Main message
Hospital doctors used broad-spectrum antibiotics more often than guidelines recommend
Doses of penicillin were often higher than those recommended by guidelines
The duration of antibiotic treatment was often longer than recommended by guidelines
Pneumonia is a frequent admission diagnosis in Norwegian hospitals. Incidence, morbidity and mortality have been shown to increase with age and other risk factors, such as comorbidity (1). In cases of pneumonia acquired outside hospital, Streptococcus pneumoniae (S. pneumoniae) is the most frequent agent, followed by Haemophilus influenzae (H. influenzae) (2, 3). In Norway, S. pneumoniae is still sensitive to penicillin, and in the national guidelines for antibiotic treatment of pneumonia acquired outside hospital (referred to in the guidelines and hereafter as 'community-acquired pneumonia'), penicillin is therefore recommended as empirical treatment (4, 5). The guidelines recommend using a scoring tool based on the patient's degree of confusion, respiratory rate, blood pressure and age to assess the severity of pneumonia (the CRB-65 scale; see Box 1). CRB-65 should therefore be preferred over other scoring systems, such as the criteria for systemic inflammatory response syndrome (SIRS), in cases of suspected pneumonia (6).
C: New-onset confusion
R: Respiratory rate ≥ 30 breaths/min
B: Blood pressure < 90 mm Hg systolic or < 60 mm Hg diastolic
65: Age > 65 years
One point is awarded for each criterion
0–2 points: the patient probably has a mild to moderate pneumonia
3–4 points: the patient probably has a severe pneumonia
A guideline is a professional recommendation prepared by experts in a field based on existing research. Guidelines are only recommendations, and are not legally binding, such that one can deviate from a guideline without legal repercussions (7). Nevertheless, the reasons for any decision not to follow the recommendations should be recorded, given that compliance with such guidelines has been shown to yield better treatment outcomes. Narrow-spectrum antibiotics should be used in place of broad-spectrum antibiotics where possible, since the former lead to less development of resistance and fewer effects on the normal bacterial flora (8, 9). Nevertheless, in Norway in recent years, there has been an increase in the prescribing of broad-spectrum antibiotics, despite a relatively low incidence of antibiotic resistance (10). Use of antibiotics in this way drives the development of resistance, which is a major and growing problem both nationally and internationally (11). Compliance with the national guidelines could help to counteract this development.
We wished to investigate compliance with the Norwegian national guidelines for antibiotic treatment of patients hospitalised with community-acquired pneumonia by examining the initial diagnosis and course of antibiotic treatment versus the recommendations in the guidelines.
Material and method
We obtained an overview of patients discharged from the Department of Infectious Diseases at Oslo University Hospital, Ullevål, in the period 1 January 2015 to 31 December 2015 with ICD-10 codes J13 and J14 (pneumonia due to S. pneumoniae and H. influenza, respectively) from the hospital's internal registers. These two diagnostic codes were included because they represent the most frequent pneumonias with known bacterial agents. We did not include patients who were hospitalised with other forms of pneumonia. We chose to disregard diagnostic code J15.9 (bacterial pneumonia, unspecified), for example, because we wished to assess whether treatment was given in accordance with the guidelines following identification of the causative agent and resistance determination. Clinical, demographic and laboratory data were recorded using a standardised form. All information was obtained from DIPS, the patient records system used by Oslo University Hospital.
Permission for data retrieval was granted by the Data Protection Officer at Oslo University Hospital, and all data were anonymised. The first and second authors reviewed all of the medical records together. The last author was consulted in the event of uncertainty.
Results
In 2015, 40 patients were discharged following hospitalisation with pneumonia due to S. pneumoniae and 30 following hospitalisation with pneumonia due to H. influenzae (Table 1). Blood and nasopharyngeal cultures, and PCR of respiratory tract agents, were performed for over 90 % of patients. Of these tests, nasopharyngeal culture most frequently resulted in pathogen growth (80 % of S. pneumoniae and 90 % of H. influenzae). The median duration of hospitalisation was four days (interquartile range three to six days). CRB-65 scores were not reported in any of the medical records, but could nevertheless be calculated based on information in the records for 67 (96 %) patients. In the three remaining patients, where there was not enough information to calculate scores for all criteria, there was nevertheless sufficient information to conclude that the total score was less than 2.
Table 1
Demographic and clinical data for 70 patients admitted to the Department of Infectious Diseases at Ullevål Hospital in 2015 with pneumonia due to S. pneumoniae or H. influenzae. Results are given as percentages unless otherwise stated.
| Variable |
Result |
|
|---|---|---|
| Sex |
||
| Men |
49 |
|
| Women |
51 |
|
| Age in years, median (interquartile range) |
68 (47–82) |
|
| Multiple diagnoses in medical records |
57 |
|
| CRB-65 score |
||
| 0 |
37 |
|
| 1 |
31 |
|
| 2 |
20 |
|
| 3 |
7 |
|
| 4 |
0 |
|
| Incomplete1 |
4 |
|
| Culture results |
||
| S. pneumoniae |
57 |
|
| H. influenzae |
43 |
|
| Duration of hospitalisation in days, median (interquartile range) |
10 (8–11) |
|
1In these three patients, there was sufficient information to conclude that the CRB-65 score was ≤ 2.
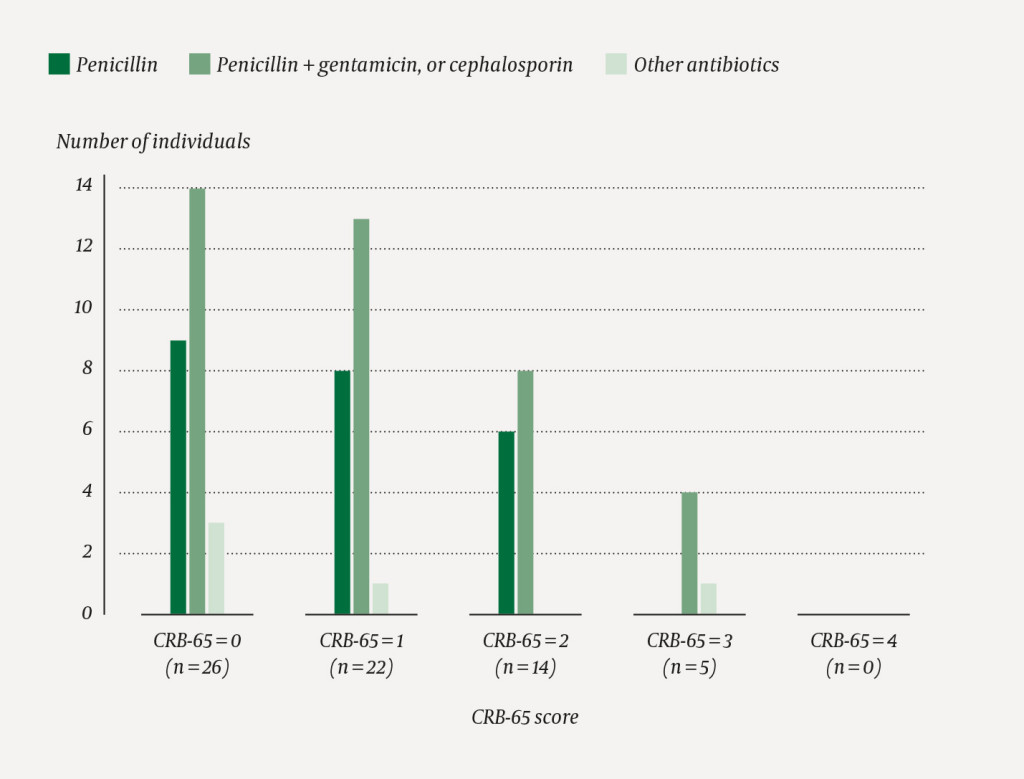
Figure 1 shows the antibiotics that patients received upon admission alongside the calculated CRB-65 scores. Penicillin monotherapy was given to 24 (34 %) patients. Of these, 15 patients received 3 g × 4 intravenously and eight patients 1.2 g × 4 intravenously, while one patient received 1 g × 4 per os. Penicillin in combination with gentamicin was given to 25 (36 %) patients, of whom 21 received 3 g × 4 intravenously. Only one patient who received combination therapy with penicillin and gentamicin had a CRB-65 score of 3 or above and thus an indication for this treatment. One patient received penicillin in combination with another antibiotic. Of the 50 patients who received penicillin as monotherapy or in combination therapy, 38 (76 %) received a dose that was too high relative to their disease severity according to the CRB-65 criteria. Of these, 25 had their dose reduced while in hospital, 18 of them upon discharge. Cephalosporin was given to 14 (20 %) patients. Of these, seven had an indication for this treatment, either a penicillin allergy or a CRB-65 score of 3 or above. Seven (10 %) patients received other antibiotics, of whom six had an appropriate indication in the form of an allergy or specific clinical issues. In total, 38 (54 %) patients received initial antibiotic treatment in accordance with the recommendations in the guidelines (Figure 2).
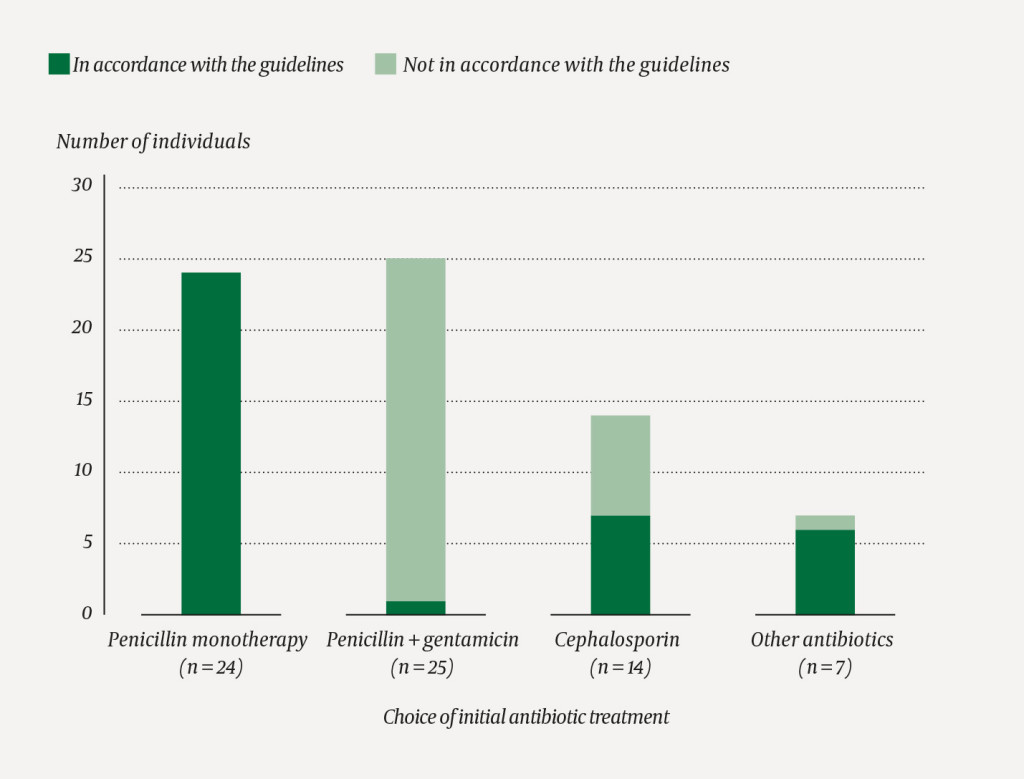
In 38 out of 40 patients with pneumococcal infection, resistance determination revealed that the bacteria were sensitive to penicillin. One patient had resistant bacteria and one had bacteria with decreased sensitivity. Thirty-six of the patients had their antibiotic therapy adjusted in line with the recommendations in the guidelines once the test results were available, including the two patients with resistant bacteria. One patient could not be assessed because of missing information in the medical records.
In 12 out of 30 patients with H. influenzae infection, the bacteria were resistant to amoxicillin and ampicillin. Twenty-six of the patients had their antibiotic therapy adjusted in line with the recommendations in the guidelines once the test results were available. Two patients could not be assessed owing to missing information in the medical records or a complicated disease course. Of the patients shown to have resistant H. influenzae, ten received ciprofloxacin, trimethoprim/sulfamethoxazole or doxycycline in accordance with the resistance determination, while one patient remained on his/her initial treatment and one patient received no treatment.
Discussion
A key finding was that many patients received treatment with excessively broad-spectrum antibiotics, particularly gentamicin and cephalosporins, relative to the severity of their pneumonia as judged using the CRB-65 criteria, and the recommendations in the national guidelines. In comparison, most patients fulfilled the criteria for SIRS; according to the medical records, this was often used to justify empirical treatment. The CRB-65 criteria have been shown to be superior to SIRS for identifying patients who have pneumonia with low mortality risk, and who can thus be treated outside hospital. However, there is debate as to whether patients with low CRB-65 scores but with hypoxaemia or high levels of comorbidity should be considered for admission, and whether the CRB-65 criteria may not therefore be optimal for assessing severity and prognosis (12, 13). Other studies have compared the CRB-65 criteria with SIRS as well as the new sepsis criteria, 'quick sepsis-related organ failure assessment' (qSOFA) (14). They concluded that CRB-65 and qSOFA are better than SIRS for initial assessment of patients with pneumonia, and that CRB-65 and qSOFA are equally good for assessing severity, but that the CRB-65 criteria are better than qSOFA for predicting mortality at initial assessment (15, 16).
CRB-65 scores were not specified in any of the medical records that we reviewed. We interpret this as indicating that few were familiar with the rating system. The qSOFA criteria were not included in the national guidelines and were therefore not applied. However, it is possible that our findings with respect to empirical antibiotic treatment may have been different had the qSOFA criteria been in use in 2015, and that these criteria could in future help ensure that more patients receive empirical treatment in accordance with the recommendations in the national guidelines.
The use of gentamicin and cephalosporins should be limited for several reasons, but may be justified in cases of suspected sepsis or severe pneumonia. Gentamicin should then be given in preference to cephalosporins since gentamicin has a faster onset of bactericidal action and results in less development of resistance (17). Doctors were, however, good at discontinuing gentamicin after 1–2 doses in our patients.
For H. influenzae, resistance rates to amoxicillin and ampicillin were more than twice as high as those in the latest report from NORM (Norwegian Surveillance System for Antimicrobial Drug Resistance), and 21 (70 %) patients with H. influenzae pneumonia did not receive covering empirical treatment (5). This may indicate a need to consider adjusting the guidelines for treatment of H. influenzae based on the local pattern of resistance. On the other hand, most pneumonias caused by H. influenzae are mild and the bacterium rarely passes into the bloodstream (18). None of our patients died.
According to the CRB-65 criteria, 38 (76 %) patients who received penicillin received too high a dose. Many of them did not have their dose reduced; of those who did, the adjustment was usually carried out only upon discharge. This is consistent with the results of an intervention study by Høgli et al. (19), in which penicillin doses were reduced by 10 % without compromising treatment outcomes. The bactericidal ability of beta-lactam antibiotics depends on the length of time for which their concentration is above the minimum inhibitory concentration (MIC) and not on the peak concentration (20). To achieve sufficient bactericidal efficacy, the concentration should be above the MIC more than 50 % of the time, and penicillin should ideally be taken four times a day. Increasing the dosage of penicillin in cases of pneumonia does not usually increase bactericidal efficacy (8, 20). There should therefore be greater emphasis on selecting the correct dosage to avoid unnecessary ecological and toxic adverse effects (8).
Of the patients in our study, 62 (89 %) had their treatment changed to narrower spectrum antibiotics once the microbiological agent and its resistance status had been determined. This shows that the testing was followed up, and that efforts were made to provide treatment that was as narrow-spectrum and targeted as possible when the agent was known and the patient's clinical condition allowed. This would seem to suggest a good level of awareness and knowledge of these issues among doctors at the department.
Eight (11 %) patients stated that they had a penicillin allergy. This figure is consistent with several other studies (19, 21). However, the actual prevalence of severe penicillin allergy (type 1) is thought to be less than 0.05 % (22). We may therefore assume that our figures do not reflect reality, but this is difficult to judge as the reaction was not described adequately in the medical records. We call for greater emphasis on the overdiagnosis of penicillin allergy to avoid unnecessary use of broad-spectrum antibiotics (21, 23).
Treatment duration was not in accordance with the recommendations in the guidelines. This is consistent with the results of the intervention study by Høgli et al., in which treatment duration was successfully reduced by one day (19). However, this reduction proved to be short-lived. Høgli et al. commented that the doctors responsible for the treatment did not feel comfortable shortening the treatment course and did not appear sufficiently up-to-date regarding the latest recommendations on treatment duration. We believe that these factors may also have contributed to our finding of excessive treatment duration. Three-day treatment for mild to moderate pneumonia and five-day treatment for severe pneumonia have been shown to be non-inferior to treatment for seven to ten days in terms of clinical success rate, symptoms and mortality up to 30 days post-discharge, provided that patients show a clinical response (24, 25). In addition, short-course antibiotic treatment offers benefits in terms of reduced development of resistance, reduced costs and increased patient compliance (26). The use of new biomarkers such as procalcitonin has also been shown to help shorten pneumonia treatment by several days, but these are not yet in use by our department (27). Our findings of incomplete compliance with the guidelines in terms of risk assessment, dosage and duration of antibiotic treatment, as well as overdiagnosis of penicillin allergy, illustrate that it often takes a long time for findings from research to be implemented in clinical practice, and that there is a need for greater focus on adopting the latest evidence-based practices to ensure that patients receive the best possible treatment (28).
Strengths and weaknesses of the study
Our findings are based on retrospective evaluation of medical records rather than on clinical examination of the patients. We cannot conclude for certain therefore that patients were treated with excessively broad-spectrum antibiotics. Our patient dataset is also relatively small. However, the dataset does reflect the realities of everyday clinical practice in a hospital department and the daily challenges facing all hospitals. We did not include patients with unspecified pneumonia. Had we included these patients, the observed effects of antibiotic treatment may have been different.
Conclusion
Compliance with national guidelines on antibiotic treatment is important to give patients the best possible treatment and to avoid the development of antimicrobial resistance. Our findings suggest a need for greater emphasis on thorough initial assessment of pneumonia severity in order to avoid unnecessary use of broad-spectrum antibiotics, as well as for increased focus on dosage and treatment duration.
- 1.
Welte T, Torres A, Nathwani D. Clinical and economic burden of community-acquired pneumonia among adults in Europe. Thorax 2012; 67: 71 - 9. [PubMed][CrossRef]
- 2.
Holter JC, Müller F, Bjørang O et al. Etiology of community-acquired pneumonia and diagnostic yields of microbiological methods: a 3-year prospective study in Norway. BMC Infect Dis 2015; 15: 64. [PubMed][CrossRef]
- 3.
Røysted W, Simonsen Ø, Jenkins A et al. Aetiology and risk factors of community-acquired pneumonia in hospitalized patients in Norway. Clin Respir J 2016; 10: 756 - 64. [PubMed][CrossRef]
- 4.
Nasjonal faglig retningslinje for bruk av antibiotika i sykehus. Oslo: Helsedirektoratet, 2016. https://helsedirektoratet.no/Retningslinjer/Antibiotika%20i%20sykehus.pdf (4.10.2018).
- 5.
NORM/NORM-VET. 2016. Usage of antimicrobial agents and occurrence of antimicrobial resistance in Norway. Tromsø/Oslo:NORM/NORM-VET, 2017. https://unn.no/Documents/Kompetansetjenester,%20-sentre%20og%20fagr%C3%A5d/NORM%20%20Norsk%20overv%C3%A5kingssystem%20for%20antibiotikaresistens%20hos%20mikrober/Rapporter/NORM%20NORM-VET%202016.pdf (4.10.2018).
- 6.
Lim WS, van der Eerden MM, Laing R et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax 2003; 58: 377 - 82. [PubMed][CrossRef]
- 7.
Helsedirektoratet. Metoder og verktøy for utarbeiding av nasjonale faglige retningslinjer. https://helsedirektoratet.no/metoderog-verktoy(4.10.2018).
- 8.
Berild D, Haug JB. Fornuftig bruk av antibiotika i sykehus. Tidsskr Nor Laegeforen 2008; 128: 2335 - 9. [PubMed]
- 9.
McCabe C, Kirchner C, Zhang H et al. Guideline-concordant therapy and reduced mortality and length of stay in adults with community-acquired pneumonia: playing by the rules. Arch Intern Med 2009; 169: 1525 - 31. [PubMed][CrossRef]
- 10.
Haug JB, Berild D, Walberg M et al. Increased antibiotic use in Norwegian hospitals despite a low antibiotic resistance rate. J Antimicrob Chemother 2011; 66: 2643 - 6. [PubMed][CrossRef]
- 11.
Laxminarayan R, Duse A, Wattal C et al. Antibiotic resistance-the need for global solutions. Lancet Infect Dis 2013; 13: 1057 - 98. [PubMed][CrossRef]
- 12.
CAPNETZ Study Group. CRB-65 predicts death from community-acquired pneumonia. J Intern Med 2006; 260: 93 - 101. [PubMed][CrossRef]
- 13.
Dwyer R, Hedlund J, Henriques-Normark B et al. Improvement of CRB-65 as a prognostic tool in adult patients with community-acquired pneumonia. BMJ Open Respir Res 2014; 1: e000038. [PubMed][CrossRef]
- 14.
Singer M, Deutschman CS, Seymour CW et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016; 315: 801 - 10. [PubMed][CrossRef]
- 15.
CAPNETZ Study Group. Comparison of the qSOFA and CRB-65 for risk prediction in patients with community-acquired pneumonia. Intensive Care Med 2016; 42: 2108 - 10. [PubMed][CrossRef]
- 16.
Ranzani OT, Prina E, Menéndez R et al. New sepsis definition (sepsis-3) and community-acquired pneumonia mortality. A validation and clinical decision-making study. Am J Respir Crit Care Med 2017; 196: 1287 - 97. [PubMed][CrossRef]
- 17.
Lindemann PC, Haldorsen BC, Smith I et al. Aminoglykosider bør fortsatt brukes i empirisk sepsisbehandling. Tidsskr Nor Legeforen 2013; 133: 1054 - 5. [PubMed][CrossRef]
- 18.
European Centre for Disease Prevention and Control Country Experts for Invasive Haemophilus influenzae Disease. Epidemiology of invasive haemophilus influenzae disease, Europe, 2007–2014. Emerg Infect Dis 2017; 23: 396 - 404. [PubMed][CrossRef]
- 19.
Høgli JU, Garcia BH, Skjold F et al. An audit and feedback intervention study increased adherence to antibiotic prescribing guidelines at a Norwegian hospital. BMC Infect Dis 2016; 16: 96. [PubMed][CrossRef]
- 20.
Andes D, Craig WA. Animal model pharmacokinetics and pharmacodynamics: a critical review. Int J Antimicrob Agents 2002; 19: 261 - 8. [PubMed][CrossRef]
- 21.
Lee CE, Zembower TR, Fotis MA et al. The incidence of antimicrobial allergies in hospitalized patients: implications regarding prescribing patterns and emerging bacterial resistance. Arch Intern Med 2000; 160: 2819 - 22. [PubMed][CrossRef]
- 22.
Park MA, Li JT. Diagnosis and management of penicillin allergy. Mayo Clin Proc 2005; 80: 405 - 10. [PubMed][CrossRef]
- 23.
Borch JE, Andersen KE, Bindslev-Jensen C. The prevalence of suspected and challenge-verified penicillin allergy in a university hospital population. Basic Clin Pharmacol Toxicol 2006; 98: 357 - 62. [PubMed][CrossRef]
- 24.
Uranga A, España PP, Bilbao A et al. Duration of antibiotic treatment in community-acquired pneumonia: a multicenter randomized clinical trial. JAMA Intern Med 2016; 176: 1257 - 65. [PubMed][CrossRef]
- 25.
el Moussaoui R, de Borgie CA, van den Broek P et al. Effectiveness of discontinuing antibiotic treatment after three days versus eight days in mild to moderate-severe community acquired pneumonia: randomised, double blind study. BMJ 2006; 332: 1355. [PubMed][CrossRef]
- 26.
Mandell LA, File TM. Short-course treatment of community-acquired pneumonia. Clin Infect Dis 2003; 37: 761 - 3. [PubMed][CrossRef]
- 27.
Schuetz P, Müller B, Christ-Crain M et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev 2012; 9: CD007498. [PubMed]
- 28.
Grimshaw JM, Eccles MP, Lavis JN et al. Knowledge translation of research findings. Implement Sci 2012; 7: 50. [PubMed][CrossRef]