Naloxone nasal spray – bioavailability and absorption pattern in a phase 1 study
Main findings
Concentrated naloxone nasal spray makes it possible to administer a therapeutic dose of opioid antagonist with a volume of 0.1 ml
The study provided a sound basis for determining the optimal dose of naloxone in nasal spray form in later studies
Altogether 250–300 Norwegians die each year from opioid overdose. Europe and the United States have seen a major increase in fatalities in recent years (1, 2). Opioid overdose can cause respiratory depression resulting in death if respiratory support or the opioid antagonist naloxone is not administered. Naloxone has been available since the 1970s and has been administered either intramuscularly or intravenously. The guidelines for the ambulance service recommend 0.4–0.8 mg naloxone given intramuscularly, followed by 0.4 mg intravenously if needed for greater effect (3, 4).
In June 2018, a naloxone nasal spray developed in Norway received marketing authorisation in 12 European countries with reversal of suspected opioid overdose as the indication (5). In this article we describe the first of five studies of different doses and modes of administration of naloxone that we have conducted, from concept to approved medication.
Naloxone nasal spray was proposed in the 1990s as an alternative to the injection treatment used at that time (6). The aim was to facilitate Take Home Naloxone programmes: distribution of naloxone to anyone who might witness cases of opioid overdose. This is consistent with the World Health Organization’s (WHO) recommendations and has constituted a key measure in the national overdose strategy since 2014 (7). Initially only low-concentration injection formulas existed (1 mg/ml, spray volume 2 ml), where the injection needle was exchanged for an atomiser for nasal administration. Naloxone spray is also relevant for healthcare personnel and may reduce the risk of needle stick injury.
Naloxone nasal spray has long been used in many countries without clinical testing or approval by pharmaceutical regulatory agencies (off label). Studies of improvised, low-concentration nasal spray solutions showed, as expected, that their bioavailability was modest, at a level of only 4–11 % (8, 9). These sprays could not deliver a systemic, therapeutic dose corresponding to 0.4–0.8 mg parenterally. Their use has therefore been controversial (10), but considered necessary in a situation of increasing overdose fatalities. In 2012, the US authorities invited the pharmaceutical industry to develop nasal sprays that could receive marketing authorisation for bystander administration (11), and a naloxone nasal spray was already approved in the United States as early as 2015. There has been no equivalent public initiative in Europe. In 2010, the Norwegian Directorate of Health rejected an application from the Norwegian University of Science and Technology (NTNU) for funding to develop a highly concentrated naloxone nasal spray, on the grounds that there was no commercial interest in such a product. Only four years later, the same authorities endorsed the use of a non-approved spray in their overdose strategy.
NTNU started the innovation process for a naloxone nasal spray in 2008. The academic formulation pharmacist Phatsawee Jansook, University of Bangkok, was commissioned by NTNU to develop an adequate nasal spray formulation. Professor Thorsteinn Loftsson, University of Iceland, supported this process.
The first trial of a new highly concentrated (20 mg/ml) naloxone nasal spray using healthy volunteers was conducted in 2013. The aim was to investigate whether it was possible to administer a therapeutic dose of naloxone with a low-volume (0.1 ml) nasal spray. In this article, we will give an account of the results of the pilot study and the journey towards marketing authorisation of the nasal spray. This illustrates how smaller academic research communities can also take the lead in developing new drugs.
Method
The study was an open, randomised, two-way phase 1 crossover study undertaken in five healthy men. The study was approved by the Regional Committee for Medical and Health Research Ethics (South-East C, REC no. 2012/1970) and the Norwegian Medicines Agency (EudraCT no. 2012–004989–18). The study was conducted in accordance with good clinical practice (GCP), and the protocol was registered at clinicaltrials.gov (NCT01939444). All the participants gave their written informed consent before inclusion, and they were insured through the Drug Liability Association and received compensation of NOK 1500 per day of the trial.
Healthy men in the age group 18–45 years were screened with anamnesis, electrocardiogram (ECG) and blood samples for haemoglobin, creatinine, alanine aminotransferase (ALT), aspartate aminotransferase (AST) and gamma-glutamyltransferase (GGT). Participants were excluded in cases of deviating test results, drug allergy, suspicion of substance use, regular use of medications, nasal disease or previous nasal surgery.
The nasal spray contained naloxone hydrochloride at a concentration of 20 mg/ml and well-known additives (12). It was administered with a single-use product that delivered 0.1 ml of solution per activation of the spray (Aptar bidose, Aptar Pharma, Louveciennes, France). The nasal sprays were manufactured by the Norwegian Institute of Public Health (Biopharmaceutical production) which was certified for good manufacturing practice. Naloxon B. Braun 0.4 mg/ml was delivered by the hospital pharmacy in Trondheim and used as a drug of comparison.
In the study, 2.0 mg intranasal naloxone (IN) was compared with 1.0 mg naloxone administered intravenously (IV). Each experiment lasted for six hours, and the participants were sedentary for the first two hours, but afterwards were free to walk around. All the participants received both treatment options with at least 72 hours between each treatment. The treatment sequence was randomised using a digital solution from the Unit for Applied Clinical Research at NTNU. Follow-up interviews were conducted with the participants two weeks after their final visit. The study was undertaken in the Clinical Research Facility at St. Olavs hospital/NTNU in August–October 2013.
A total of 15 blood samples were taken at time = –10, 2, 5, 10, 15, 20, 25, 30, 35, 45, 60, 90, 120, 240 and 360 minutes, where naloxone was administered at time = 0. The blood samples were collected in Vacuette tubes without gel and allowed to coagulate before centrifuging at 2 200 G. The serum was frozen at –80 °C until analysis. Adverse effects were recorded in accordance with good clinical practice and national legislation.
The primary endpoint was absolute intranasal bioavailability. Secondary endpoints were maximum concentration (Cmax), time to maximum concentration (Tmax), and safety.
Five people were included, fewer than the 12 that is usually considered the minimum for such studies. The aim was to investigate whether the formulation had potential for further development and, if so, to provide a basis for deciding the strengths/doses to be used in subsequent studies.
Quantification of naloxone was performed using liquid chromatography-tandem mass spectrometry (LC-MS/MS). The method was validated and has previously been described in full (12).
Serum concentrations were assayed using non-compartmental analysis. The area under the curve up to the last measurement at 360 minutes (AUC0→t), half-life, Cmax and Tmax were calculated using WinNonlin version 6.4 (Pharsight Corp., USA). The dose-corrected AUC0→t was used to calculate bioavailability. Any samples with more than double or less than half the expected concentration were discarded (2/150 samples). SPSS version 23 was used for descriptive statistics. Data are presented as mean and minimum–maximum values unless otherwise stated.
Results
Seven men agreed to participate in the study. Five met the inclusion criteria and were randomised, all of whom completed the study. Their mean age was 23.4 years (21–25), height 179.6 cm (175–187), weight 73.9 kg (64.0–91.8), and body mass index 22.8 (20.9–26.2).
Absolute bioavailability was 47 % (24–66). For two of the participants, a drop of nasal spray ran out of the nose immediately after spray administration. The bioavailability in these individuals was 24 % and 53 %, respectively. AUC0→t was 264 min*ng/ml for 2.0 mg intranasal and 282 min*ng/ml for intravenous administration.
The maximum concentration after use of the nasal spray was 4.2 (1.5–7.1) ng/ml, and the time to maximum concentration was 16 (5–25) minutes. The mean terminal half-life was 80 and 90 minutes for intranasal and intravenous naloxone, respectively (Table 1).
Table 1
Pharmacokinetic variables in healthy volunteers following intranasal and intravenous administration of naloxone in an open, randomised, two-way crossover study (N = 5). Data are presented as mean (minimum–maximum values). Cmax = maximum concentration, Tmax = time to maximum concentration, AUC0→t = area under the curve up to and including the last measurement at 360 minutes.
| Treatment |
Cmax |
Tmax (min) |
AUC0→t (min*ng/ml) |
Distribution volume (l) |
Clearance (ml/min) |
Half-life (min) |
|---|---|---|---|---|---|---|
| 2.0 mg intranasal naloxone |
4.2 (1.5–7.1) |
16 (5–25) |
264 (150–408) |
430 (172–688) |
3 615 (2 198–4 431) |
80 (50–132) |
| 1.0 mg intravenous naloxone |
22.7 (7.7–49.2) |
2.6 (2–5) |
282 (211–451) |
482 (224–713) |
3 656 (2 191–4 623) |
90 (66–133) |
Intravenous administration resulted immediately in a high concentration of naloxone, which then decreased rapidly. The concentration after nasal spray exceeded that following intravenous administration after ten minutes and remained higher until the 240-minute sample (Figure 1).
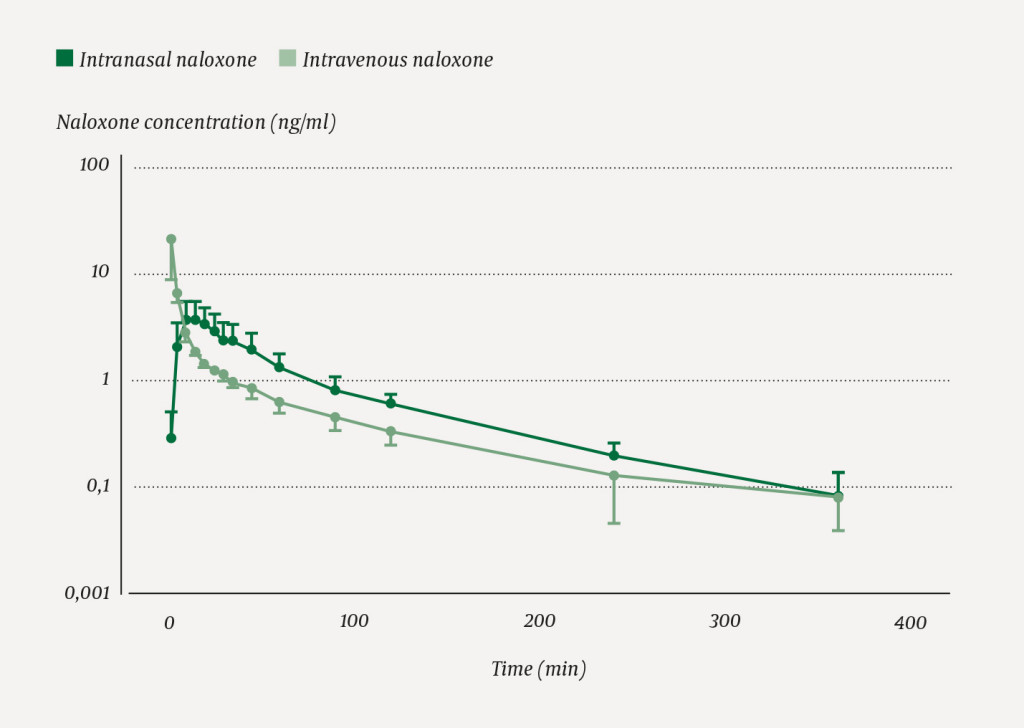
Only mild taste sensations were reported in response to the nasal spray. Participants experienced no other adverse events.
Discussion
Bioavailability of just under 50 % in the pilot study showed that it was possible to administer a therapeutic dose of naloxone in 0.1 ml via a highly concentrated nasal spray. Absorption was also rapid, with serum concentrations after nasal administration remaining sufficiently high throughout the study. However, there was a need to confirm the findings in more comprehensive studies. Adjustment of the dose was also required to be able to offer a spray that would be equivalent to an intramuscular dose of 0.4–0.8 mg. The results for central pharmacokinetic variables were confirmed in subsequent studies.
Four different concentrations of the nasal spray were produced: 4, 8, 16 and 20 mg/ml. We chose to test the strongest solution of 20 mg/ml first, as previous reports on naloxone had indicated low bioavailability (8). In order to determine absolute bioavailability, we chose in the initial studies to use intravenous naloxone as a comparator, as this is the recommended route of administration in the Summary of Product Characteristics for injectable naloxone. In subsequent studies, naloxone was administered via the intramuscular route, currently the most commonly used route of administration by the Norwegian ambulance service.
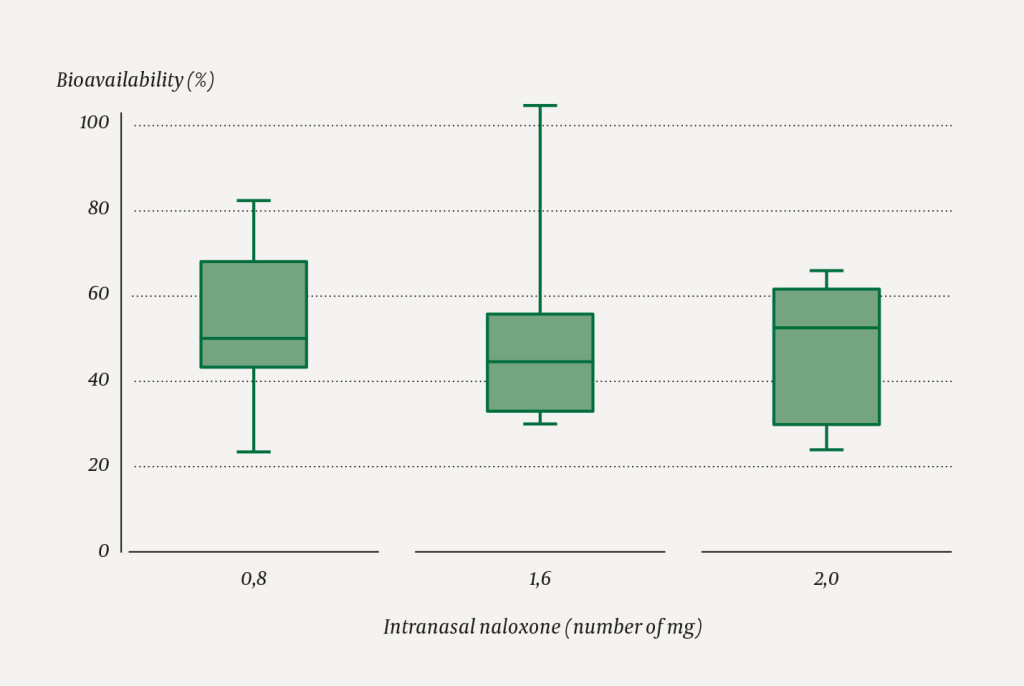
The bioavailability of 47 % was markedly higher than previously reported and corresponds well to the 47–54 % bioavailability in subsequent studies by ourselves and others (12–14). Our results were thus a good reflection of the central tendency for absolute bioavailability. Variability was underestimated in the pilot study, where minimum–maximum values were 24–66 % (range 42) versus 23–83 % (range 60) and 30–105 % (range 75) for two different doses of nasal spray in the following study (12). This is illustrated in Figure 2 with a quartile-based box and whisker plot. A bioavailability of approximately 50 % means in practice that the dose must be doubled if naloxone is to be administered as a nasal spray rather than by injection.
Intranasal and intramuscular naloxone have a relative bioavailability of about 44–54 % in healthy volunteers (13–15). However, relative bioavailability was 75 % in individuals who received naloxone during ongoing infusion of the opioid remifentanil (16). There may therefore be a pharmacokinetic interaction between remifentanil and naloxone. If this is a general opioid effect, and not just specific to remifentanil, it may have an impact on the current approval processes for new naloxone formulations in opioid overdose.
Our maximum concentration (Cmax) was 4.2 ng/ml after 2 mg intranasal naloxone. When the same dose (2 mg) was administered in a more dilute solution, the Cmax was 0.5 ng/ml. Our highly concentrated solution thus yielded a Cmax eight times higher than when the same dose was given in a more dilute solution, equivalent to that used until recently in the Norwegian Take Home Naloxone programme (9). Our Cmax was also four times higher than the standard regulatory comparison dose of 0.4 mg given intramuscularly (Cmax 1.1 ng/ml) (17). The 2 mg dose is therefore higher than that required for the nasal spray to be compatible with the 0.4 mg intramuscular comparison standard.
Intravenous administration of naloxone yields very high serum naloxone concentrations (22.7 ng/ml). The estimated time to maximum concentration (Tmax) after intravenous administration of naloxone was 2.6 minutes in this study, compared to 16 minutes for the nasal spray. In later studies, we observed a somewhat higher Tmax (18–20 min), comparable with the 15–30 minutes reported for other high-concentration naloxone sprays (12–15) and with the Tmax of 10–23 minutes reported for intramuscular naloxone (13, 15, 17).
Rapid attainment of a high antidote concentration may be necessary in the minority of patients who cannot be ventilated. However, it comes at a cost of increased risk of withdrawal. Opioid withdrawal is not just an unpleasant adverse effect, but can also increase the risk of further overdoses and may cause patients to refuse follow-up or to discharge themselves from hospital (18). In order to reduce acute withdrawal, many ambulance services have switched to intramuscular naloxone as first-line treatment, with intravenous naloxone administered only when necessary. Intranasal administration (Figure 1) results in a serum concentration time course similar to that seen following intramuscular injection. Our later studies in healthy volunteers showed a rapid onset and potent effect of intravenous naloxone (1 mg) measured in terms of pupillary size, while an intramuscular dose of 0.8 mg yielded less potent and slower onset effects (16, 19), consistent with a lower risk of withdrawal.
Little attention has been paid to the distribution volume and clearance of naloxone. The latter in particular is interesting. We confirmed previously reported clearance values of approximately 3500 ml/min (20, 21), more than double the maximum capacity of the liver. This indicates extensive extrahepatic elimination of naloxone. In principle, all organs with demonstrable expression of drug-metabolising enzymes, including the nasal mucosa, may be involved in this process.
The U.S. Food and Drug Administration (FDA) has recommended that new naloxone products achieve serum concentrations comparable to those seen with 0.4 mg intramuscular naloxone, especially in the first few minutes after administration (11). We have not performed this comparison in the current study, but our Cmax and Tmax were similar to those found for intramuscular naloxone, which also gave us a clear indication that we would be able to develop a commercial product.
The pilot study showed that our formulation could yield a serum concentration on a level with those achieved with established modes of administration. This was important knowledge early in the development process and laid the foundations for our subsequent studies (12, 14, 16, 19). What at first glance appears to be a modest study with five men, initiated a process that has led to five further studies. From the birth of an idea at NTNU, we have progressed all the way to obtaining marketing authorisation of Ventizolve 1.26 mg naloxone through an industry collaboration with dne pharma as and Farma Industri AS. This demonstrates that small academic communities that receive funding and the freedom to conduct experimental research can deliver results amid fierce competition from the international pharmaceutical industry.
This article has been peer-reviewed.
- 1.
European Monitoring Centre for Drugs and Drug Addiction. European Drug Report 2016: Trends and Developments. Luxembourg: Publications Office of the European Union, 2016. http://www.drugsandalcohol.ie/25579/1/Final_EDR_report.pdf Lest 19.6.2019.
- 2.
Rudd RA, Seth P, David F et al. Increases in drug and opioid-involved overdose deaths - United States, 2010–2015. MMWR Morb Mortal Wkly Rep 2016; 65: 1445–52. [PubMed][CrossRef]
- 3.
Oslo universitetssykehus. Tiltaksbok ambulanse. Opiatoverdose.
- 4.
Helse Midt-Norge. Felles retningslinjer ambulanse Midt-Norge: Naloxon.
- 5.
Midling AS. Nå skal norsk nesespray redde liv i tolv land. Forskning.no 26.6.2018. https://web.archive.org/web/20190618103133/https://forskning.no/partner-rus-og-avhengighet-alkohol-og-narkotika/na-skal-norsk-nesespray-redde-liv-i-tolv-land/1192214 Lest 18.6.2019.
- 6.
Strang J, Darke S, Hall W et al. Heroin overdose: the case for take-home naloxone. BMJ 1996; 312: 1435–6. [PubMed][CrossRef]
- 7.
Nasjonal overdosestrategi 2014–2017. Ja visst kan du bli rusfri – men først må du overleve. IS-0418. Oslo: Helsedirektoratet, 2014. https://www.regjeringen.no/contentassets/43121155483947d79316af20c68e6d7d/overdosestrategi_230414.pdf Lest 19.6.2019.
- 8.
Dowling J, Isbister GK, Kirkpatrick CM et al. Population pharmacokinetics of intravenous, intramuscular, and intranasal naloxone in human volunteers. Ther Drug Monit 2008; 30: 490–6. [PubMed]
- 9.
McDonald R, Danielsson Glende Ø, Dale O et al. International patent applications for non-injectable naloxone for opioid overdose reversal: Exploratory search and retrieve analysis of the PatentScope database. Drug alcohol rev 2018; 37: 205–15.
- 10.
Strang J, McDonald R, Tas B et al. Clinical provision of improvised nasal naloxone without experimental testing and without regulatory approval: imaginative shortcut or dangerous bypass of essential safety procedures? Addiction 2016; 111: 574–82. [PubMed][CrossRef]
- 11.
Hertz S. Naloxone for outpatient use: Data required to support an NDA. U.S. Food and Drug Administration, 2012. https://web.archive.org/web/20161024174802/http://www.fda.gov/downloads/Drugs/NewsEvents/UCM300874.pdf Lest 19.6.2019.
- 12.
Tylleskar I, Skulberg AK, Nilsen T et al. Pharmacokinetics of a new, nasal formulation of naloxone. Eur J Clin Pharmacol 2017; 73: 555–62. [PubMed][CrossRef]
- 13.
McDonald R, Lorch U, Woodward J et al. Pharmacokinetics of concentrated naloxone nasal spray for opioid overdose reversal: Phase I healthy volunteer study. Addiction 2018; 113: 484–93. [PubMed][CrossRef]
- 14.
Skulberg AK, Åsberg A, Khiabani HZ et al. Pharmacokinetics of a novel, approved, 1.4-mg intranasal naloxone formulation for reversal of opioid overdose-a randomized controlled trial. Addiction 2019; 114: 859–67. [PubMed][CrossRef]
- 15.
NARCAN® (naloxone hydrochloride) nasal spray – Prescribing Information, 2017. https://web.archive.org/web/20190618112914/https://s3-us-west-2.amazonaws.com/narcan-assets-uswest/NARCAN-Prescribing-Information.pdf Lest 18.6.2019.
- 16.
Skulberg AK, Tylleskar I, Nilsen T et al. Pharmacokinetics and -dynamics of intramuscular and intranasal naloxone: an explorative study in healthy volunteers. Eur J Clin Pharmacol 2018; 74: 873–83. [PubMed][CrossRef]
- 17.
Evzio – Prescribing Information. 2016. https://web.archive.org/web/20190618114658/https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/209862lbl.pdf Lest 18.6.2019.
- 18.
Neale J, Strang J. Naloxone–does over-antagonism matter? Evidence of iatrogenic harm after emergency treatment of heroin/opioid overdose. Addiction 2015; 110: 1644–52. [PubMed][CrossRef]
- 19.
Tylleskar I, Skulberg AK, Skarra S et al. Pharmacodynamics and arteriovenous difference of intravenous naloxone in healthy volunteers exposed to remifentanil. Eur J Clin Pharmacol 2018; 74: 1547–53. [PubMed][CrossRef]
- 20.
Yassen A, Olofsen E, van Dorp E et al. Mechanism-based pharmacokinetic-pharmacodynamic modelling of the reversal of buprenorphine-induced respiratory depression by naloxone : a study in healthy volunteers. Clin Pharmacokinet 2007; 46: 965–80. [PubMed][CrossRef]
- 21.
Olofsen E, van Dorp E, Teppema L et al. Naloxone reversal of morphine- and morphine-6-glucuronide-induced respiratory depression in healthy volunteers: a mechanism-based pharmacokinetic-pharmacodynamic modeling study. Anesthesiology 2010; 112: 1417–27. [PubMed][CrossRef]