MAIN POINTS
None of our patients with exercise-induced rhabdomyolysis developed severe kidney injury or other complications
All patients showed decreasing creatine kinase levels upon outpatient follow-up
More patients with exercise-induced rhabdomyolysis should be considered for outpatient treatment
Our patients with exercise-induced rhabdomyolysis are treated as outpatients or considered for discharge with creatine kinase < 40 000 IU/l in the absence of risk factors or complications
Rhabdomyolysis is characterised by the breakdown of myocytes, with subsequent release of the intracellular components creatine kinase, myoglobin and electrolytes into the circulation (1).
The incidence of exercise-induced rhabdomyolysis has been increasing in Norway in recent years, with an estimated hospitalisation rate of 4.6/100 000 inhabitants (2, 3).
The classic symptom triad of muscle pain, muscle weakness and dark urine is seen in about 10 % of patients (1). Clinical presentation typically includes swollen, sore muscles, reduced joint mobility and loss of strength. The condition can vary from an asymptomatic elevation of creatine kinase levels to severe disease with kidney injury, electrolyte imbalance, compartment syndrome and disseminated intravascular coagulation (DIC) (4).
Kidney injury occurs as a result of renal vasoconstriction as well as obstructive and direct nephrotoxic effects of myoglobin, which are exacerbated by an acidic environment (5). Electrolyte imbalance is common. Hypocalcaemia can occur as a result of complex formation between calcium and phosphate in the muscles. There is no indication for treatment in the absence of serious symptoms (such as seizures or arrhythmia).
The release of phosphate and potassium upon myocyte damage and kidney injury can give rise to hyperphosphatemia and hyperkalaemia. These are treated in accordance with standard guidelines and usually resolve upon adequate fluid therapy and proper diuresis (5). Elevated creatine kinase levels may also be seen after strenuous exercise without necessarily indicating clinical rhabdomyolysis (6).
Exercise-induced rhabdomyolysis is diagnosed on the basis of clinical symptoms plus a creatine kinase level that is more than 5–10 times the upper reference limit, or > 5 000 IU/l (4, 7), and is often a consequence of workouts that are too strenuous, too long and/or repetitive, often with major eccentric muscle loading. Treatment recommendations are based on small retrospective observational studies, animal models, case reports, and expert opinions (7). Exercise-induced rhabdomyolysis is often treated as rhabdomyolysis due to other causes, with intensive care, large volumes of intravenous fluid and urinary alkalinisation. This increases the risk of overhydration and disturbances in the acid-base-electrolyte balance.
The purpose of this study was to describe the treatment, potential complications, and follow-up of patients with exercise-induced rhabdomyolysis in our hospital.
Material and method
This is a retrospective observational study from 2011 up to and including 2015. Patients were identified on the basis of a measured creatine kinase level exceeding 5 000 IU/l in Acute Admissions at Diakonhjemmet Hospital, and their medical records were then reviewed to determine the cause. Patients ≥ 18 years with exercise-induced rhabdomyolysis were included; those with rhabdomyolysis due to other causes were excluded.
Various types of data were obtained from the medical records and medication charts (Box 1). Since we did not have a baseline value for creatinine, we assumed that the last measured value represented the patient's normal level. The peak creatinine level during hospitalisation was used to assess kidney injury in accordance with the KDIGO classification of acute kidney injury: Stage 1 corresponds to an increase in creatinine level ≥ 26.5 μmol/l or to 1.5–1.9 times the baseline value, stage 2 an increase to > 2.0–2.9 times the baseline value, and stage 3 an increase to > 3 times the baseline value or to ≥ 353.6 μmol/l (8).
Box 1 Data obtained from the medical records and medication charts
Gender, age, weight, medical history, regular medications
Time from exercise to first medical contact, type of strength training and affected muscle group
Laboratory values (including creatine kinase, creatinine, eGFR and electrolytes) upon admission, during hospitalisation (including time of peak creatine kinase), at discharge and upon follow-up
Duration of hospitalisation, department to which the patient was admitted or in which they received outpatient treatment
Type and duration of treatment, including intravenous fluid volume, urinary alkalinisation, complications (kidney injury, electrolyte imbalance, compartment syndrome) and number of patients examined by a surgeon due to suspected compartment syndrome
Exercise advice given to patients upon discharge, type of follow-up and any readmissions
Diagnostic codes used for patients with exercise-induced rhabdomyolysis
The Data Protection Officer at Oslo University Hospital, Ullevål, approved the study – on the condition that patients provided written consent for access to their medical records.
Statistics
The data were not normally distributed. We therefore used non-parametric tests and have provided the median plus the lower and upper quartiles as a measure of spread. The relationship between creatine kinase levels upon arrival in patients that were hospitalised and those that were treated as outpatients, and comparison of urine pH and creatine kinase levels in cases of urinary alkalinisation, were analysed using Mann-Whitney's test. A p-value < 0.05 was considered statistically significant.
The analyses were performed in STATA SE (version 14.2, Stata Corporation, College Station, TX, USA).
Results
We identified a total of 128 patients with rhabdomyolysis, of whom 42 had exercise-induced rhabdomyolysis. Of these, 31 patients (74 %) provided written consent for access to their medical records. In 2011, there were no patients diagnosed with exercise-induced rhabdomyolysis, whereas in 2015 there were 16 (52 % of all the rhabdomyolysis cases).
The patients were previously healthy, although six (19 %) reported other conditions with no known association with rhabdomyolysis. Eleven patients (35 %) used medications with no known relation to rhabdomyolysis, two used medications associated with rhabdomyolysis, and the remainder used no regular medications (Table 1).
Table 1
Patient characteristics (2011–15)
| Gender (n = 31) |
Number (%) |
|---|---|
| Men |
15 (48) |
| Women |
16 (52) |
| Age (years) |
Median (lower–upper quartile) |
| Men (n = 15) |
27 (22–32) |
| Women (n = 16) |
25 (23–30) |
| Weight (kg) |
|
| Men (n = 14) |
76 (75–95) |
| Women (n = 11) |
62 (55–74) |
| Time (days) from exercise to first medical contact (n = 30) |
3 (2–4) |
| Reason for medical contact (n = 31) |
Number (%) |
| Muscle pain |
30 (97) |
| Muscle swelling |
23 (74) |
| Dark urine |
7 (23) |
| Triad (pain, swelling, dark urine) |
2 (6) |
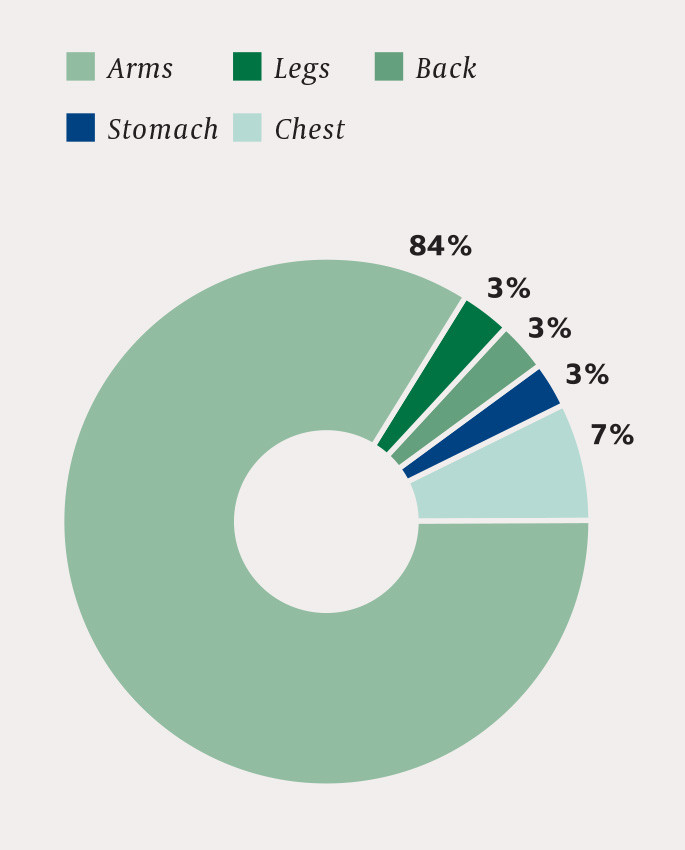
All cases of exercise-induced rhabdomyolysis were the result of strength training – 81 % after weight training, 13 % after Crossfit and 6 % after a workout with a personal trainer. Muscles in the upper extremities were most frequently affected (Fig. 1).
The median creatine kinase level on arrival was 36 797 IU/l (17 172–53 548 IU/l). Four patients had creatine kinase levels > 100 000 IU/l. Creatine kinase levels peaked at a median of three days post-exercise. Median creatine kinase levels were significantly higher in those who were hospitalised (44 768 IU/l) than in those who were treated as outpatients (17 172 IU/l) (p < 0.001).
No further marked abnormalities in test results were seen on arrival, but urine strips tested positive for blood in 12 patients (55 %). Myoglobin also causes urine strips to test positive for blood, as the strips do not distinguish between myoglobin and the haemoglobin in blood. Microscopy would provide clarification as erythrocytes will not be seen in cases of myoglobinuria. Unfortunately, the urine of our patients was not examined by microscopy.
Twenty patients (65 %) were hospitalised and 11 were treated as outpatients (Table 2). All hospitalised patients received intravenous fluid therapy with 9 mg/ml sodium chloride (NaCl) or Ringer acetate. The aim was to achieve an hourly diuresis of 200–300 ml (9), which was accomplished in all patients. Three patients received loop diuretics owing to signs of overhydration (oedema). Those who received outpatient treatment were advised to drink plenty of fluids, usually at least three litres daily.
Table 2
Therapeutic interventions during the period 2011–2015
| Hospitalisation |
Median (lower–upper quartile) |
|---|---|
| Duration (days) of hospitalisation (n = 20) |
2.5 (1–4) |
| Place of treatment |
Number (%) |
| Outpatient clinic (n = 31) |
11 (35.5) |
| Intensive care (n = 31) |
10 (32.3) |
| Observational ward (n = 31) |
9 (29.0) |
| Standard ward (n = 31) |
1 (3.2) |
| Fluid therapy |
Median (lower–upper quartile) |
| Total volume (ml) (n = 24) |
6 000 (1 000–27 700) |
| Volume per kg bodyweight (ml) (n = 22) |
90 (41–170) |
| Duration (days) (n = 21) |
2 (1–4) |
| Urinary alkalinisation |
Number (%) |
| Patients (n = 31) |
11 (35) |
| Median (lower–upper quartile) |
|
| Duration (days) (n = 11) |
2 (1–3) |
| Volume (mmol) (n = 11) |
250 (175–584) |
There was no significant difference in median urine pH between those who underwent urinary alkalinisation and those who did not (p = 0.24). Median creatine kinase levels upon hospitalisation were significantly higher in those who underwent urinary alkalinisation (59 271 IU/l) than those who did not (19 198 IU/l) (p = 0.001). In our dataset, creatine kinase levels appeared to be the deciding factor for whether or not alkalinisation was initiated.
Some patients had mild electrolyte imbalances, all of which were normalised with fluid therapy alone. The median creatinine level was 73 μmol/l (64–107 μmol/l) at the time of hospitalisation. Two patients had possible mild kidney injury (stage 1), with peak creatinine levels of 103 μmol/l and 107 μmol/l. Both were young and healthy and had peak creatine kinase levels of 45 538 IU/l and 28 988 IU/l, respectively. Their kidney function had normalised prior to discharge. Three patients received > 20 litres of intravenous fluid, developed overhydration with peripheral oedema and received loop diuretics. Four were assessed by a surgeon with respect to possible compartment syndrome, but none required surgical intervention.
Twenty-two patients (71 %) attended a follow-up appointment, either at an outpatient clinic or with their general practitioner. All of the patients assessed in outpatient clinics were found to have decreasing creatine kinase levels (Table 3). Two patients experienced a recurrence of exercise-induced rhabdomyolysis during the study period, one of whom was referred for further assessment.
Table 3
Post-discharge follow-up of patients in the period 2011–15
| Laboratory tests |
Median (lower–upper quartile) |
|---|---|
| S-creatine kinase at discharge (IU/l) (n = 21) |
16 051 (11 845–26 505) |
| S-creatine kinase upon follow-up (IU/l) (n = 12) |
9 633 (4 161–13 764) |
| Number of days until follow-up appointment (n = 22) |
2.5 (2–5) |
| Number (%) |
|
| Follow-up appointment in hospital (n = 30) |
13 (43) |
| Advised to attend follow-up appointment with general practitioner (n = 30) |
9 (30) |
| No follow-up appointment arranged (n = 30) |
8 (27) |
| Advice about exercise given at discharge (n = 31) |
20 (65) |
Discussion
We describe the treatment, complications and follow-up of 31 patients with exercise-induced rhabdomyolysis with creatine kinase levels ranging from 9 065 IU/l to > 100 000 IU/l.
Incidence
No attempt was made to estimate disease incidence because our patient population varied throughout the study period. There is no specific diagnostic code for exercise-induced rhabdomyolysis in ICD-10. Four of our 31 patients (13 %) would have been overlooked if we had sought to identify them only on the basis of the most commonly used codes, T79.6 (traumatic ischaemia of muscle) and M62.8 (other specified muscle disease). The estimated hospitalisation rate of 4.6/100.000 inhabitants (2) does not take this into account. Thus it is probably an underestimate.
The number of patients with exercise-induced rhabdomyolysis who self-manage their condition or who contact their general practitioner or a sports physician is also unknown. However, the current study, as well as two further Norwegian studies (2, 3), suggest that the incidence is increasing and that a substantial number of affected patients are admitted to departments of general medicine.
Acute kidney injury
Patients with exercise-induced rhabdomyolysis appear to have a very low risk of kidney injury provided there are no other risk factors present, such as dehydration, hyperthermia, concomitant infection, medications (e.g. NSAIDs and statins), performance-enhancing drugs (e.g. creatine and anabolic steroids), myopathy or sickle cell anaemia (10). Two of our patients had one of these risk factors.
One Norwegian study reported two patients with severe kidney injury following exercise-induced rhabdomyolysis, but no further characterisation of the patients was presented (3). Another Norwegian study and several international studies reported no patients with severe kidney injury (2, 11, 12). The relationship between creatine kinase and kidney injury remains uncertain (13).
Fatalities and kidney injury requiring dialysis have both been reported in cases of exercise-induced rhabdomyolysis, but all of the patients concerned had at least one risk factor (14, 15). In our study, creatine kinase levels did not predict the risk of kidney injury. Whether creatine supplements increase the risk of exercise-induced rhabdomyolysis and kidney injury, and whether creatine can affect the serum creatinine level, is subject to debate. Creatine supplements can make it difficult to assess potential kidney injury (16).
Treatment and follow-up
Diuresis should guide fluid infusion, and should be 200–300 ml/h to avoid the risk of overhydration and disturbances in the acid-base electrolyte balance (1). Large volumes of saline (9 mg/ml NaCl) may lead to hyperchloremic metabolic acidosis, whereas large volumes of Ringer acetate may lead to hypochloremic metabolic alkalosis, due to conversion of acetate into bicarbonate. Alternating between these two infusion fluids should therefore be considered.
Urinary alkalinisation with sodium bicarbonate is often suggested as a means of reducing myoglobin-induced kidney injury. However, the efficacy of this intervention has not been evaluated in randomised trials and routine use is not recommended (17). When performed, urinary alkalinisation should begin once diuresis is underway and should be discontinued if the urine pH does not increase > 6.5 after six hours of infusion or if the arterial pH exceeds 7.5 (5, 18).
The creatine kinase level seemed to be the factor that determined whether patients received inpatient or outpatient treatment. There was no specific creatine kinase threshold at which patients were discharged, and the criteria for discharge varied between doctors. The criteria used appeared to be improvement in symptoms, decreasing creatine kinase levels and the absence of new complications.
Patients with exercise-induced rhabdomyolysis often receive the same treatment as patients with rhabdomyolysis due to other causes. We believe this to be incorrect and have changed our local treatment practice.
Patients hospitalised with exercise-induced rhabdomyolysis may receive treatment on either standard or observational wards, and diuresis should preferably be measured noninvasively. Our patients are considered for discharge when their creatine kinase levels have fallen below 40 000 IU/l at least three days post-workout, provided that their symptoms are improving and there are no new complications (9). If such patients present to Acute Admissions, they may be considered for outpatient treatment with follow-up as described below.
The creatine kinase threshold is intended to provide clinical guidance for doctors. It was selected on the basis of experience which suggested that patients who were fit for discharge had creatine kinase levels below 40 000 IU/l. A similar threshold has been discussed in another study (13).
The creatine kinase level should be checked in an outpatient clinic, or by a general practitioner, after a further 2–3 days. This is because the time from exercise to peak creatine kinase can vary from one to five days (19, 20). It is especially important to identify patients who show recurrent increases in creatine kinase after exercise, as these individuals may have underlying myopathy. It is unclear how many patients this may apply to.
Strengths and weaknesses
We identified patients on the basis of creatine kinase levels as opposed to diagnosis codes, and it is therefore unlikely that we have missed any cases. We carefully examined fluid therapy and urinary alkalinisation as well as laboratory test results, including electrolytes: data that many previous studies failed to provide a full account of.
Weaknesses of the study include small sample size, retrospective design, lack of blood samples upon discharge, imprecise creatine kinase levels if they exceeded 100 000 IU/l (laboratory upper limit), lack of urine microscopy and limited follow-up. Moreover, we lacked complete information about the use of supplements – of which creatine is particularly important.
The educational value of this project lies in areas including preventing exercise-induced rhabdomyolysis and in considering what constitutes appropriate training for patients that have been affected. We therefore offer some straightforward, practical, experience-based advices.
Prevention
Most cases of exercise-induced rhabdomyolysis are the result of strength training. Those who are out of shape or who are trying strength training for the first time should not exercise to the point of exhaustion – about half the number of repetitions one thinks one could have managed is enough. The same applies to those who take a break from training, omit an exercise for a few weeks, or switch to a new exercise. The muscles' resistance improves after the first session with a new exercise, and this effect lasts from days to weeks (21). Nevertheless, it is inadvisable to perform sets to the point of exhaustion the second time the exercise is attempted.
The acquired muscle protection is relatively specific to the range of motion and load/velocity profile of an exercise. Even if two exercises look relatively similar, it is best to hold back a little when switching to a new exercise, and to be particularly cautious with eccentric muscle training. In the third or fourth session, it is acceptable to perform sets to the point of exhaustion. If an exercise is performed regularly at least once a week, the body can tolerate substantial stresses without sustaining muscle damage. At this point one can perform sets to the point of exhaustion. Note, however, that training appears to be at least as effective when the body is not pushed to complete exhaustion in every set.
Training after exercise-induced rhabdomyolysis
Necrosis occurs in segments of the muscle fibres in cases of exercise-induced rhabdomyolysis. Depending on severity, anything from a few to more than half of the muscle fibres in the overloaded muscle group will need to regenerate before normal muscle function can be restored (22). Regeneration of fibres in the necrotic regions usually takes a few weeks. This process is almost complete: all fibres can regenerate, provided that the circulation to the necrotic regions is intact (22).
Although most of the muscle fibres will have been rebuilt after 1–2 months, they will be smaller than the original fibres. Strength will therefore be reduced for some time (several months) after severe rhabdomyolysis. It is important to begin normal training in order to quickly normalise muscle function. Allow the muscle group to rest until all symptoms have resolved and clinical tests show normal results (creatine kinase < 1 000 IU/l), usually about two weeks after the injury. Strength training can then be resumed with care, and the load on the affected muscle group can slowly be increased. Unaffected muscle groups can be trained as normal all the time.
- 8.
Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 2012; 120: c179 - 84 [PubMed].. [PubMed]
- 9.
Tazmini K. Metodebok for leger - medisinsk avdeling. 4. utg. Oslo: Diakonhjemmet Sykehus, 2016.
- 22.
Paulsen G, Mikkelsen UR, Raastad T et al. Leucocytes, cytokines and satellite cells: what role do they play in muscle damage and regeneration following eccentric exercise? Exerc Immunol Rev 2012; 18: 42 - 97 [PubMed].. [PubMed]
Studien gir ny nødvendig kunnskap, men reiser også noen spørsmål:
1) Hvordan definerte/målte man muskelødem som inn symptom?
2) Brukte man noe måleinstrumenter ved vurderingen av kompartmentsyndrom?
3) Treningsanbefalingene i avsnittet om forebygging samsvarer vel ikke med studien (?).
Takk uansett for fin lesing.
Kjære Pavels,
Vi takker for hyggelig interesse for vårt arbeid. Vi har også fått tilbakemeldinger blant annet på sosiale medier som bekrefter at dette er et tema som angår flere.
Vi takker Tazmini og kollegaer på Diakonhjemmet for en interessant artikkel om treningsindusert rabdomyolyse. Vi er enige med forfatterne i fortolkningen om at friske personer med treningsindusert rabdomyolyse generelt har lav risiko for komplikasjoner. Dette samsvarer med erfaringen fra vår egen avdeling og det støttes av litteraturen – dog uten at det finnes noen større studier på dette.
Artikkelen beskriver 31 pasienter med CK > 5000 E/l i perioden 2011- 2015, der over 50% ble inkludert i 2015. Dette understreker den økende trenden med treningsindusert rabdomyolyse som vi har sett de siste årene – også hos oss.
Vi er enige med forfatterne i at dette er en gruppe pasienter som ikke trenger like intensiv behandling som pasienter med rabdomyolyse av andre årsaker. Men vi mener forfatterne trekker konklusjonen om at det ”ikke er riktig å legge inn pasienter med CK verdier < 40 000 E/l” litt langt når populasjonen kun består av 31 pasienter. Studien det refereres til som har diskutert tilsvarende grenser, ble utført med endepunktene alvorlig nyresvikt med behov for dialyse og/eller død (1). Dermed mener vi den vanskelig kan brukes til å forsvare konklusjonen.
Vi savner også måling av serum myoglobin. Myoglobin er mest sannsynlig hovedaktøren bak rabdomyolyseindusert nyresvikt og er sannsynligvis også en bedre prognostisk markør (2-4). En retrospektiv studie fra 2015 fant at myoglobin kan være en god og tidlig prediktor for nyresvikt (4), det samme har noen mindre studier. Vi har nylig publisert en retrospektiv studie som inkluderte 341 pasienter med rabdomyolyse på medisinsk avdeling og fant der at serum myoglobin var en bedre prediktor for utvikling av akutt nyresvikt enn CK. Vi fant også at risikoen for å utvikle nyresvikt økte med en økende myoglobin/CK ratio- der en ratio på > 0.2 var assosiert med en økt sannsynlighet for utvikling av nyresvikt (5).
Vi foreslår derfor at myoglobin også bør måles hos alle pasienter med rabdomyolyse og at dette sammenholdes med CK før man kan si noe sikkert om hvem som bør legges inn og hvem som kan behandles poliklinisk. Men først og fremst så trenger vi større studier for å kunne si noe sikkert om treningsindusert rabdomyolyse.
Litteratur
1. McMahon GM, Zeng X, Waikar SS. A risk prediction score for kidney failure or mortality in rhabdomyolysis. JAMA internal medicine. 2013;173(19):1821-8.
2. Bosch X, Poch E, Grau JM. Rhabdomyolysis and acute kidney injury. The New England journal of medicine. 2009;361(1):62-72.
3. Chavez LO, Leon M, Einav S, Varon J. Beyond muscle destruction: a systematic review of rhabdomyolysis for clinical practice. Critical care (London, England). 2016;20(1):135.
4. Premru V, Kovac J, Ponikvar R. Use of myoglobin as a marker and predictor in myoglobinuric acute kidney injury. Therapeutic apheresis and dialysis : official peer-reviewed journal of the International Society for Apheresis, the Japanese Society for Apheresis, the Japanese Society for Dialysis Therapy. 2013;17(4):391-5.
5. Vangstad M, Bjornaas MA, Jacobsen D. Rhabdomyolysis: a 10-year retrospective study of patients treated in a medical department. Eur J Emerg Med. 2017.
Takk for gode og lærerike kommentarer til artikkelen vår. Vi er enige om at komplikasjonsraten er liten og et lavere behandlingsnivå er tilstrekkelig for denne pasientgruppen som synes økende i antall.
Vi har ikke skrevet i vår artikkel at det ikke er riktig å legge inn pasienter med kreatinkinase (CK) < 40 000E/l. Vi har skrevet at det ikke er riktig å behandle pasienter med treningsindusert rabdomyolyse på samme måte som pasienter med rabdomyolyse av andre årsaker.
Den nevnte veiledende CK-verdien på 40 000 E/l var omdiskutert også hos oss. Dette er kun ment veiledende for leger ved vårt sykehus og ikke nødvendigvis ment som en retningslinje for andre. Vi er enige i at vår studie har en liten populasjon, hvorfor vi ikke har kommet med en generell anbefaling. Vi ser også at vi kunne ha formulert oss noe mer moderat gitt relativt lite pasientgrunnlag. Vår hovedkonklusjon var imidlertid at det ikke er riktig å behandle pasienter med treningsindusert rabdomyolyse på samme måte som pasienter med rabdomyolyse av andre årsaker.
Det kan være at myoglobin eller myoglobin/CK-ratio er en bedre prediktor for utvikling av akutt nyresvikt enn kreatinkinase hos pasienter med rabdomyolyse. Vårt sykehus analyserer ikke myoglobin per i dag, men har den som sendeprøve. Dette gjør prøven ikke godt egnet som initialt beslutningsverktøy hos oss. Myoglobin ble i vår retrospektive studie ikke tatt, men vi er enige i at det ville vært ønskelig å ha hatt myoglobin svaret. Men myoglobin og kreatinkinase har forskjellige halveringstid og følgelig forskjellige tidskurver for maksimale verdier (1). Dette kan komplisere bruken av ratioen som dere anbefaler. Skal man bruke ratio mellom myoglobin og kreatinkinase så vil den være påvirket av tidspunktet man tar blodprøven på. I en studie kom maks myoglobin etter 72 timer mens kreatinkinase fortsatt var på vei opp etter 96 timer (2). Som et varsko i travel klinikk, tar man for eksempel blodprøven 24 timer etter den utløsende treningsøkten, kan man få meget lave verdier av både myoglobin og kreatinkinase selv om det foreligger store muskelødeleggelser (2). Ser man bare på maksverdier uavhengig av tidspunkt, vil det være en god sammenheng mellom kreatinkinase og myoglobin fordi de begge reflekter godt hvor mye muskelvev som får nekrose (egen observasjon, T. Raastad).
Studien til Vangstad og kolleger er viktig. Blant deres pasienter med rabdomyolyse var 30 (9 %) treningsinduserte, hvor av syv fikk akutt nyresvikt (3). Det fremgår ikke av artikkelen hvor høye CK-verdier disse pasientene hadde eller om de hadde andre risikofaktorer for utvikling av akutt nyresvikt. Det er diskutabelt om det er riktig å generalisere deres konklusjon, at myoglobin eller myoglobin/CK-ratioen er en bedre prediktor for utvikling av nyresvikt hos alvorlig syke pasienter, til friske personer som får treningsindusert rabdomyolyse.
Fordi det er relativ få gode studier på treningsindusert rabdomyolyse, er vi helt enige i at man trenger større studier, gjerne multisenter, for å øke kunnskapen om dette. Forståelse av muskelmetabolisme kan være mangelfull i indremedisinen.
Litteratur
1. Huerta-Alardin AL, Varon J, Marik PE. Bench-to-bedside review: Rhabdomyolysis -- an overview for clinicians. Crit Care 2005; 9: 158-69.
2. Lavender AP, Nosaka K. Changes in markers of muscle damage of middle-aged and young men following eccentric exercise of the elbow flexors. J Sci Med Sport 2008; 11: 124-31.
3. Vangstad M, Bjornaas MA, Jacobsen D. Rhabdomyolysis: a 10-year retrospective study of patients treated in a medical department. Eur J Emerg Med 2017.