Fertility after cryopreservation of ovarian tissue due to cancer treatment
MAIN POINTS
Premature ovarian failure is a possible complication of cancer treatment
The probability of premature ovarian failure depends on the patient's cancer diagnosis, treatment and age
Fertility preserving measures should always be considered prior to cancer treatment for prepubescent girls and women younger than 35 years
Advances in cancer treatment have led to a considerably higher survival rate, including for prepubescent girls and women of reproductive age (1). It is therefore important to take future quality of life into consideration, including future fertility.
Cancer therapy may entail autologous or allogeneic stem cell transplantation, aggressive use of chemotherapy, radiotherapy or surgery. The ovaries are highly sensitive to these treatment regimes, and one of the adverse effects in prepubescent girls and women of reproductive age may be premature ovarian failure, defined as loss of function of the ovaries before the age of 40 (2).
Spontaneous premature ovarian failure occurs in approximately 1 % of women before the age of 40, and in 0.1 % before they turn 30 (2). The risk of reduced ovarian function will depend on the type of chemotherapy and total dose of irradiation to which pelvic organs and hypophysis are subjected (3).
Fertility can be maintained by cryopreserving embryos, oocytes or ovarian tissue (4). Assisted reproduction treatment, including cryopreservation of embryos, is regulated by the Norwegian Act relating to the application of biotechnology in human medicine, etc. (the Biotechnology Act), which requires that the patient must be in a stable cohabitational relationship or married. Cryopreserved embryos can be stored for a maximum of five years (5), which is a disadvantage if the patient is to undergo prolonged cancer treatment. The five-year storage limit and cohabitation requirements do not apply to cryopreservation of oocytes or ovarian tissue (5).
Cryopreservation of oocytes or embryos is not an option if the patient has not reached puberty, if there is not sufficient time to mature oocytes prior to cancer treatment, or if harvesting of oocytes is regarded as too great a risk for the patient because of the danger of infection, bleeding or spread of cancer cells. Cryopreservation of ovarian tissue is therefore the only fertility preservation option for some patient groups.
Cryopreservation of ovarian tissue has been approved in Norway as a means of preserving fertility since 2004, and is performed by the Norwegian National Unit for Fertility Preservation with Autologous Ovarian Tissue, Oslo University Hospital. Laparoscopy is used to harvest ovarian tissue by removing one ovary or resectates from one or both ovaries. This is done without postponement of the cancer treatment.
If a woman is in remission after completing cancer treatment and experiences premature ovarian failure, the ovarian tissue can be retransplanted, usually in the remaining ovary or, alternatively, under the peritoneum. If ovarian function resumes, the woman can become pregnant, either spontaneously or through assisted reproduction. From 2004 until the end of 2014, cryopreservation of ovarian tissue was performed for 164 women with benign or malignant primary disorders. During the same period, six women had applied to have their cryopreserved tissue reimplanted. Two of them had retransplantation approved and carried out (6).
In a Danish study, 22 % of the patients who had ovarian tissue cryopreserved in connection with cancer treatment (7) suffered premature ovarian failure. Because the indication for offering fertility-preserving treatment is more liberal in Denmark than in Norway (8), and no-one had previously reviewed the Norwegian material, we wanted to conduct a similar study of women in Norway with a malignant primary disorder who had ovarian tissue cryopreserved. We wanted to find out how many of these women had suffered premature ovarian failure, and whether they wanted to use the cryopreserved tissue should it prove necessary.
We hoped that the results of this study would enable us to improve our recommendations to future patients with respect to fertility-preserving measures prior to cancer therapy.
Material and method
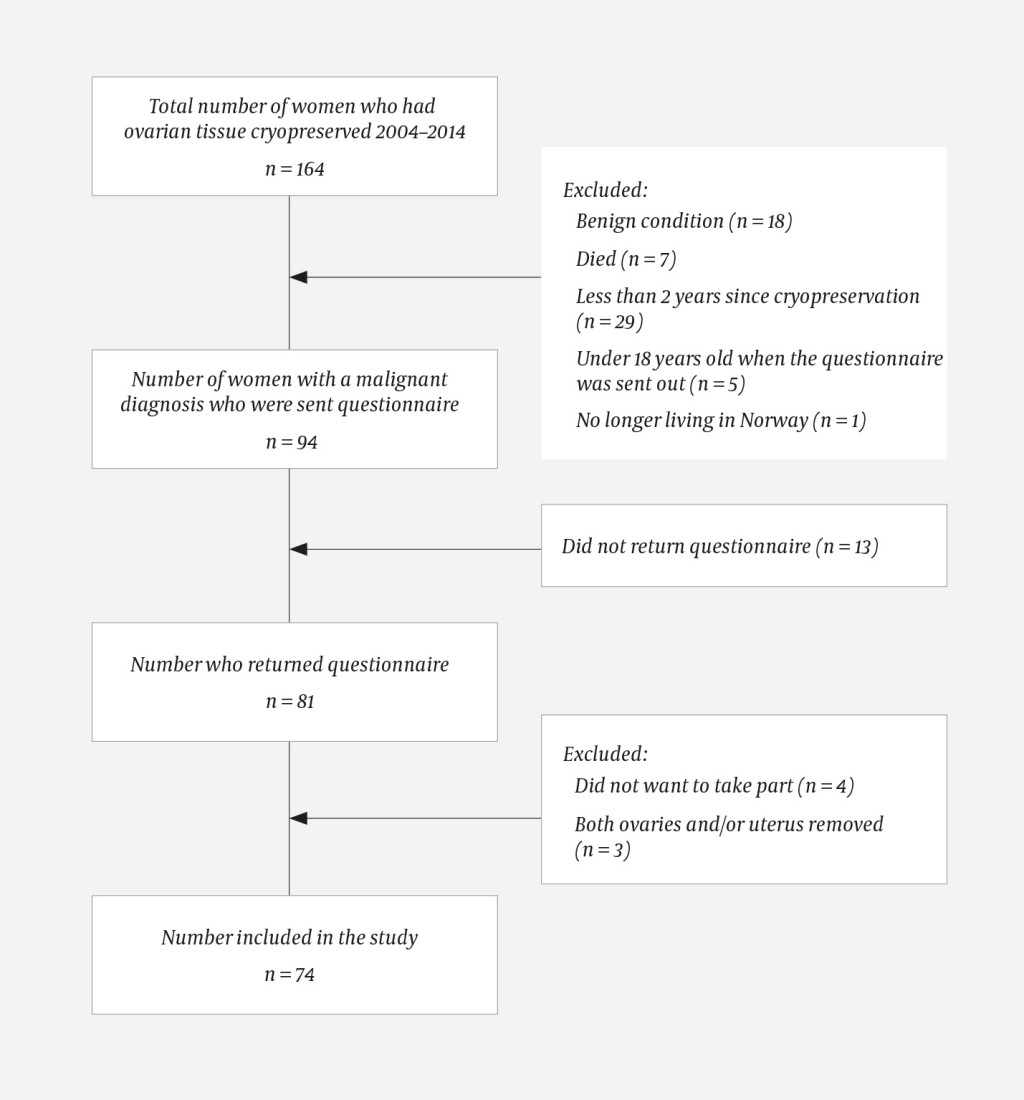
In the period 2004–2014, a total of 164 women had ovarian tissue cryopreserved, 146 of them due to a malignant condition and 18 of them due to a non-malignant condition. In December 2014, women aged over 18 living in Norway who had ovarian tissue cryopreserved as a result of a malignant condition, and a cryopreservation period of more than two years, were sent a questionnaire with an invitation to take part in the study. Of the 94 women who were asked, 74 who had cryopreservation in the period 2004–2012 were included in the study (Fig. 1). The questionnaire was an edited version of a similar form used for the same patient group that was developed at the Rigshospitalet in Copenhagen, Denmark (7). Information regarding the indication for cryopreservation, and whether a whole ovary was removed or resectates from one or both ovaries, was obtained from the patient's answers and from electronic medical records. Approval was given by the Data Protection Officer at Oslo University Hospital (case no. 2014/6637).
Results
Table 1 shows diagnosis, number with premature ovarian failure, age at the time of cryopreservation and at the time of sending out the questionnaire. Premature ovarian failure was defined as the woman answering 'Yes' to the question of amenorrhoea occurring before the age of 40 without another, concurrent reason to explain the lack of menstruation.
Table 1
Patients over the age of 18 with more than two years of follow-up after cryopreservation of ovarian tissue as a means of preserving fertility due to treatment for a malignant condition in the period 2004–2012 (n = 74)
| Diagnosis | Number (n) | Number with premature ovarian failure (n) | Median age at time of cryopreservation | Median age when questionnaire was sent out |
|---|---|---|---|---|
| Breast cancer | 38 | 2 | 30 (22–36) | 35 (26–43) |
| Lymphoma | 22 | 10 | 19 (14–35) | 25 (19–43) |
| Sarcoma | 7 | 3 | 16 (15–27) | 21 (18–31) |
| Leukaemia | 4 | 3 | 24 (22–29) | 32 (28–39) |
| Colorectal cancer | 3 | 2 | 33 (26–35) | 41 (33–42) |
A total of 20 of 74 women (27 %) had developed premature ovarian failure. The proportion was highest among women with leukaemia and colorectal cancer and lowest among those with breast cancer.
As part of their fertility-preserving treatment, 43 of 74 (58 %) had a whole ovary removed for cryopreservation, while ovarian biopsies had been taken from one or both ovaries in the remaining 31 women (42 %). Premature ovarian failure occurred in 16 of the 43 who had a whole ovary removed, compared with four of the 31 with ovarian biopsies.
Patients with breast cancer were evenly divided between the two methods: 18 had one ovary removed, while 20 had ovarian biopsies. Of the 29 lymphoma and sarcoma patients, 21 had one ovary cryopreserved and eight had biopsies.
Breast cancer
Two of the 38 breast cancer patients reported premature ovarian failure after cytotoxic treatment. A further three women were classified as uncertain because they were still being treated with antioestrogenic agents. Sixteen (42 %) of the women had been pregnant before treatment, and eight of them had borne children. Fourteen (37 %) of the women achieved pregnancy after treatment, and had given birth to a total of 18 children. Ten of the 14 women responded that they had achieved spontaneous pregnancy in the course of the first year of wanting a child, and two had used assisted reproduction treatment. Of the 24 women who had not become pregnant after treatment, ten reported that they were still receiving antioestrogenic therapy, four had been advised against pregnancy or were themselves uncertain about the risk associated with pregnancy, two had relapses and were in poor health, five had no partner or no active desire for a child. As indicated above, two women had premature ovarian failure and one did not answer the question. Four of the patients with breast cancer did not want children and they did not want to use the frozen tissue. They were generally older; three of the four were over 40 years old.
Lymphoma
Of 22 patients with lymphoma, seven of them with non-Hodgkin's lymphoma and 15 with Hodgkin's lymphoma, ten suffered premature ovarian failure. Seven of them received high-dose therapy and autologous stem cell transplant (HMAS), one received an allogeneic stem cell transplant (bone marrow transplant), one received a total of 20 Gy radiotherapy below the diaphragm, and one patient aged 35 developed ovarian failure after chemotherapy alone. Of a total of eight patients who received an autologous stem cell transplant, only one did not report premature ovarian failure.
Five patients had been pregnant before treatment, and two of them had given birth. Seven became pregnant after treatment: two after retransplantation of ovarian tissue, one after in-vitro fertilisation, one after egg donation and three spontaneously. Of the two women who underwent retransplantation of ovarian tissue, one fell pregnant spontaneously, and the other became pregnant with the aid of in-vitro fertilisation.
Seventeen women reported that they had either an active or a future desire for children, and 19 would use their ovarian tissue if necessary. One was uncertain. Both the women who did not want to use the stored tissue had given birth after completing treatment. Of the 15 who did not have an active desire for children, lack of a partner or that the time was not right were cited as reasons for the absence of pregnancy. Three women wanted children, but had not achieved this yet.
Sarcoma
Three of the seven women treated for sarcoma reported premature ovarian failure. These three had all received an autologous stem cell transplant, and one had additionally received a total of 50 Gy of pelvic irradiation. None of them had been pregnant either before or after treatment. Reasons given were the lack of a partner, or that that they were too young. Six wanted children in the future. All of them would want to use frozen ovarian tissue if necessary to achieve pregnancy.
Leukaemia
Of the four women treated for leukaemia, three had premature ovarian failure. They had all undergone allogeneic stem cell transplantation, in one case combined with total body irradiation. All reported a desire for children in the future and a desire to use the ovarian tissue. One woman had two children after completing treatment. She had not had an allogeneic stem cell transplant.
Colorectal cancer
Two of the three women treated for colorectal cancer had premature ovarian failure after chemotherapy and a total of 50 Gy pelvic irradiation. Neither had been pregnant before or after treatment, but wanted to use the ovarian tissue if necessary.
Discussion
In this study, the incidence of premature ovarian failure was highest in women treated for leukaemia, colon cancer and lymphoma. Those at greatest risk had undergone allogeneic stem cell transplantation, autologous stem cell transplantation and/or total body irradiation, abdomen or pelvis. This corresponds with previously published studies (9–11).
There was lowest risk of premature ovarian failure after treatment for breast cancer, but because many women with breast cancer were still receiving treatment with antioestrogenic agents, it was difficult to assess ovarian function before the treatment had been completed. The breast cancer group had the highest fertility both before and after treatment. A large proportion of the pregnancies, particularly in women treated for lymphoma, occurred after infertility treatment with retransplantation of ovarian tissue, assisted reproduction and the use of egg donation. Our results concerning premature ovarian failure are largely consistent with a previously published Danish study (7) and a similar Belgian follow-up study, in which breast cancer patients were at lower risk of premature ovarian failure than patients with haematological cancer: 11 % versus 34.5 % (12).
The probability of premature ovarian failure was highest in the group that had a whole ovary frozen as part of fertility-preserving treatment. The method was used more often for women who had autologous or allogeneic stem cell transplantation and pelvic radiotherapy.
We therefore believe that premature ovarian failure depends on the type of gonadotoxic treatment administered, and not on the operating procedure that is used.
Laparoscopic unilateral oophorectomy is currently used at Oslo University Hospital as standard procedure for reducing the risk of inappropriate biopsies and to avoid scar tissue in the remaining or residual ovary that makes retransplantation difficult. The procedure can take place at the patient's own regional hospital, and the tissue is then sent to Oslo University Hospital for further processing (13).
The first child born following a pregnancy achieved after retransplantation of ovarian tissue was born in Belgium in 2004 (14). The literature describes almost 100 births achieved by this method (15). Cryopreservation of ovarian tissue is still regarded as an experimental treatment. The probability of premature ovarian failure after treatment should be high before it is offered, in light of the fact that many patients preserve their fertility after undergoing treatment for cancer.
The Norwegian guidelines recommend a risk of infertility of about 50 %, as assessed by the referring oncologist or haematologist (16). The upper age limit for cryopreservation of ovarian tissue in Norway is 35 years, because the number and quality of eggs diminishes with increasing age (16). There is no lower limit, so this is a potential method for both children and adolescents. Malignant cells can survive the freeze-thaw process, and there is therefore a risk of reintroducing the disease through retransplantation. Metastasis of lymphoma and breast cancer to the ovaries is rare (17), while the risk is particularly high for leukaemia (18). Leukaemia patients are therefore not offered retransplantation today. Nonetheless, cryopreservation of ovarian tissue is offered to this patient group today in the expectation of a future option of treatment, but when, or indeed whether, this will be available is still uncertain. If cryopreservation is offered in cases of disseminated disease, the upper age limit is currently set at 25 years (16).
According to the current Norwegian Biotechnology Act, storage of ovarian tissue and unfertilised eggs can only be offered if the patient has to undergo treatment that may harm her fertility (5). Because treatment regimes are constantly changing, this may be difficult to predict. Girls and young women retain their menstruation and fertility to a greater extent than women over the age of 30, but they too enter menopause earlier than expected (11).
The disadvantage of removing up to half of the egg reserves of girls and young women through cryopreservation of ovarian tissue must be taken into consideration. On average, women from whom one ovary is removed enter menopause one year earlier than women with two ovaries (19). What the situation is for those who have additionally undergone gonadotoxic therapy, we do not know. Since egg donation is still not permitted in Norway, it is conceivable that a number of the women will request fertility-preserving measures despite the low risk of ovarian failure.
The risk of premature ovarian failure following treatment for breast cancer appears to be lower than previously assumed (7, 12), which our results also support. Cryopreservation of ovarian tissue is therefore currently offered to breast cancer patients aged between 30 and 35 (20). Cryopreservation of oocytes and/or embryos is recommended for patients below the age of 30 (20). This is viewed as a less radical intervention for the patient and, in contrast to cryopreservation of ovarian tissue, is not an experimental treatment. Cryopreservation of oocytes is predominantly used in connection with egg donation, and so far there are few reports of children born by this method to cancer patients (21).
Cryopreservation of embryos is a daily routine at the laboratory in connection with ordinary in-vitro fertilisation. The disadvantages of cryopreserving oocytes and/or embryos are that this, as with ordinary in-vitro fertilisation, entails 7–14 days of hormone stimulation of the ovaries, harvesting of oocytes and that the patient share the cost of drugs. Demand for cryopreservation of ovarian tissue and oocytes is growing rapidly, not only for cancer patients, but also for patients with non-malignant diseases such as multiple sclerosis and rheumatic diseases who are to be treated with cytostatics, radiotherapy or stem cell transplantation.
There are no data in Norway on how many patients have been offered fertility-preserving treatment, or how many have declined the offer of this type of treatment. On the basis of our survey, we cannot make any pronouncements about the post-treatment fertility of cancer patients generally, only regarding those patients who have had ovarian tissue cryopreserved. However, the percentage of responses in our study was high, strengthening the results for our patient population.
Summary
The greatest risk of premature ovarian failure was found in patient groups who had undergone autologous or allogeneic stem cell transplantation and total body irradiation and/or abdomen and pelvis. These women should still be offered cryopreservation of ovarian tissue.
The breast cancer patients in our study seldom experienced premature ovarian failure, and most of those who tried achieved pregnancy without the use of infertility treatment. We recommend nonetheless that fertility counselling be offered to all women of reproductive age in order to determine which fertility preserving measures can be recommended for the individual.
Most of those who had cryopreserved ovarian tissue wanted the possibility of retransplantation should this prove necessary to achieve pregnancy, irrespective of whether they had been pregnant previously or not.
We would like to thank Dr Kirsten Tryde Macklon, senior consultant at the Fertility Clinic, Rigshospitalet, Copenhagen, for permission to use the Danish questionnaire in our survey.
- 1.
Kreftregisteret. Kreftstatistikk. https://www.kreftregisteret.no/Registrene/Kreftstatistikk/ (18.2.2018).
- 2.
Rebar RW. Premature ovarian failure. Obstet Gynecol 2009; 113: 1355 - 63. [PubMed][CrossRef]
- 3.
American Society of Clinical Oncology. Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 2013; 31: 2500 - 10. [PubMed][CrossRef]
- 4.
Storeng R, Åbyholm T, Tanbo T. Kryopreservering av ovarialvev. Tidsskr Nor Laegeforen 2007; 127: 1045 - 8. [PubMed]
- 5.
Lov om humanmedisinsk bruk av bioteknologi m.m. (bioteknologiloven) https://lovdata.no/dokument/NL/lov/2003-12-05-100? q=bioteknologiloven(27.4.2017).
- 6.
Tanbo T, Greggains G, Storeng R et al. Autotransplantation of cryopreserved ovarian tissue after treatment for malignant disease – the first Norwegian results. Acta Obstet Gynecol Scand 2015; 94: 937 - 41. [PubMed][CrossRef]
- 7.
Schmidt KT, Nyboe Andersen A, Greve T et al. Fertility in cancer patients after cryopreservation of one ovary. Reprod Biomed Online 2013; 26: 272 - 9. [PubMed][CrossRef]
- 8.
Dansk Fertilitetsselskab, Fertilitetsbevaring ved malign sykdom. http://www.fertilitetsselskab.dk/images/2015_dok/guideline/guideline%20dfs%20fertilitetsbevaring%20150315.pdf (27.4.2017).
- 9.
Barrett A, Nicholls J, Gibson B. Late effects of total body irradiation. Radiother Oncol 1987; 9: 131 - 5. [PubMed][CrossRef]
- 10.
Locatelli F, Giorgiani G, Pession A et al. Late effects in children after bone marrow transplantation: a review. Haematologica 1993; 78: 319 - 28. [PubMed]
- 11.
Larsen EC, Müller J, Schmiegelow K et al. Reduced ovarian function in long-term survivors of radiation- and chemotherapy-treated childhood cancer. J Clin Endocrinol Metab 2003; 88: 5307 - 14. [PubMed][CrossRef]
- 12.
Imbert R, Moffa F, Tsepelidis S et al. Safety and usefulness of cryopreservation of ovarian tissue to preserve fertility: a 12-year retrospective analysis. Hum Reprod 2014; 29: 1931 - 40. [PubMed][CrossRef]
- 13.
Oslo universitetssykehus. eHåndboken. https://oslo-universitetssykehus.no/fag-og-forskning/kvalitet/ehandboken (20.6.2017).
- 14.
Donnez J, Dolmans M-M, Demylle D et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet 2004; 364: 1405 - 10. [PubMed][CrossRef]
- 15.
Jensen AK, Macklon KT, Fedder J et al. 86 successful births and 9 ongoing pregnancies worldwide in women transplanted with frozen-thawed ovarian tissue: focus on birth and perinatal outcome in 40 of these children. J Assist Reprod Genet 2017; 34: 325 - 36. [PubMed][CrossRef]
- 16.
Veileder i gynekologi 2015, Norsk Gynekologisk forening, Fertilitetsbevarende tiltak hos kvinner med kreft: http://legeforeningen.no/Fagmed/Norsk-gynekologisk-forening/Veiledere/Veileder-i-gynekologi-2015/Fertilitetsbevarende-tiltak-hoskvinner-med-kreft/ (3.5.2017).
- 17.
Seshadri T, Gook D, Lade S et al. Lack of evidence of disease contamination in ovarian tissue harvested for cryopreservation from patients with Hodgkin lymphoma and analysis of factors predictive of oocyte yield. Br J Cancer 2006; 94: 1007 - 10. [PubMed][CrossRef]
- 18.
Dolmans MM, Marinescu C, Saussoy P et al. Reimplantation of cryopreserved ovarian tissue from patients with acute lymphoblastic leukemia is potentially unsafe. Blood 2010; 116: 2908 - 14. [PubMed][CrossRef]
- 19.
Bjelland EK, Wilkosz P, Tanbo TG et al. Is unilateral oophorectomy associated with age at menopause? A population study (the HUNT2 Survey). Hum Reprod 2014; 29: 835 - 41. [PubMed][CrossRef]
- 20.
Helsedirektoratet 2017 http://www.helsebiblioteket.no/retningslinjer/brystkreft/fertilitetsbevarende-tilbud (22.1.2018).
- 21.
Cobo A, Garcia-Velasco JA, Domingo J et al. Is vitrification of oocytes useful for fertility preservation for age-related fertility decline and in cancer patients? Fertil Steril 2013; 99: 1485 - 95. [PubMed][CrossRef]