probable gout have been invited to participate in the standardised care pathway. The patients are referred to the department by their GP or other departments. The diagnosis is made by a rheumatologist before inclusion in the care pathway, and inclusion is independent of comorbidity. The only requirement is that the patient must be capable of following the programme. The aim of the treatment is reduction in the number of gout flares, with the objective of achieving a serum urate concentration below 360 µmol/l, or below 300 µmol/l for tophaceous disease.
The first care pathway visit is interdisciplinary. Nurses provide information about the follow-up, while doctors investigate how the disease has manifested itself to date, inform the patient about the disease and recommend treatment. A pharmacist notes use of medication and gives further information about the drug and its adverse effects.
The patients receive drug treatment in line with international guidelines (6, 7, 11). Untreated patients start with allopurinol 50–100 mg daily, and the dose is adjusted based on renal function and any adverse effects experienced previously. Allopurinol is then increased by 50–100 mg every four weeks until the treatment goal is achieved. If the patient is already using allopurinol, the dose is optimised according to the same principle. In cases of intolerance to allopurinol, febuxostat is used. The starting dose of febuxostat is 40 mg, and the maximum therapeutic dose is 120 mg daily. Colchicine is recommended as prophylaxis for acute flares in the first six to twelve months at a dose of 0.5 mg × 1–2 (lower in case of reduced renal function and intolerance).
The patients are monitored through monthly nurse-led consultations, face-to-face or by telephone. The nurses increase the urate-lowering therapy based on blood test results. When the target value for serum urate is achieved, monthly check-ups continue for three months, followed by a new doctor's assessment. Further follow-up is assessed on an individual basis. The intention is to transfer check-ups back to the primary health service after two years of follow-up.
Upon inclusion in the care pathway, all patients are asked for their consent to participate in a study that will assess the efficacy of standardised, close follow-up. In this article we use data from the care pathway to investigate the time to achievement of the treatment goal for serum urate in this patient group. The study design is an intervention study without a control group. At the time of analysis, 103 patients were included in the care pathway. All consented to inclusion in the study. Patient characteristics are presented in Table 1.
Table 1
Patient characteristics at inclusion for 103 patients1 (average (min.– max.) or percentage)
| Variable |
Value |
|
|---|---|---|
| Age, sex and anthropometry |
||
| Age (years) |
63 (27–89) |
|
| Men (%) |
90 |
|
| BMI (kg/m2) |
28.5 (20.7–41.5) |
|
| Comorbidity (%) |
||
| Diabetes |
17 |
|
| Renal disease |
43 |
|
| Cardiovascular disease |
28 |
|
| Exercise (%) |
||
| Daily/weekly |
57 |
|
| Rarely/never |
43 |
|
| Smoking (%) |
||
| Never smoked |
40 |
|
| Former smoker |
47 |
|
| Smoker |
11 |
|
| Alcohol (%) |
||
| ≥ 4 days/week |
6.2 |
|
| 1–3 days/week |
34 |
|
| Rarely |
48 |
|
| Never |
12 |
|
| Characteristics of the gout |
||
| Duration of disease (years) |
9.3 (0–36)2 |
|
| Flares per year |
4.6 (0–25) |
|
| Sodium urate (µmol/l) |
446 (144–751) |
|
| Allopurinol at inclusion (%) |
55 |
|
1Information on BMI (body mass index) was available for 89 patients, exercise for 92, smoking status for 98 and alcohol anamnesis for 96 patients. Data for the remaining variables were available for all 193 patients.
2Duration 0 years indicates new-onset gout.
The project has been approved by the regional committee for medical and health research ethics (REC North Norway, 2017/801).
Statistical analyses
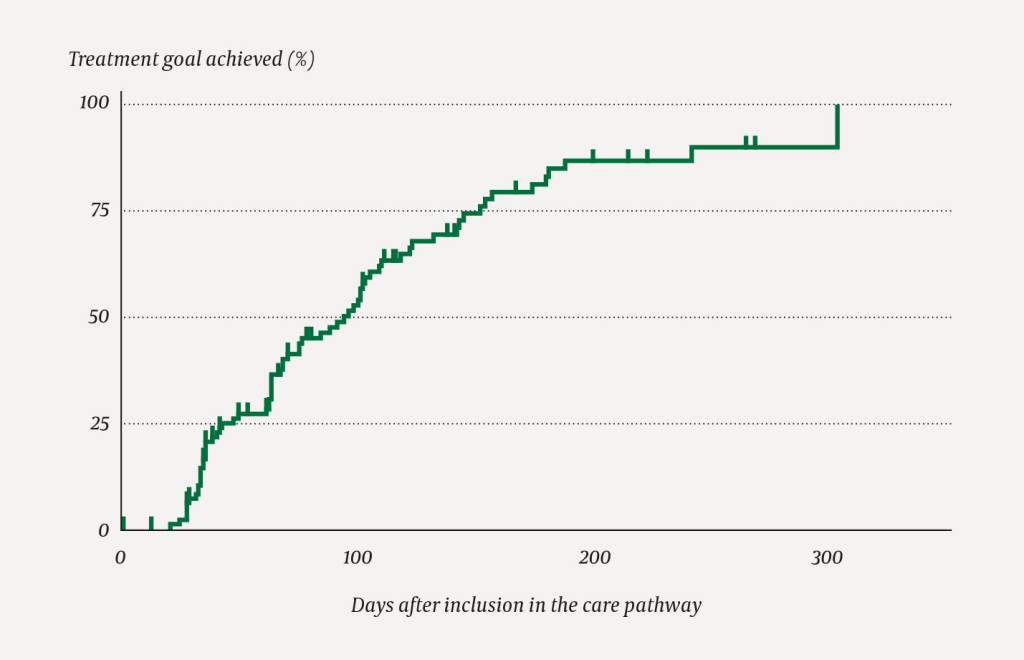
Since the follow-up time varied for the different patients, survival analyses in the form of Kaplan-Meier analysis and Cox regression analysis were used to find the time period that elapsed before the treatment goal was achieved, and the degree to which different factors affected the likelihood of achieving the goal. Achievement of the treatment goal constituted the endpoint in these analyses. For patients who achieved the treatment goal, the observation time lasted until the first date on which the treatment goal was achieved. Patients who failed to achieve the treatment goal in the course of the study were censored at the time of their last registered check-up, and the observation time was calculated up to that date. For patients who died or withdrew from the study without having achieved the treatment goal, the observation time was calculated up to the date of death or the time of withdrawal. The cumulative proportion of patients who achieved the treatment goal three and six months after inclusion in the care pathway was calculated with the aid of Kaplan-Meier analysis (see Figure 1). Significance testing was performed using the log-rank test. The analyses were performed in SPSS, version 24.
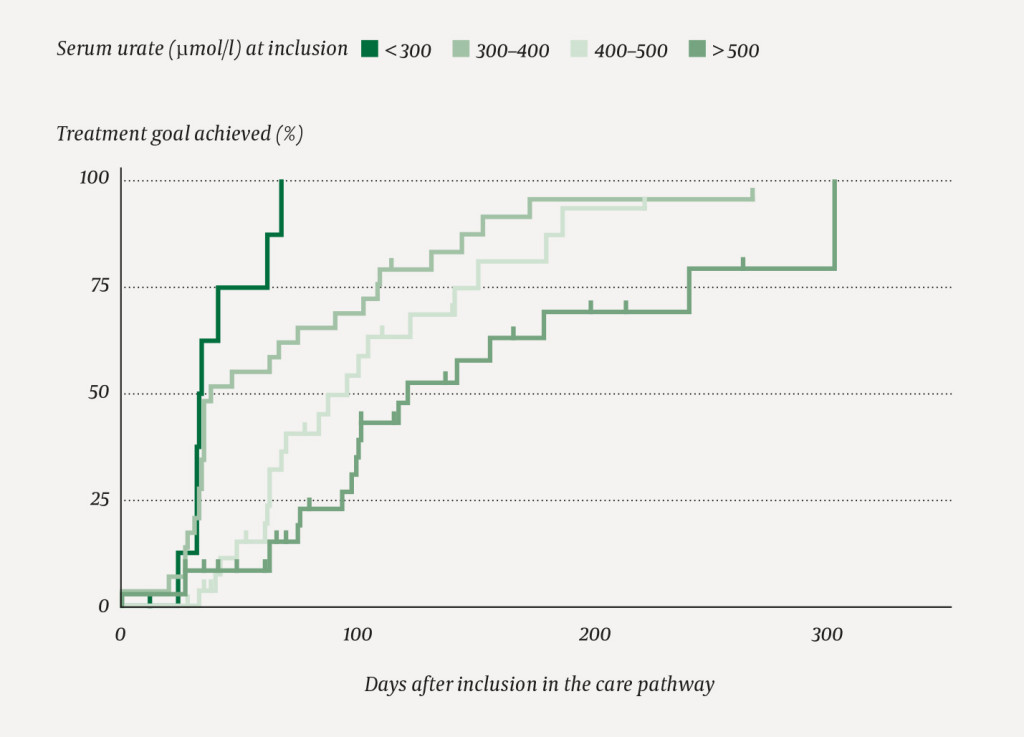
We performed unadjusted Cox regression analyses in order to assess how different factors affected achievement of the treatment goal. Based on findings in previous studies and clinical relevance, we analysed the following variables: sex, age, alcohol consumption (self-reported at inclusion, categorised: ≥ 4 days/week, 1–3 days/week, rarely or never), smoking status (self-reported at inclusion: never smoked, former or current smoker), serum urate (at inclusion: < 300, 300–400, 400–500 or > 500 µmol/l), body mass index (BMI, patient-reported height and weight at inclusion) and reduced renal function (defined as serum creatinine > 100 µmol/l at inclusion). Only serum urate had a significant effect on the outcome and was included, together with sex and age, in the adjusted Cox regression analysis (Table 2).
Table 2
Chance of achieving the treatment goal related to various factors with a possible effect on the outcome, calculated using Cox regression analysis. The analysis is adjusted for age and sex. Values marked in bold are significant.
| n1 |
uHR2 |
HR2 |
95 % CI3 |
||
|---|---|---|---|---|---|
| Serum urate (µmol/l) |
|||||
| < 300 |
9 |
10.8 |
11.0 |
4.4–27.3 |
|
| 300–400 |
29 |
3.0 |
3.00 |
1.6–5.7 |
|
| 400–500 |
29 |
1.8 |
1.8 |
0.9–3.5 |
|
| > 500 |
36 |
1 |
1 |
||
1Data for one or more of the variables are missing for 3 patients. 100 patients were therefore included in the analysis.
2uHR and HR are the unadjusted and adjusted hazard ratio, respectively.
395 % confidence interval (CI) for adjusted HR.
Results
At the time of analysis, the gout project had been in progress for 18 months. Of a total of 103 included patients, 21 (20.4 %) had reached their one-year check-up. Altogether 95 patients were still being followed up in the care pathway, while 8 (7.7 %) had left the project due to death, termination of the follow-up by their own choice, or exclusion by their doctor.
Altogether 13 patients reported previous use of allopurinol, with adverse effects or advice from their GP as reasons for its discontinuation. In cases of mild or uncertain adverse effects, the drug was reintroduced at a low starting dose (50 mg) to 10 of these patients.
The maximum dose of allopurinol for patients who had achieved the analytical treatment goal was an average of 333 mg (min.–max. 100–700 mg). Four out of five patients taking febuxostat achieved the target value with a dose of 40 mg daily. For all five of these patients, mild and somewhat non-specific adverse effects of allopurinol formed the basis for the use of febuxostat, tried either before or after inclusion in the care pathway.
No serious adverse effects of allopurinol were observed. Among mild adverse effects, gastrointestinal effects of colchicine were frequent (14 patients). One case of colchicine-induced bone marrow suppression was identified and categorised as severe. Self-discontinuation of allopurinol or another drug was rare.
A total of 71 patients had achieved the analytical treatment goal for serum urate by the end of the study. Of these, 66 received treatment with allopurinol alone; the remainder received febuxostat. Median time to achievement of the target value for serum urate was 69 days (interquartile range 81). Survival analyses showed that the probability of achieving the analytical goal at three months was 49 %, and at six months (181 days) it was 83 % (Figure 1). Sex, age, alcohol consumption, smoking, body mass index and renal function had no significant effect on the time to achievement of the analytical goal. However, the serum urate value correlated with the chance of achieving the treatment goal (Table 2). As expected, a high baseline value for serum urate resulted in the need for a higher allopurinol dose and a longer escalation phase (Figure 2).
Of a total of 71 patients who achieved the analytical goal for serum urate, 50 (70 %) were free of flares in the first three months after achieving the goal. The remainder generally reported one or two flare-ups in the same period. These also involved more fleeting symptoms reported by the patient, with no detectable arthritis.
Discussion
Use of a structured care pathway for patients with gout provided excellent results. Only four patients withdrew, and all the patients who completed one year of the care pathway achieved the treatment goal. Since the patients who were offered the care pathway had been in contact with the specialist health service, we believe that they represent those who are worst affected. More than four acute flares per year, as well as an average disease duration of more than nine years also point in this direction. The 83 % probability of achieving the treatment goal within six months indicates that the programme was effective.
Previous studies have shown a high degree of self-discontinuation of medication for this disease (10). One of the reasons for self-discontinuation is most likely an increase in the frequency of flares in the initial phase of the treatment (1), which the patient may have interpreted as treatment failure. However, this does not point to treatment failure, but indicates the mobilisation of stored uric acid by allopurinol (6). Colchicine for prophylaxis of acute flares in the first six months during dose escalation with allopurinol is essential to counteract this effect; it may have resulted in few observed flares in the study period, and to the fact that the patients maintained the recommended treatment. Long periods without flares during treatment with allopurinol may be misinterpreted as regression of the disease and can also lead to self-discontinuation. Lifelong reduction of serum urate concentration below 360 µmol/l is recommended (6).
It took more than three months for the majority of the patients to achieve the treatment goal. Urate-lowering therapy is therefore time-consuming, and it is important that treatment continues until the point when the target value for serum urate is reached. Sufficient patient information with regard to these conditions, as well as close follow-up by nurses during the escalation phase, were important factors in the care pathway that we believe may have contributed to good results.
Few patients experienced adverse effects, and we saw no serious reactions to allopurinol. The gastrointestinal effects of colchicine are well known, and studies show that approximately 10 % of patients experience these effects when it is used as prophylaxis (12), which also corresponds with our data. One case of serious bone marrow toxicity triggered by colchicine was observed, related to accumulation in chronic renal failure. Use of colchicine in patients with renal failure should be restricted to the utmost extent (12).
In the period 2010–17, a total of 640 patients were admitted to Haukeland University Hospital with gout as the main diagnosis. Median hospitalisation time was three days. We believe that many of these hospitalisations could have been avoided with the aid of simple basic treatment and better knowledge about gout on the part of the individual patient and doctor. It remains to be seen whether the care pathway may have an impact on hospitalisation figures.
Strengths and weaknesses
A possible source of error that may have contributed to the good results is the fact that the participants were particularly motivated due to serious, prolonged disease. We believe that use of a similar treatment strategy among more healthy patients with disease of a shorter duration will also yield good results, as based on lower initial serum urate levels, a shorter adjustment time and lower target dose for allopurinol can be expected. Because we did not undertake a comparative study, we cannot say with certainty that the results are superior in relation to normal practice, but systematisation and structuring of treatment has previously been shown to significantly improve medication compliance in cases of gout (10). A study that randomised to either 'normal practice' or inclusion in a structured care pathway might have ascertained whether the pathway yielded better results. There were several reasons for our choice not to include a control group. The efficacy of the medical treatment in the care pathway is well documented (6, 7, 11). Nor was the purpose of the study to compare different drug treatment regimens, but to facilitate correct use of the recommended treatment. The fact that 'normal practice' frequently entails treatment in the primary health service would also have rendered it difficult to follow up the patients in this group.
A disadvantage of the programme of check-ups in the follow-up process is the extensive nature of the initial phase. Telephone-based check-ups and blood sampling by GPs simplify the follow-up. In our opinion, the fact that we included an unselected group of patients with regard to comorbidity results in good external validity, so that the model can be used for the vast majority of patients with gout, whether they are treated in the primary health service or in hospital.
The article has been peer reviewed.
- 2.
Kuo CF, Grainge MJ, Zhang W et al. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol 2015; 11: 649–62. [PubMed][CrossRef]
- 3.
Pascart T, Lioté F. Gout: state of the art after a decade of developments. Rheumatology (Oxford) 2019; 58: 27–44. [PubMed]
- 4.
Kiadaliri AA, Uhlig T, Englund M. Burden of gout in the Nordic region, 1990-2015: findings from the Global Burden of Disease Study 2015. Scand J Rheumatol 2018; 47: 410–7. [PubMed][CrossRef]
- 5.
Lim SY, Lu N, Oza A et al. Trends in gout and rheumatoid arthritis hospitalizations in the United States, 1993–2011. JAMA 2016; 315: 2345–7. [PubMed][CrossRef]
- 6.
Richette P, Doherty M, Pascual E et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis 2017; 76: 29–42. [PubMed][CrossRef]
- 7.
Khanna D, Fitzgerald JD, Khanna PP et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res (Hoboken) 2012; 64: 1431–46. [PubMed][CrossRef]
- 8.
Kuo CF, Grainge MJ, Mallen C et al. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann Rheum Dis 2015; 74: 661–7. [PubMed][CrossRef]
- 9.
De Vera MA, Marcotte G, Rai S et al. Medication adherence in gout: a systematic review. Arthritis Care Res (Hoboken) 2014; 66: 1551–9. [PubMed][CrossRef]
- 10.
Abhishek A, Jenkins W, La-Crette J et al. Long-term persistence and adherence on urate-lowering treatment can be maintained in primary care-5-year follow-up of a proof-of-concept study. Rheumatology (Oxford) 2017; 56: 529–33. [PubMed][CrossRef]
- 11.
Khanna D, Khanna PP, Fitzgerald JD et al. 2012 American College of Rheumatology guidelines for management of gout. Part 2: therapy and antiinflammatory prophylaxis of acute gouty arthritis. Arthritis Care Res (Hoboken) 2012; 64: 1447–61. [PubMed][CrossRef]
- 12.
Pascart T, Richette P. Colchicine in gout: an update. Curr Pharm Des 2018; 24: 684–9. [PubMed][CrossRef]