Main findings
At the time of referral to the Department of Oncology, 43 % of cancer patients aged ≥ 70 had had cardiovascular disease that was symptomatic or requiring intervention.
20 % of the patients had impaired renal function and 26 % took at least six drugs regularly.
The number of older people with cancer is increasing (1). At the same time, new treatment options that may also be suitable for older patients are constantly being developed. Individual differences in health status become more pronounced with age, and chronological age alone provides limited information about tolerance for cancer treatment and life expectancy. An international consensus recommends that older people with cancer undergo a geriatric assessment that systematically assesses comorbidity, use of medication, functional level, mobility, cognitive and emotional function, nutritional status and social network (2). The American Society of Medical Oncology presented similar recommendations in 2018, where the degree of evidence was regarded as high and the recommendations as strong (3).
The purpose of surveying frailty indicators is partly to detect the need for measures that should be implemented prior to and during cancer treatment. It also provides a basis for individual adaptation of the cancer-specific therapy. It is particularly important to identify patients who are frail, with a higher risk of complications and shorter life expectancy. Frailty is a clinical condition characterised by increased vulnerability when the patient is subjected to stressors such as chemotherapy and surgical interventions. The condition is due to a reduction in the body's homeostatic capacity. A frail patient is therefore prone to complications, hospitalisations and mortality (4). Patients are classified as frail if they have severe comorbidity, are dependent in basic activities of daily living, and/or suffer from dementia, malnutrition or severe depression. A systematic review from 2018 has shown that geriatric assessment of older cancer patients leads to treatment being altered in a median of 28 % of patients, usually in a less aggressive direction, to non-oncological interventions in a median of 72 % of patients, and a trend towards more patients managing to complete the treatment (5).
In this study, we have surveyed frailty indicators in a group of older patients with cancer.
Material and method
We have conducted a retrospective, records-based survey of all patients aged ≥ 70 who were initially assessed at the Department of Oncology, Drammen Hospital, in 2017. Patients who were referred after a previously completed course were also included. Drammen Hospital serves as a local hospital for over 160 000 people, and a regional hospital for cancer surgery for about 475 000. The main activities of the Department of Oncology target adults in the local hospital area requiring non-surgical treatment for solid tumour cancers.
Sex, year of birth and cancer diagnosis were recorded. Treatment intent was classified as curative, palliative (life extension and/or relief of symptoms) or follow-up (after completion of curative treatment). The number of regular medicines was defined as prescription drugs taken regularly prior to the initial consultation with the exception of nutritional/electrolyte supplements and laxatives. Performance status was classified according to the Eastern Cooperative Oncology Group (ECOG) scale from 0–4 (6): 0 = Normal, unlimited activity level, 1 = self-sufficient with a slightly reduced capacity for strenuous activity, 2 = self-sufficient but not capable of work / confined to a chair or bed < 50 % of the day, 3 = confined to a chair or bed > 50 % of the day / partly in need of care, 4 = completely reliant on care.
In cases where ECOG status was not entered in the initial record, the first author conducted a retrospective assessment if there was sufficient description of functioning in the records. Comorbidity was broken down into four categories to permit identification of relevant groups with an increased risk of complications arising from cancer treatment. The category of cardiovascular disease was assigned if a review of records yielded positive answers to whether the patient was receiving/had received treatment for at least one of the following conditions: coronary heart disease, heart failure, heart valve disease, heart rhythm disturbance, stroke/TIA or peripheral vascular disease, such as deep vein thrombosis or intermittent claudication. The category pulmonary disease was assigned for asthma and/or COPD. The use of antidiabetic drugs was categorised under diabetes. The category impaired renal function was assigned for estimated GFR < 60 ml/min based on the MDRD formula (7), which includes calculated body surface area according to Du Bois & Du Bois' height and weight formula (8). If height and weight were not available, body surface area was set at 1.73 m2.
Geriatric 8 (G-8) is the first screening tool that was developed specifically for older cancer patients with the aim of identifying patients who could benefit from a geriatric assessment (9). G-8 consists of eight questions about food intake, weight loss, mobility, neuropsychological problems (depression and dementia), body mass index, use of medication, the patient's own assessment of their health status and chronological age. The result points add up to a maximum of 17. An international consensus recommends this tool for selecting older patients with cancer for a geriatric assessment in the case of an overall score of < 15 (10). G-8 was developed in France and translated into Norwegian, but has not been validated in Norwegian. It takes about five minutes to fill in, and has a sensitivity of 85 % and a specificity of 64 % for detecting frailty (11). A low score has been shown to be a strong and consistent predictor of death in patients with cancer, irrespective of metastatic status and type of tumour (12). At the Department of Oncology, the form was used in 2017 to systematically identify frailty indicators in older cancer patients. The doctors in the department were encouraged to use the form for all new patients aged ≥ 70. The form was completed by the attending doctor as part of structured record-taking and scanned into the patient's records. In this review, we have recorded the G-8 responses where the form was used.
De-identified data were collected in the department's local quality register, which was established after consulting the Data Protection Official and stored according to the hospital's procedures for local quality registers. Publication of the results has been cleared with the Data Protection Official. Descriptive analyses were performed with the aid of SPSS statistics software.
Results
We identified 235 patients born in 1947 or earlier who had had an initial consultation at the Department of Oncology in 2017. They accounted for 48 % of the total number of initial consultations. The median age was 75 (range 60–93 years), and 130 (55 %) were men (Table 1). Treatment intent was curative for 66 patients (28 %), palliative for 160 (68 %) and follow-up after previous treatment with curative intent for 9 (4 %). Performance status was given or sufficiently described for 202 patients (86 %). Of these, 44 (22 %) had ECOG 0, 88 (44 %) ECOG 1, 49 (24 %) ECOG 2, 20 (10 %) ECOG 3 and 1 (0.4 %) ECOG 4. The description of the functioning of 33 of the patients (14 %) was not sufficient to allow assessment of their ECOG status.
Table 1
Distribution of sex, cancer diagnoses, treatment intent, performance status, medication used and comorbidity in patients born in 1947 or earlier who were assessed for the first time at the Department of Oncology, Drammen Hospital in the course of 2018 (N = 235), and for a sub-group of patients who were scored using the Geriatric 8 (G-8) form as part of structured record keeping. Number with rounded off percentage in parentheses unless otherwise stated.
| All, N = 235 (%) |
G-8 form completed, N = 144 (%) |
||
|---|---|---|---|
| Men |
130 (55) |
92 (64) |
|
| Age (years), median (range) |
75 (69–93) |
76 (69–93) |
|
| Type of cancer |
|||
| Colorectal |
43 (18) |
30 (21) |
|
| Lung |
41 (17) |
18 (13) |
|
| Breast |
38 (16) |
16 (11) |
|
| Prostate |
29 (12) |
27 (19) |
|
| Pancreatic |
18 (8) |
14 (10) |
|
| Lymphoma |
15 (6) |
8 (6) |
|
| Melanoma |
13 (6) |
8 (6) |
|
| Other |
38 (16) |
23 (16) |
|
| Treatment intent |
|||
| Palliative |
160 (68) |
106 (74) |
|
| Curative |
66 (28) |
34 (24) |
|
| Follow-up |
9 (4) |
4 (3) |
|
| ECOG status |
|||
| 0 |
44 (19) |
32 (22) |
|
| 1 |
88 (37) |
58 (40) |
|
| 2 |
49 (21) |
37 (26) |
|
| 3 |
20 (9) |
7 (5) |
|
| 4 |
1 (0) |
1 (1) |
|
| Not described |
33 (14) |
9 (6) |
|
| Number of regular drugs, median (range) |
4 (0–14) |
4 (0–14) |
|
| Comorbidity |
|||
| Cardiovascular disease |
101 (43) |
64 (44) |
|
| Impaired renal function |
47 (20) |
35 (24) |
|
| Pulmonary disease |
37 (16) |
19 (13) |
|
| Diabetes |
25 (11) |
20 (14) |
|
The median number of regular drugs was four (range 0–14). Eighteen patients took no drugs regularly (8 %). Six or more regular drugs were taken by 61 patients (26 %) (Fig. 1). Cardiovascular disease was found in 101 patients (43 %), impaired renal function in 47 (20 %), pulmonary disease in 37 (16 %) and diabetes in 25 (11 %). Eighty five patients (36 %) had none of the defined forms of comorbidity. The G-8 form was used for 144 patients (61 %). The median age of patients with a G-8 score was 76 (range 60–93), and 92 (64 %) were men (Table 1). Treatment intent was palliative for 74 %. Of the patients scored using G-8, 54 (38 %) reported a moderate to severe decrease in food intake in the previous three months, 50 (35 %) reported weight loss of > 3 kg, 29 (20 %) neuropsychological problems and 29 (20 %) considered their own health to be poorer than that of others of the same age. Of the 144 patients for whom G-8 was used, 100 (69 %) scored < 15 points (Table 2).
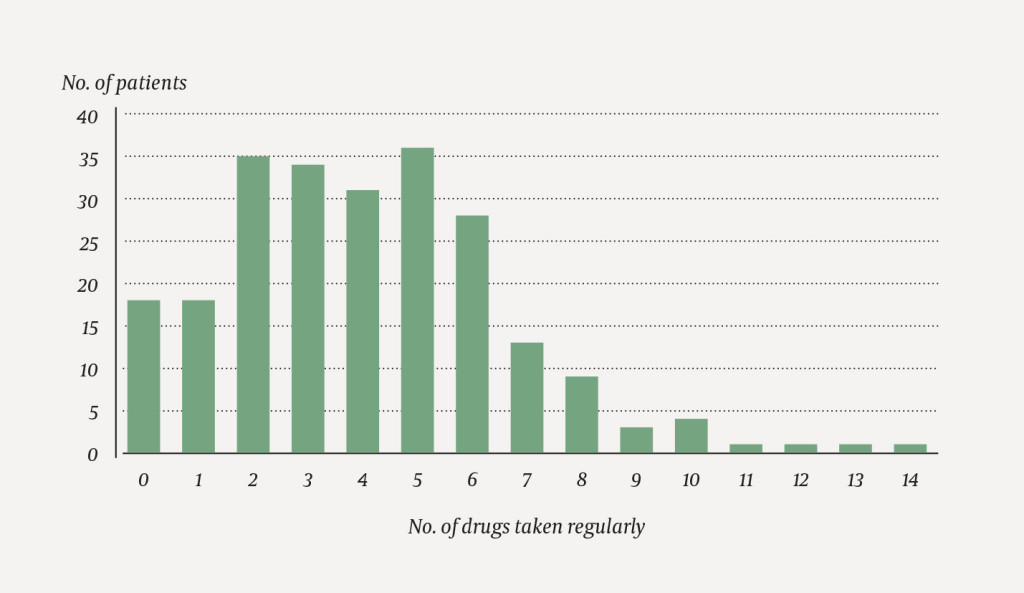
Table 2
Results for 144 patients born in 1947 or earlier where attending doctor filled in a Geriatric 8 form in connection with initial assessment at the Department of Oncology, Drammen Hospital, in 2017. Total score < 15 indicates that the patient is potentially frail and may benefit from a geriatric assessment.
| Questions in Geriatric 8 |
Possible answers |
Distribution of answers n (%) |
|---|---|---|
| Has food intake declined over the past 3 months due to loss of appetite, digestive problems, chewing or swallowing difficulties? |
0: Severe decrease in food intake |
16 (11) |
| 1: Moderate decrease in food intake |
38 (26) |
|
| 2: No decrease in food intake |
90 (63) |
|
| Weight loss during the last 3 months |
0: > 3 kg |
50 (35) |
| 1: Does not know |
2 (1) |
|
| 2: 1–3 kg |
21 (15) |
|
| 3: No weight loss |
71 (49) |
|
| Mobility |
0: Confined to bed or chair |
1 (1) |
| 1: Able to get out of bed or chair, but does not go out |
19 (13) |
|
| 2: Goes out |
124 (86) |
|
| Neuropsychological problems |
0: Severe dementia or depression |
4 (3) |
| 1: Mild dementia or depression |
25 (17) |
|
| 2: No psychological problems |
115 (80) |
|
| Body mass index (BMI) |
0: < 19 |
7 (5) |
| 1: 19–21 |
15 (10) |
|
| 2: 21–23 |
20 (14) |
|
| 3: > 23 |
102 (71) |
|
| Takes more than three prescription drugs a day? |
0 = Yes |
92 (64) |
| 1 = No |
52 (36) |
|
| In comparison with other people of the same age, how does the patient consider his/her health status? |
0: Not as good |
29 (20) |
| 0.5: Does not know |
10 (7) |
|
| 1: As good |
48 (33) |
|
| 2: Better |
57 (40) |
|
| Age |
0: > 85 |
13 (9) |
| 1: 80–85 |
34 (24) |
|
| 2: < 80 |
97 (67) |
|
| Total score |
0–14 |
100 (69) |
| 15–17 |
44 (31) |
Discussion
In our survey of 235 patients ≥ 70 who had an initial consultation at the Department of Oncology at Drammen Hospital, we found a high prevalence of frailty indicators that could potentially have provided a basis for supplementary assessment and intervention before and after medical cancer treatment and/or individually adapted treatment.
Medical cancer treatment carries an additional risk for cardiovascular conditions, both acute and as a contributory factor in long-term impairment (13, 14). The fact that as many as 43 % of our patients had established cardiovascular disease at the initial consultation indicates that there is a need to prevent such complications, and that some patients would probably benefit from systematic cooperation between oncologists and cardiologists. Impaired renal function, which was found in 20 % of our population, has been demonstrated to increase the risk of chemotherapy-related toxicity in older cancer patients when renal function is calculated taking account of patients' weight, and not merely their creatinine level (15). In difficult cases, weighing the degree of impaired renal function against the risk associated with medical cancer treatment will require cooperation with a nephrologist. The patients in the study took a median of four drugs, and 26 % took six or more on a regular basis. In busy everyday clinical practice, maintaining an overview of which drugs a patient actually takes and the potential interactions can be demanding. It may be useful to discuss the medication list with a specialist in internal medicine or geriatrics. Studies in geriatric oncology indicate that a geriatric assessment leads to changes in medication for 31 % of patients (5).
Neuropsychological problems will often affect the treatment trajectory of a cancer patient, for example assessment of capacity to consent, handling of complications and compliance with oral medication regimens. A mild degree of cognitive impairment may be difficult to detect during short, targeted examinations in the specialist health service.
Of the patients in this study who underwent the G-8 survey, 20 % reported mild or severe forms of neuropsychological problems. These included both dementia and depression.
Older cancer patients are under-represented in clinical studies (16). Those who are included in studies are often those with a high-performance status (ECOG 0–1) and limited comorbidity. Our review shows that almost half of all new patients at the Department of Oncology were aged 70 or over, and that 34 % of them had a performance status that was poorer than ECOG 1. This means that evidence-based knowledge of the efficacy and adverse effects of cancer therapy is based on a small percentage of the older patients.
In the sub-group where G-8 was used, 69 % were at risk of frailty and according to international recommendations could have benefited from a geriatric assessment. The G-8 tool was not used on 39 % of the patients, and the prevalence of frailty may be overestimated, partly because doctors may have been more inclined to use G-8 for patients who were perceived to have a need for further assessment. The prevalence of potential frailty and comorbidity is high regardless, and consistent with other studies in geriatric oncology. Of 439 Belgian cancer patients over 70 years old, 75 % had a G-8 score of <15 (17). Similarly, the prevalence of potential frailty based upon G-8 was 76 % among lung-cancer patients aged over 70 in the Netherlands (18). In a Norwegian study from Innlandet Hospital Trust, where frailty was defined on the basis of a geriatric assessment, the prevalence was 49 % among patients aged over 70 (19).
In the period in question, G-8 was used at the Department of Oncology to survey frailty indicators, and it was up to the individual doctor to decide what consequences the survey was to have. We have no follow-up data in this study, and therefore do not know whether surveying of frailty indicators led to patients being referred for assessment by a geriatrician or other specialist, or the extent to which such an evaluation influenced planned oncotherapy.
The main strength of this study is that we have included all new patients aged ≥ 70 at a medium-sized general oncology clinic within one calendar year. The fact that surveying of performance status and comorbidity was performed retrospectively is a limitation, as is the fact that we have not related the findings to clinical outcome measures or further management of the patients.
Thanks to Leiv Rusten, Åse Hollender, Wenche Gustafson, Odd Terje Brustugun, Alina Porojnicu and Linn Reichborn for contributing to this article.
The article has been peer-reviewed.
- 1.
Syse A, Veenstra M, Aagnes B et al. Cancer incidence, prevalence and survival in an aging Norwegian population. Nor Epidemiol 2012; 22. doi: 10.5324/nje.v22i2.1556. [CrossRef]
- 2.
Wildiers H, Heeren P, Puts M et al. International Society of Geriatric Oncology consensus on geriatric assessment in older patients with cancer. J Clin Oncol 2014; 32: 2595–603. [PubMed][CrossRef]
- 3.
Mohile SG, Dale W, Somerfield MR et al. Practical assessment and management of vulnerabilities in older patients receiving chemotherapy: ASCO guideline for geriatric oncology. J Clin Oncol 2018; 36: 2326–47. [PubMed][CrossRef]
- 4.
Huisingh-Scheetz M, Walston J. How should older adults with cancer be evaluated for frailty? J Geriatr Oncol 2017; 8: 8–15. [PubMed][CrossRef]
- 5.
Hamaker ME, Te Molder M, Thielen N et al. The effect of a geriatric evaluation on treatment decisions and outcome for older cancer patients - A systematic review. J Geriatr Oncol 2018; 9: 430–40. [PubMed][CrossRef]
- 6.
Oken MM, Creech RH, Tormey DC et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982; 5: 649–55. [PubMed][CrossRef]
- 7.
Levey AS, Bosch JP, Lewis JB et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Ann Intern Med 1999; 130: 461–70. [PubMed][CrossRef]
- 8.
Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. 1916. Nutrition 1989; 5: 303–11, discussion 312 - 3. [PubMed]
- 9.
Bellera CA, Rainfray M, Mathoulin-Pélissier S et al. Screening older cancer patients: first evaluation of the G-8 geriatric screening tool. Ann Oncol 2012; 23: 2166–72. [PubMed][CrossRef]
- 10.
Decoster L, Van Puyvelde K, Mohile S et al. Screening tools for multidimensional health problems warranting a geriatric assessment in older cancer patients: an update on SIOG recommendations. Ann Oncol 2015; 26: 288–300. [PubMed][CrossRef]
- 11.
van Walree IC, Scheepers E, van Huis-Tanja L et al. A systematic review on the association of the G8 with geriatric assessment, prognosis and course of treatment in older patients with cancer. J Geriatr Oncol 2019; 10: 847–58. [PubMed][CrossRef]
- 12.
Martinez-Tapia C, Paillaud E, Liuu E et al. Prognostic value of the G8 and modified-G8 screening tools for multidimensional health problems in older patients with cancer. Eur J Cancer 2017; 83: 211–9. [PubMed][CrossRef]
- 13.
Chang HM, Moudgil R, Scarabelli T et al. Cardiovascular complications of cancer therapy: Best practices in diagnosis, prevention, and management: Part 1. J Am Coll Cardiol 2017; 70: 2536–51. [PubMed][CrossRef]
- 14.
Chang HM, Okwuosa TM, Scarabelli T et al. Cardiovascular complications of cancer therapy: Best practices in diagnosis, prevention, and management: Part 2. J Am Coll Cardiol 2017; 70: 2552–65. [PubMed][CrossRef]
- 15.
Peterson LL, Hurria A, Feng T et al. Association between renal function and chemotherapy-related toxicity in older adults with cancer. J Geriatr Oncol 2017; 8: 96–101. [PubMed][CrossRef]
- 16.
Hurria A, Dale W, Mooney M et al. Designing therapeutic clinical trials for older and frail adults with cancer: U13 conference recommendations. J Clin Oncol 2014; 32: 2587–94. [PubMed][CrossRef]
- 17.
Kenis C, Decoster L, Bastin J et al. Functional decline in older patients with cancer receiving chemotherapy: A multicenter prospective study. J Geriatr Oncol 2017; 8: 196–205. [PubMed][CrossRef]
- 18.
Schulkes KJG, Souwer ETD, van Elden LJR et al. Prognostic value of geriatric 8 and identification of seniors at risk for hospitalized patients screening tools for patients with lung cancer. Clin Lung Cancer 2017; 18: 660–666.e1. [PubMed][CrossRef]
- 19.
Kirkhus L, Šaltytė Benth J, Rostoft S et al. Geriatric assessment is superior to oncologists' clinical judgement in identifying frailty. Br J Cancer 2017; 117: 470–7. [PubMed][CrossRef]