Main findings
A follow-up study undertaken after three years showed that one-fifth of the older patients who were admitted to departments of old-age psychiatry in specialist healthcare service with depression had not suffered any relapse of depression or depressive symptoms during this period.
The prevalence of dementia among older patients with depression admitted to departments of old-age psychiatry in specialist healthcare services tripled over a period of three years.
Various studies report a prevalence of depression at diagnostic level in older people (commonly defined as ≥ 60 years or ≥ 65 years) of 1–5 % (1). Depressive symptoms that combined fail to satisfy the criteria for depression at a diagnostic level may occur up to 2–3 times more frequently than depression. These symptoms may nevertheless be clinically significant in the form of reduced quality of life, reduced social functioning and an increased risk of developing depression (2). The prevalence of depressive conditions (depression at the diagnostic level or depressive symptoms) is higher among older people admitted to hospital or a nursing home than among those who are living at home (3).
The rates of response and remission of depressive episodes are the same among older and younger adults, but older patients are at a higher risk of relapse (4). It has also been shown that in older people depression tends to become chronic, cause problems in daily functioning and is associated with a heightened risk of developing dementia and early death, when compared to older people with no depression (1, 5, 6). Overlapping symptoms of dementia and depression in older people may represent a challenge for differential diagnostics (6, 7). In addition to the negative clinical consequences, depressive conditions in older people may incur major costs for society (1, 8).
Departments of old-age psychiatry in specialist healthcare services in Norway will typically receive older people who are suffering from severe depression and who are unable to take care of themselves, are at a high risk of suicide or have a high degree of comorbidity. No systematic longitudinal studies of depression in older people have been undertaken in Norway, and only a few have been undertaken in the Nordic region. We have therefore undertaken a multi-centre, longitudinal observation study of older patients who have been treated for depression in departments of old-age psychiatry in Norway, Prognosis of Depression in the Elderly (PRODE), to investigate the prognosis with regard to the course of depression, development of dementia and daily functioning over a three-year follow-up period (9, 10).
Material and method
In the PRODE study we included patients who were 60 years or older and referred for admission to departments of old-age psychiatry in specialist health care service because of depression. Patients with severe dementia or other illnesses that prevented verbal communication and patients with short life expectancy due to somatic diseases were not asked to participate. Nine departments of old-age psychiatry (Oslo University Hospital, Ullevål and Aker; Diakonhjemmet Hospital; Vestre Viken Hospital Trust; Innlandet Hospital Trust, Sanderud and Reinsvoll; Stavanger University Hospital; St. Olavs Hospital, Trondheim University Hospital; and Haukeland University Hospital) recruited participants during the period 1 December 2009–1 January 2013.
Healthcare workers at the various study centres collected clinical and demographic information with the aid of a standardised protocol upon admission to and discharge from the department of old-age psychiatry. Training sessions in use of the relevant measurement scales were held before the start of the study and twice annually during the study period. Psychiatrists or specialists in psychology diagnosed dementia and depression in accordance with the criteria in the diagnostic classification system ICD-10 (11). The severity of depressive symptoms was assessed with the Montgomery and Aasberg Depression Rating Scale (MADRS) (12) and the Cornell Scale for Depression in Dementia (CSDD) (13). The former contains ten questions for the patient about depressive symptoms that are assessed on a scale from 0 to 6 (total score 0–60), where higher scores indicate more serious symptoms. Cornell's scale includes information from next of kin or healthcare workers and contains 19 depressive symptoms which is each scored 0–2 according to their degree of severity (total score 0–38). The higher the score, the more serious the symptoms. Cognition was assessed using the Mini Mental State Examination (MMSE), with a score range of 0–30. Here, higher scores indicate better cognitive function (14). Instrumental functions in activities of daily living (I-ADL) were assessed on Lawton and Brady's scale (15). We added together the scores for the eight different I-ADL functions in the scale (total score 8–31), and lower scores indicated better functioning. The doctors assessed physical health on the General Medical Health Rating Scale (GMHR) (16), a graded scale (excellent/good/fair/poor), based on their assessment of medical diagnoses and the number of medications. In the analysis, scores were dichotomised into excellent/good and fair/poor.
The patients received the treatment that was common practice at the study centres. A total of 41 out of 160 (25.6 %) received electroconvulsive therapy, while all 160 received some form of counselling. Upon discharge 134 out of 160 (83.8 %) were using antidepressants, and 42 out of 160 (26.3 %) were using antipsychotic drugs. The corresponding figures at the time of admission (inclusion in the study) were 114 out of 160 (71.3 %) and 38 out of 160 (23.8 %), respectively (9).
One year after inclusion the patients were followed up with a clinical examination at the study centres, and corresponding data were collected as upon admission (Table 1). The results from the hospitalisation period and the one-year follow-up have been published previously (9, 10)
Table 1
Data collected with the aid of measurement instruments at the different times of measurement in the study. T0 = upon inclusion in the study; T1 = upon discharge from a department of old-age psychiatry; T2 = follow-up examination after one year; T3 = follow-up examination after three years.
| T0–T11 | T2 | T3 | ||
|---|---|---|---|---|
| Depression | ||||
| Depression according to the ICD-10 criteria (11) | X | X | ||
| Montgomery and Aasberg Depression Rating Scale (MADRS) (12, 17) | X | X | X | |
| Cornell Scale for Depression in Dementia (CSDD) (13) | X | X | X | |
| Clinical course of depression | X | X | ||
| Cognition | ||||
| Dementia according to the ICD-10 criteria (11) | X | X | X | |
| Mild cognitive impairment (MCI) according to the Winblad criteria (23) | X | X | ||
| X | X | X | ||
| Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) (19) | X | X | X | |
| The Clinical Dementia Rating (CDR) (20) | X | |||
| Physical health | ||||
| General Medical Health Rating Scale (GMHR) (16) | X | X | X | |
| Daily functioning | ||||
| Lawton & Brody's scale for instrumental activities in daily living (I-ADL) (15) | X | X | X | |
1During hospitalisation in department of old-age psychiatry
Follow-up examination after three years
Three years after inclusion, the patients and their next of kin, if any, as well as healthcare workers from nursing homes or home-based care wherever relevant, were contacted in telephone surveys administered from Innlandet Hospital, Sanderud. In the interviews we collected information on the course of depression, cognition, physical health (GMHR) and functioning (Lawton and Brody's scale for I-ADL). If there were discrepancies between the information provided by the patient and the next of kin/healthcare workers, the interviewers would make a discretionary decision regarding what information should be deemed most valid. The patients were asked to describe their experience of the course of depression (cured of depression / relapse of depression / persistently ill from depression) after the one-year follow-up. They were interviewed with a validated telephone version of the MADRS (17) (n = 105). In addition, the next of kin and/or healthcare workers completed the Cornell scale for depression in dementia (n = 106).
The patients (n = 95) went through a validated telephone version of the Mini Mental State Examination (MMSE) (18). Eighteen further patients whose health condition precluded implementation of the test by telephone went through a traditional MMSE test (14) in the primary health service. Cognitive change over time was measured by the Informant Questionnaire on Cognitive Decline in the Elderly (19), and the Clinical Dementia Rating (20) was completed by the interviewer based on all information on cognition derived from the telephone interviews. The Informant Questionnaire on Cognitive Decline in the Elderly contains 16 questions on cognitive changes over time, for the purposes of this study over the last two years. The Clinical Dementia Rating is used to evaluate cognition and functioning based on available information on memory, orientation, judgement and problem solving, community affairs, home and hobbies, and personal care.
Based on the data that the study had collected on depression at the one-year and three-year follow-up surveys, we classified the course of depression over three years in the categories favourable/less favourable/unfavourable/unclassifiable. The category unfavourable course included patients who had attempted suicide, suffered at least one relapse that required hospitalisation or were assessed by the interviewer as persistently ill from a depressive condition. The less favourable category included patients who had shown symptoms of depression (MADRS > 9 (21) and/or CSDD > 8 (22)), but did not fulfil the criteria for a favourable or unfavourable course. A favourable course meant that the patients had not suffered a relapse of depression or depressive symptoms (MADRS > 9 and/or CSDD > 8) in the follow-up period.
Three of the authors, all psychiatrists with a PhD degree, used all available information from the surveys to independently classify cognition in the three-year survey into the categories no cognitive impairment, mild cognitive impairment according to the Winblad criteria (23), and dementia according to ICD-10 (11). The three experts were in initial agreement on the classification of the cognitive status of 102 out of 131 patients (77.9 %), and held a consensus meeting to classify those 29 patients (22.1 %) for whom there was no correspondence.
Ethics
The patients and their next of kin received written and oral information about the study and provided written consent to participation. The next of kin gave written consent on behalf of patients who had no capacity to provide consent. The study was approved by the Regional Committee for Medical and Health Research Ethics, South-Eastern Norway (ref. no. 2009/1774) and by the Data Protection Officer at Oslo University Hospital, and is registered in ClinicalTrials.gov (NCT01952366).
Statistics
Independent and paired t-tests were used for continuous data, and the chi-square test and McNemar's test were used for categorical data. All analyses were performed in SPSS, version 25.
Results
In six of the nine participating study centres, the sex and age of patients who declined to participate could be established. In these six study centres altogether 174 eligible patients were asked to participate, and 38 declined. There were no differences in terms of sex and age between the patients who agreed or declined to participate in the study (9).
Figure 1 shows the patient flow in the study, and Table 2 shows patient characteristics upon inclusion in the study (T0), discharge from the department of old-age psychiatry (T1) and three years after inclusion (T3). Compared to the 29 patients who did not complete the study, those 131 who completed T3 were younger (p = 0.028), had a higher MMSE score (p < 0.001), a higher MADRS score (p = 0.034), more often reported excellent/good physical health (p = 0.001) at T0 and more rarely dementia at T1 (p = 0.004).
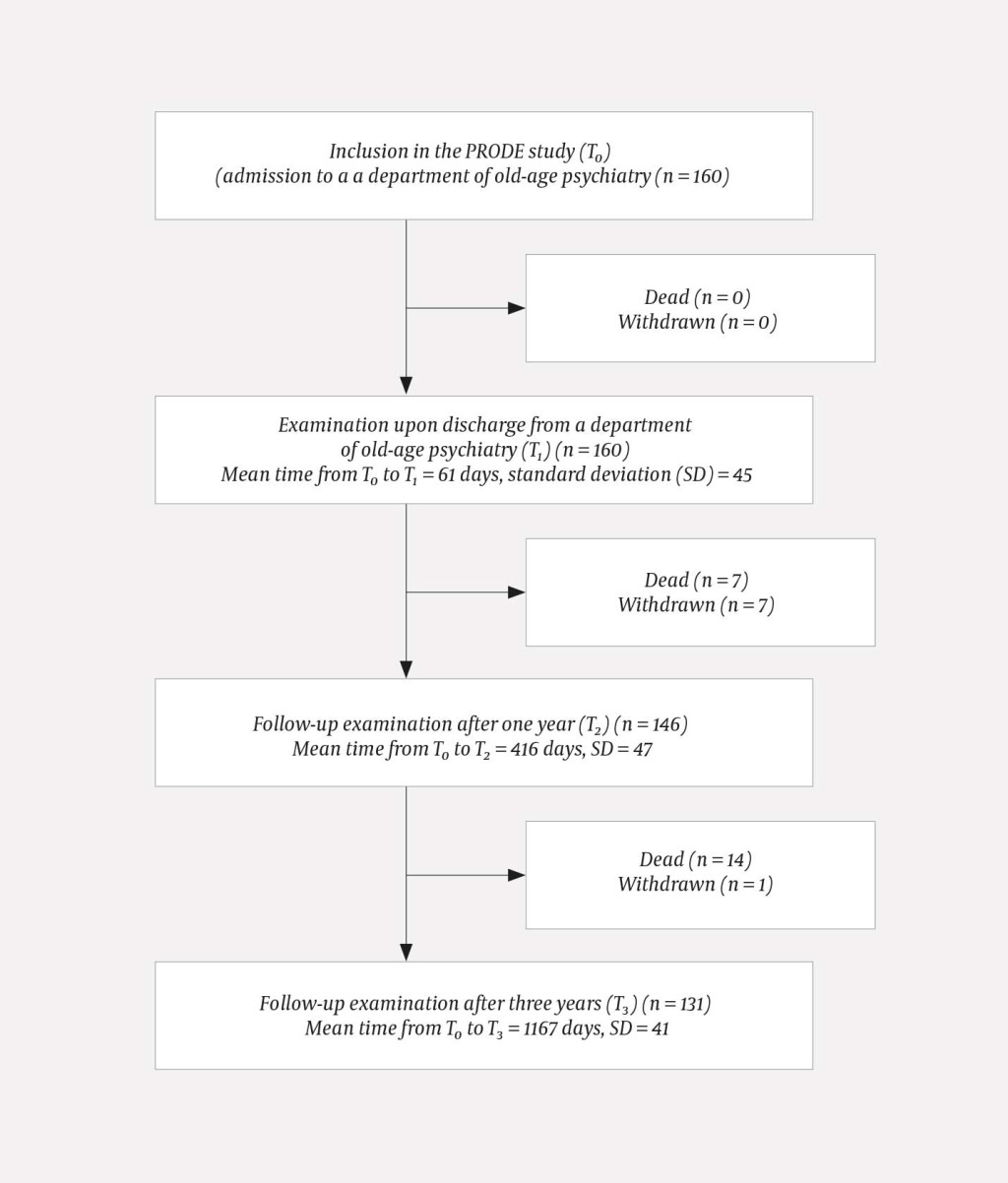
Table 2
Demographic and clinical data for the study sample upon admission to a department of old-age psychiatry and after three years. The data are presented with the values prevailing at the times of measurement. T0 = upon inclusion in the study; T1 = upon discharge from a department of old-age psychiatry; T3 = follow-up examination after three years.
| T0 | T1 | T3 | ||
|---|---|---|---|---|
| Demographics | ||||
| Age in years, mean (standard deviation, SD) | 76.1 (6.8) | 78.8 (6.4) | ||
| Proportion of women | 72.5 % | 73.9 % | ||
| Years of education, mean (SD) (n = 151 at T0, n = 124 at T3) | 10.0 (3.0) | 10.0 (3.0) | ||
| Marital status | ||||
| Married/co-habiting | 40.0 % | 38.9 % | ||
| Widow/widower/divorced/single | 60.0 % | 61.1 % | ||
| Depression | ||||
| First-onset depression upon inclusion | 30.6 % | |||
| Bipolar disorder according to ICD-10 | 6.3 % | |||
| Psychotic depression according to ICD-10 | 9.4 % | |||
| Age at first onset of depression in categories (n = 157 at T0) | ||||
| < 60 years | 49.0 % | |||
| ≥ 60 years | 51.0 % | |||
| MADRS1 score, mean (SD) (n = 157 at T0) | 26.1 (8.6) | |||
| Cognition | ||||
| Mini Mental State Examination (MMSE), mean (SD) (n = 156 at T0) | 25.9 (3.6) | |||
| Dementia diagnosis according to ICD-10 | 8.8 % | 30.5 % | ||
| Mild cognitive impairment (MCI) | 33.6 % | |||
| Physical health | ||||
| GMHR2 categories | ||||
| Excellent/good | 48.1 % | 27.5 % | ||
| Fair/poor | 51.9 % | 72.5 % | ||
| Daily functioning | ||||
| I-ADL3 score, mean(SD) (n = 136 at T0, n = 117 at T3) | 15.3 (6.1) | 17.9 (7.9) | ||
| Nursing home resident | 1.3 % | 15.3 % | ||
1MADRS = Montgomery and Aasberg Depression Rating Scale
2GMHR = General Medical Health Rating Scale
3Lawton and Brody's scale for instrumental activities in daily living
At T3 altogether 21 patients had died (Figure 1). There was no difference between the sexes in terms of mortality (p = 0.480).
Status after three years
At T3, when compared to T0, a larger proportion of the patients had fair/poor physical health (p < 0.001), a larger proportion lived in nursing homes (p < 0.001), and we found a higher I-ADL score (p = 0.001 for n = 103 with measurements at both time points) (Table 2). A larger proportion had been diagnosed with dementia (p < 0.001). The course of depression over three years showed that 24 out of 131 patients (18.3 %) were in the 'favourable' category, 55 out of 131 (42.0 %) in the 'less favourable', 51 out of 131 (38.9 %) in the 'unfavourable', and 1 (0.8 %) was unclassifiable.
Discussion
This study shows that older people who have been treated for depression in Norwegian departments for old-age psychiatry have an unfavourable prognosis with regard to relapse of depression and persistent depressive symptoms. Of the 131 patients who were followed up over three years, only 24 (18.3 %) had been free from depression and depressive symptoms. Moreover, the study shows that a larger proportion of the patients suffered from dementia after three years when compared to the start of the study: 40 out of 131 (30.5 %) and 14 out of 160 (8.8 %) respectively.
Our results concerning the course of depression correspond to findings in international studies, but differences in patient samples, the use of different definitions of depression and different follow-up times render any direct comparisons difficult (5, 24). In our definition, a relapse of depression during the follow-up period implied an unfavourable course of the illness, even if the patient recovered from the relapse. Other studies have defined this differently, and this may partly explain why relatively many participants were classified in the least favourable category in our study. Many previous studies have excluded patients with dementia, while this study is one of very few longitudinal studies that have also included patients with dementia (1, 5). This may have had an impact on the results, since depression and dementia have overlapping symptoms.
The patient sample in our study was a selected group and included many patients with severe depression and multimorbidity. The results are therefore not transferable to all older people with depression. A Dutch study undertaken over two years of 285 patients ≥ 60 years who suffered from depression without dementia and had been recruited from both health institutions and general practice found that 61 % had depressive symptoms that were of a more or less chronic nature, 20 % had intermittent symptoms and 19 % remained healthy after the initial treatment (24). Furthermore, the study showed that 56 % of the patients who did not fulfil the diagnostic criteria for depression at the time of the two-year follow up had depressive symptoms. The study included a different patient sample and used different screening tools than those in our study, but it also shows that depressive conditions in older people are often chronic.
Depression in the elderly can be associated with dementia in several ways (6). Depression in earlier life may be a risk factor for dementia, depression may be a prodromal symptom of dementia, or both conditions may occur in parallel (1, 6). We found that a larger proportion of the patients suffered from dementia at the time of the three-year follow-up, when compared to the examination three years earlier, 30.5 % and 8.8 % respectively. Our findings concur with studies that describe how depression in older people may be a prodromal symptom of dementia (25). The depressive illness may also have accelerated/exacerbated an ongoing pre-clinical dementia condition, as other studies have found (6). More severe depression and persistent depressive symptoms seem to be especially associated with an increased risk of developing dementia (6, 25). At the three-year follow-up, 64.1 % of the patients suffered from either dementia or mild cognitive impairment. This shows that varying degrees of cognitive impairment are common in the course of depression among older people. Studies have shown that symptoms of cognitive impairment in cases of depression in older patients are more persistent that previously assumed (1).
Strengths and weaknesses of the study
One of the study's weaknesses is the absence of a control group consisting of older people without depression. A further weakness is the change in the procedure for data collection from face-to-face interviews to telephone interviews at the three-year follow-up. This may have had an impact on the results, but it is hard to tell in what direction with regard to dementia development and the course of depression. Depressive symptoms may fluctuate and ought to have been assessed more often during the follow-up period. Many different healthcare workers were involved in the assessment of the patients, and their assessments may therefore diverge. To ensure a more consistent assessment of the patients we therefore held training sessions before the start of the study and twice annually during the study period.
The study participants had a high degree of comorbidity and constituted a selected group of hospitalised patients with depression, and the results are therefore not transferable to all older people with depression. Some demographic and clinical variables had no valid values. One of the study's strengths lies in our use of few exclusion criteria. Another strength is that we are able to account for all patients at the three-year follow-up, and that only eight participants out of 160 withdrew during the process (24). Furthermore, we have used validated measurement instruments in a longitudinal design. The findings are clear and robust.
Conclusion
Our study shows that older people who suffer from depression and have been treated in departments of old-age psychiatry at specialist healthcare services have an unfavourable prognosis in terms of the course of depression and development of dementia over a three-year period. Doctors and other healthcare workers who are in contact with similarly older patients with depression should be aware of the risk of relapse and development of dementia.
The study was funded by South-Eastern Norway Regional Health Authority and Innlandet Hospital Trust, but these institutions have had no influence on the ideas behind the study, the collection of data, interpretation of results or the drafting of the article.
The article has been peer reviewed.
- 1.
Thomas A. Depression in older people. I: Dening T, Thomas A, red. Oxford Textbook of Old-age psychiatry. 2. utg. Oxford: Oxford University Press, 2013: 544–69.
- 2.
Meeks TW, Vahia IV, Lavretsky H et al. A tune in "a minor" can "b major": a review of epidemiology, illness course, and public health implications of subthreshold depression in older adults. J Affect Disord 2011; 129: 126–42. [PubMed][CrossRef]
- 3.
Rosenvinge BH, Rosenvinge JH. Occurrence of depression in the elderly–a systematic review of 55 prevalence studies from 1990-2001. Tidsskr Nor Lægeforen 2003; 123: 928–9. [PubMed]
- 4.
Mitchell AJ, Subramaniam H. Prognosis of depression in old age compared to middle age: a systematic review of comparative studies. Am J Psychiatry 2005; 162: 1588–601. [PubMed][CrossRef]
- 5.
Cole MG, Bellavance F. The prognosis of depression in old age. Am J Geriatr Psychiatry 1997; 5: 4–14. [PubMed][CrossRef]
- 6.
Bennett S, Thomas AJ. Depression and dementia: cause, consequence or coincidence? Maturitas 2014; 79: 184–90. [PubMed][CrossRef]
- 7.
Bystad M, Pettersen K, Grønli OK. Depression or Alzheimer-type dementia? Tidsskr Nor Legeforen 2014; 134: 525–8. [PubMed][CrossRef]
- 8.
Tollånes MC, Knudsen AK, Vollset SE et al. Disease burden in Norway in 2016. Tidsskr Nor Legeforen 2018; 138. doi: 10.4045/tidsskr.18.0274. [PubMed][CrossRef]
- 9.
Borza T, Engedal K, Bergh S et al. The course of depression in late life as measured by the Montgomery and Asberg Depression Rating Scale in an observational study of hospitalized patients. BMC Psychiatry 2015; 15: 191. [PubMed][CrossRef]
- 10.
Borza T, Engedal K, Bergh S et al. Trajectories of depression in late life: A 1-year follow-up study. Dement Geriatr Cogn Disord 2017; 43: 180–92. [PubMed][CrossRef]
- 11.
The ICD-10 Classification of Mental and Behavioural Disorders – Diagnostic Criteria for Research. Geneva: World Health Organization, 1993.
- 12.
Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry 1979; 134: 382–9. [PubMed][CrossRef]
- 13.
Alexopoulos GS, Abrams RC, Young RC et al. Cornell Scale for Depression in Dementia. Biol Psychiatry 1988; 23: 271–84. [PubMed][CrossRef]
- 14.
Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189–98. [PubMed][CrossRef]
- 15.
Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist 1969; 9: 179–86. [PubMed][CrossRef]
- 16.
Lyketsos CG, Galik E, Steele C et al. The General Medical Health Rating: a bedside global rating of medical comorbidity in patients with dementia. J Am Geriatr Soc 1999; 47: 487–91. [PubMed][CrossRef]
- 17.
Hermens ML, Adèr HJ, van Hout HP et al. Administering the MADRS by telephone or face-to-face: a validity study. Ann Gen Psychiatry 2006; 5: 3. [PubMed][CrossRef]
- 18.
Newkirk LA, Kim JM, Thompson JM et al. Validation of a 26-point telephone version of the Mini-Mental State Examination. J Geriatr Psychiatry Neurol 2004; 17: 81–7. [PubMed][CrossRef]
- 19.
Jorm AF. A short form of the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE): development and cross-validation. Psychol Med 1994; 24: 145–53. [PubMed][CrossRef]
- 20.
Hughes CP, Berg L, Danziger WL et al. A new clinical scale for the staging of dementia. Br J Psychiatry 1982; 140: 566–72. [PubMed][CrossRef]
- 21.
Rush AJ, Kraemer HC, Sackeim HA et al. Report by the ACNP Task Force on response and remission in major depressive disorder. Neuropsychopharmacology 2006; 31: 1841–53. [PubMed][CrossRef]
- 22.
Barca ML, Engedal K, Selbaek G. A reliability and validity study of the cornell scale among elderly inpatients, using various clinical criteria. Dement Geriatr Cogn Disord 2010; 29: 438–47. [PubMed][CrossRef]
- 23.
Winblad B, Palmer K, Kivipelto M et al. Mild cognitive impairment–beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med 2004; 256: 240–6. [PubMed][CrossRef]
- 24.
Comijs HC, Nieuwesteeg J, Kok R et al. The two-year course of late-life depression; results from the Netherlands study of depression in older persons. BMC Psychiatry 2015; 15: 20. [PubMed][CrossRef]
- 25.
Mirza SS, Wolters FJ, Swanson SA et al. 10-year trajectories of depressive symptoms and risk of dementia: a population-based study. Lancet Psychiatry 2016; 3: 628–35. [PubMed][CrossRef]