Ulcerations may have a number of causes and can occur as a result of circulatory, traumatic, infectious, malignant and immunological conditions. Our patient had an unusual presentation involving multiple ulcerations of the head and neck region. The cause would prove difficult to determine.
A previously healthy boy in his mid-teens developed a rapidly growing, acne-like lesion in his neck. His GP suspected an abscess and the lesion was lanced. However, it continued to develop into a deep, painful ulceration. In the weeks that followed, several similar lesions appeared that opened spontaneously and exuded copious quantities of pus in the neck, laterally on the throat, preauricularly, and finally on his forehead. All these elements rapidly developed over a short period into intensely painful ulcerations. The patient was assessed by a local dermatologist who took microbial cultures that showed growth of staphylococcus aureus. On suspicion of ecthyma, oral treatment with dicloxacillin was initiated but there was no improvement after one week of treatment. He was admitted to a dermatology ward as an acute case, on suspicion of ecthyma or pyoderma gangrenosum.
In cases of skin and soft tissue infection, the most common agents are streptococci or staphylococcus aureus. Signs of deeper and more serious infections may include severe pain that is inconsistent with clinical findings, rapid progression, degradation of intact skin structure, and production of cutaneous gas. The patient history is essential to uncover potential sources of infection. This will have a bearing on microbiological assessments and treatment (1).
The patient lived on a farm with cats and sheep in the vicinity. He used a helmet sporadically when engaging in leisure activities. The patient had not been travelling, and had no prior joint pain, respiratory symptoms or changes in bowel movement pattern. He had attended school until a few days before admission. There was no hereditary disease in the family.
Upon admission the patient was in good general condition with a pulse of 120 beats/min, blood pressure 130/80 mm Hg, rectal temperature 37.0 °C and respiratory rate 16 breaths/min. His body mass index was 28 kg/m2. Examination of the oral cavity, heart, lungs, abdomen and lower extremities yielded normal findings, and there was no lymphadenopathy in the axillae or groin. The lymph nodes on his neck could not be palpated due to the intense pain.
The boy had multiple, purulent, necrotic ulcerations on his neck, preauricularly, and on his forehead (Figure 1 and 2).
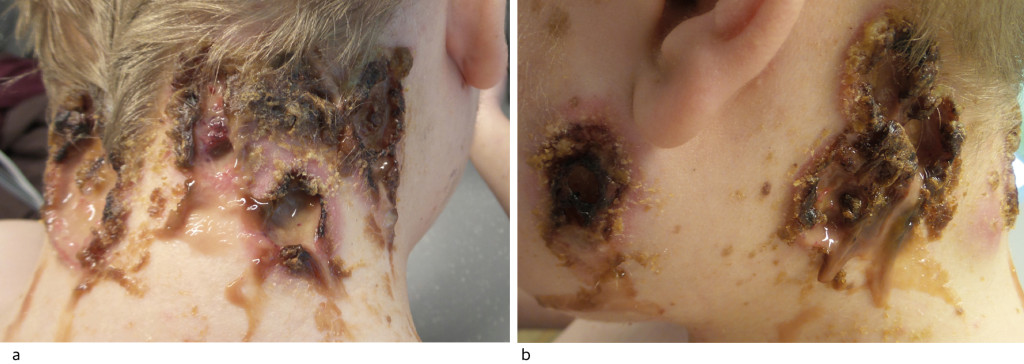
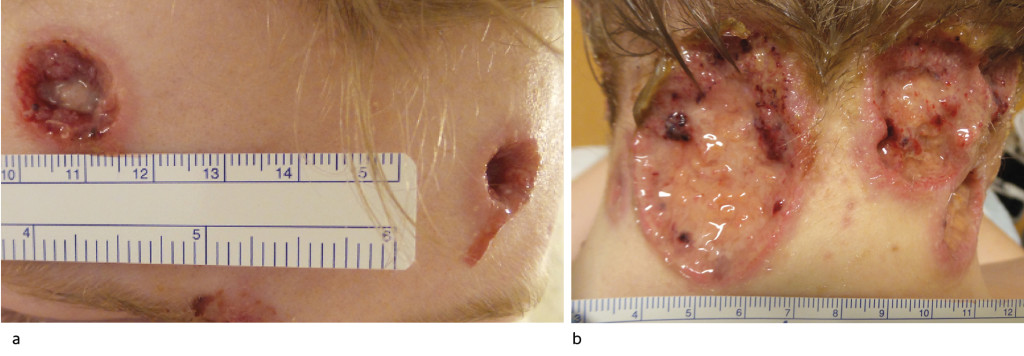
Blood samples showed leukocytes 12.4 ∙ 109/l (reference range 4.4–10.5 ∙ 109/l), neutrophilic granulocytes 9.2 ∙ 109/l (2.1–7.1 ∙ 109/l), monocytes 1.26 ∙ 109/l (0.3–0.6 ∙ 109/l) and CRP 52 mg/l (< 5), but otherwise normal values for thrombocytes, creatinine, ALT and carbamide. Urine dipstick tests showed 2+ for proteins, otherwise normal findings.
The ulcerations had occurred in close proximity to pressure points from the helmet and in areas with acne on the forehead. This gave rise to suspicion of pathergy. Based on the patient history, clinical findings and virtually normal infection parameters, immunological wounds were suspected, most likely with atypical pyoderma gangrenosum.
Pyoderma gangrenosum is a rare, ulcerative dermatological disease of unknown aetiology. The disease belongs to the group of neutrophilic dermatoses, which are histologically associated with neutrophilic infiltration, but with no signs of infection (2). Pyoderma gangrenosum is frequently associated with inflammatory bowel disease, inflammatory arthritis and malignancy. Characteristic of the disease is that it can start or flare up in an area exposed to trauma, so-called pathergy. Pathergy may occur, for example, in a surgical scar, around a stoma or as a result of pressure on the skin. The ulcerative variant of pyoderma gangrenosum often appears first as a pustule. The ulceration is characterised by a purulent and necrotic area centrally, surrounded by a reddish purple, often irregular wound edge with undermining (3). Pyoderma gangrenosum is most often localised to the lower extremities, and only 5 % of cases are described in the head or neck region (4). Patients usually need treatment with immunosuppressive drugs, of which prednisolone is the first choice (3).
The localisation of the ulcerations, as well as the appearance of the wound edges, was atypical for pyoderma gangrenosum. Infection, malignancy and vasculitis were therefore considered as possible differential diagnoses. Bacteriological cultures of pus showed scant growth of normal skin flora consistent with colonisation. There was no growth of Actinomyces or Nocardia, and fungal culture tests were negative. There was no evidence of mycobacteria, and blood culture tests were also negative.
CT of the head, collum, thorax and abdomen showed no deep abscesses or signs of malignancy, but an 8 mm angular apical lesion in the right lung was described as a secondary finding. MRI of the head and collum also revealed no deep abscesses or signs of malignancy, but there was a secondary finding of slight thickening of the mucous membrane in the roof of the left sphenoid sinus. Histological analysis of skin biopsies revealed no signs of vasculitis, granulomatous inflammation or malignancy, but could not exclude the diagnosis of pyoderma gangrenosum.
Treatment with prednisolone 50 mg × 1 was initiated upon admission, and increased to 90 mg × 1 (1 mg/kg) over a period of eight days. The patient received a pain pump, and wound dressings were changed under anaesthesia. Treatment with dicloxacillin was continued. The ulcerations were treated with hydrogel under polyurethane plates; hydrogel was subsequently replaced by tacrolimus ointment 0.1 %. After 14 days of treatment there was good improvement with decreasing inflammation at the wound edges and incipient epithelialisation from the edges. The ulcerations were still painful, and wound dressings needed to be changed under anaesthesia. Test results for anti-neutrophil cytoplasmic antibodies (ANCA) were received concurrently and showed positive for anti-proteinase 3 (PR3-ANCA) 5.1 IU/ml, (reference range < 1).
ANCA is particularly associated with primary vasculitis in small- and medium-sized vessels and is seen in granulomatosis with polyangiitis (previously called Wegener's granulomatosis), microscopic polyangiitis and eosinophilic granulomatosis with polyangiitis (previously called Churg-Strauss syndrome) (5). In patients with ANCA-positive granulomatosis with polyangiitis, 80–90 % have PR3-ANCA (6, 7). However, ANCA may also occur, for example, in infectious, malignant and other autoimmune diseases (8–10).
An interdisciplinary discussion took place as to whether the condition could be associated with granulomatosis with polyangiitis. The patient had no symptoms or findings from the upper or lower respiratory tract that were consistent with this, and creatinine testing and a urine dipstick test as well as urine microscopy did not give rise to suspicion of nephritis. Renal biopsy was therefore not indicated. Nor were there histological findings from the skin biopsies that supported the diagnosis of granulomatosis with polyangiitis. The tentative diagnosis of pyoderma gangrenosum was therefore upheld. The patient continued treatment with high doses of prednisolone and to reduce the risk of potential adverse effects, the treatment was supplemented with infusion of infliximab 5 mg/kg. One week later the pain pump was withdrawn and the prednisolone dose was reduced. When the patient was discharged after six weeks of hospitalisation, the ulcerations were still healing well. The boy was followed up with outpatient check-ups, and intravenous therapy with infliximab every eight weeks was envisaged, with a simultaneous, gradual reduction of the prednisolone dose.
At the check-up one month after discharge, the condition had deteriorated with increasing pain and secretion. The prednisolone dose was therefore increased from 20 to 40 mg daily. He had also developed symptoms of sinusitis and headache. An interdisciplinary discussion again took place regarding the possibility of granulomatosis with polyangiitis. CT of the facial skeleton showed filling in several sinuses, but no signs of skeletal destruction. Biopsy of the respiratory mucosa showed no significant pathological changes. Bacterial sinusitis secondary to immune suppression was suspected. An attempt was made to treat this with dicloxacillin and later with clarithromycin combined with daily saline irrigation, fluticasone furoate and oxymetazoline nasal spray, but the treatment was ineffective. There was still no definitive basis for the diagnosis of granulomatosis with polyangiitis, as the sinusitis symptoms had arisen acutely and because the skin lesions were potentially consistent with pyoderma gangrenosum, both morphologically and according to the biopsy.
Granulomatosis with polyangiitis is a necrotising vasculitis disorder. The aetiology is unknown, and the condition is rare: in a Swedish study, the incidence in children (0–17 years) was 1.4 per million (11). The diagnosis is normally based on clinical findings such as inflammation in the otorhinolaryngeal area, changes in the lower respiratory tract, or nephritis – in combination with elevated inflammation parameters and positive ANCA, and preferably confirmed by a positive biopsy from an affected organ. General symptoms such as fever, fatigue and weight loss are common. Skin manifestations may occur in 14–50 % of cases and are most frequently localised to the lower extremities. The most common cutaneous lesions are palpable purpura, but the clinical features may be polymorphic and include pyoderma gangrenosum-like ulcerations, papules, nodules, vesicles, bullae, petechiae, erythema and livedo reticularis (12–16). Acneiform and folliculitis-like papules have been described in children (17). Oral involvement may occur in the form of ulceration or gingivitis (14, 18, 19). When untreated, granulomatosis with polyangiitis is associated with high mortality. Early diagnosis and treatment are essential to prevent irreversible organ damage (20).
One month later the ulcerations had almost healed. However, in the course of a few days the boy had developed complete loss of vision in the left eye. Orbital MRI revealed filled sinuses contrast enhancement along the sphenoidal plane and assumed involvement of the optic nerve. He underwent surgery with decompression of the nerve, which revealed polypoid tissue in the area in question. Biopsy showed an indication of geographic necrosis, but no obvious vasculitis or signs of granulomas. Pronounced refractory sinusitis in combination with positive ANCA and histology resulted in the patient now receiving a diagnosis of granulomatosis with polyangiitis. The treatment was changed to methylprednisolone and rituximab. He also initially received three experimental doses of cyclophosphamide for more rapid therapeutic effect. The new treatment regime had a beneficial effect on the patient, both with regard to vision and sinusitis symptoms, and the ulcerations healed completely.
Discussion
The pattern of onset of granulomatosis with polyangiitis in our patient was highly unusual. The patient developed multiple, purulent, necrotic ulcerations in the head and neck region which we could best describe as atypical pyoderma gangrenosum. We found through later literature searches that in 1968, Perry and colleagues described a condition with ulcerations which he named malignant pyoderma. This clinical picture was characterised by pyoderma gangrenosum-like ulcerations in the head and neck region or the upper part of the trunk. Preauricular lesions were typically present (21). This is entirely consistent with the findings in our patient. A number of case studies describe malignant pyoderma as a manifestation of granulomatosis with polyangiitis (22–26).
In up to 50 % of cases of granulomatosis with polyangiitis, biopsy from affected skin may reveal leukocytoclastic vasculitis, in some cases granulomatous inflammation, and in rare cases geographic necrosis. Histological examinations may also show non-specific perivascular lymphocytic infiltrates. Direct immunofluorescence of skin biopsy may show deposits of IgM and complement factor C3 around small blood vessels in the dermis (13, 14). In our patient, no typical signs of granulomatosis with polyangiitis were found in biopsies taken from the skin and nasal mucous membrane, only in biopsies from the sinus membrane, which revealed geographic necrosis. A new review of the skin biopsies after the diagnosis of granulomatosis with polyangiitis was made revealed signs of geographic necrosis. This case history is a reminder that differential diagnoses cannot be excluded based on non-specific histological findings. It illustrates that granulomatosis with polyangiitis can be a diagnostic challenge and requires broad interdisciplinary collaboration. The patient is now followed up regularly by a rheumatologist, and the disease is in sustained remission.
The patient and his next of kin have consented to the publication of this article.
- 1.
Stevens DL, Bisno AL, Chambers HF et al. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis 2005; 41: 1373–406. [PubMed][CrossRef]
- 2.
Prat L, Bouaziz JD, Wallach D et al. Neutrophilic dermatoses as systemic diseases. Clin Dermatol 2014; 32: 376–88. [PubMed][CrossRef]
- 3.
Brooklyn T, Dunnill G, Probert C. Diagnosis and treatment of pyoderma gangrenosum. BMJ 2006; 333: 181–4. [PubMed][CrossRef]
- 4.
von den Driesch P. Pyoderma gangrenosum: a report of 44 cases with follow-up. Br J Dermatol 1997; 137: 1000–5. [PubMed][CrossRef]
- 5.
Seo P, Stone JH. The antineutrophil cytoplasmic antibody-associated vasculitides. Am J Med 2004; 117: 39–50. [PubMed][CrossRef]
- 6.
Wegener's Granulomatosis Etanercept Trial Research Group. Limited versus severe Wegener's granulomatosis: baseline data on patients in the Wegener's granulomatosis etanercept trial. Arthritis Rheum 2003; 48: 2299–309. [PubMed][CrossRef]
- 7.
Damoiseaux J, Csernok E, Rasmussen N et al. Detection of antineutrophil cytoplasmic antibodies (ANCAs): a multicentre European Vasculitis Study Group (EUVAS) evaluation of the value of indirect immunofluorescence (IIF) versus antigen-specific immunoassays. Ann Rheum Dis 2017; 76: 647–53. [PubMed][CrossRef]
- 8.
Knight A, Ekbom A, Brandt L et al. What is the significance in routine care of c-ANCA/PR3-ANCA in the absence of systemic vasculitis? A case series. Clin Exp Rheumatol 2008; 26 (suppl 49): S53–6. [PubMed]
- 9.
Mahr A, Batteux F, Tubiana S et al. Brief report: prevalence of antineutrophil cytoplasmic antibodies in infective endocarditis. Arthritis Rheumatol 2014; 66: 1672–7. [PubMed][CrossRef]
- 10.
Hoffman GS, Specks U. Antineutrophil cytoplasmic antibodies. Arthritis Rheum 1998; 41: 1521–37. [PubMed][CrossRef]
- 11.
Mossberg M, Segelmark M, Kahn R et al. Epidemiology of primary systemic vasculitis in children: a population-based study from southern Sweden. Scand J Rheumatol 2018; 47: 295–302. [PubMed][CrossRef]
- 12.
Zycinska K, Wardyn K, Zielonka TM et al. Cutaneous changes: an initial manifestation of pulmonary Wegener's granulomatosis. Adv Exp Med Biol 2013; 755: 307–10. [PubMed][CrossRef]
- 13.
Daoud MS, Gibson LE, DeRemee RA et al. Cutaneous Wegener's granulomatosis: clinical, histopathologic, and immunopathologic features of thirty patients. J Am Acad Dermatol 1994; 31: 605–12. [PubMed][CrossRef]
- 14.
Marzano AV, Raimondo MG, Berti E et al. Cutaneous manifestations of ANCA-associated small vessels vasculitis. Clin Rev Allergy Immunol 2017; 53: 428–38. [PubMed][CrossRef]
- 15.
Comfere NI, Macaron NC, Gibson LE. Cutaneous manifestations of Wegener's granulomatosis: a clinicopathologic study of 17 patients and correlation to antineutrophil cytoplasmic antibody status. J Cutan Pathol 2007; 34: 739–47. [PubMed][CrossRef]
- 16.
Genovese G, Tavecchio S, Berti E et al. Pyoderma gangrenosum-like ulcerations in granulomatosis with polyangiitis: two cases and literature review. Rheumatol Int 2018; 38: 1139–51. [PubMed][CrossRef]
- 17.
Wright AC, Gibson LE, Davis DM. Cutaneous manifestations of pediatric granulomatosis with polyangiitis: a clinicopathologic and immunopathologic analysis. J Am Acad Dermatol 2015; 72: 859–67. [PubMed][CrossRef]
- 18.
Stewart C, Cohen D, Bhattacharyya I et al. Oral manifestations of Wegener's granulomatosis: a report of three cases and a literature review. J Am Dent Assoc 2007; 138: 338–48. [PubMed][CrossRef]
- 19.
Patten SF, Tomecki KJ. Wegener's granulomatosis: cutaneous and oral mucosal disease. J Am Acad Dermatol 1993; 28: 710–8. [PubMed][CrossRef]
- 20.
Mukhtyar C, Flossmann O, Hellmich B et al. Outcomes from studies of antineutrophil cytoplasm antibody associated vasculitis: a systematic review by the European League Against Rheumatism systemic vasculitis task force. Ann Rheum Dis 2008; 67: 1004–10. [PubMed][CrossRef]
- 21.
Perry HO, Winkelmann RK, Muller SA et al. Malignant pyodermas. Arch Dermatol 1968; 98: 561–76. [PubMed][CrossRef]
- 22.
Salassa JR, Winkelmann RK, McDonald TJ. Malignant pyoderma. Otolaryngol Head Neck Surg 1981; 89: 917–22. [PubMed][CrossRef]
- 23.
Gibson LE, Daoud MS, Muller SA et al. Malignant pyodermas revisited. Mayo Clin Proc 1997; 72: 734–6. [PubMed][CrossRef]
- 24.
Mantovani L, Zauli S, Sarno O et al. Treatment of a relapsing facial pyoderma gangrenosum (malignant pyoderma). Int J Dermatol 2013; 52: 753–6. [PubMed][CrossRef]
- 25.
Kędzierska K, Sindrewicz K, Smektała T et al. Wegener's granulomatosis and pyoderma gangrenosum–rare causes of facial ulcerations. Postepy Hig Med Dosw 2016; 70: 210–8. [PubMed][CrossRef]
- 26.
de Boysson H, Martin Silva N, de Moreuil C et al. Neutrophilic dermatoses in antineutrophil cytoplasmic antibody-associated vasculitis: A French multicenter study of 17 cases and literature review. Medicine (Baltimore) 2016; 95: e2957. [PubMed][CrossRef]