Stroke is a frequent cause of death and disability. Most strokes are ischaemic. In recent years there has been a significant improvement in the treatment of acute cerebral infarction, including thrombolytic treatment and thrombectomy.
Approximately 13 % of all strokes in Norway are caused by a spontaneous intracerebral haemorrhage (1). These patients are at an elevated risk of death or serious disability when compared to patients with a cerebral infarction. Mortality is at its highest during the first days after the onset of the disease. In contrast to cerebral infarction, no similar improvement has occurred in the emergency treatment of spontaneous intracerebral haemorrhages.
Whether or not the incidence of intracerebral haemorrhages has changed in recent decades is uncertain (2). There are relatively few data available on the incidence and progression in Norway. A study from the Innherred district in 1994 – 96 reported an incidence of 32/100 000/year (3), and a relatively recent study from the southernmost region of Norway showed an incidence of 17.6/100 000/year (4).
With this study, we wished to estimate the annual incidence of first-time spontaneous intracerebral haemorrhages in women and men in different age groups in Vestfold county and to describe the progression of and prognosis for this condition.
Material and method
In Vestfold county, all patients showing symptoms of cerebral stroke are admitted to Vestfold Hospital in Tønsberg. All patients admitted with a newly occurred spontaneous intracerebral haemorrhage were registered prospectively during the period 1 September 2010 – 31 May 2014, including whether this was a first-time or recurrent haemorrhage. Patients with traumatic cerebral haemorrhage, haemorrhage in a tumour, haemorrhage following thrombolytic therapy, subdural haematoma and subarachnoid haemorrhage were excluded.
Symptom onset was defined as the time of newly occurred neurological symptoms. The time from symptom onset to admission, the use of anticoagulants and platelet inhibitors, previous disease such as hypertension, diabetes mellitus, atrial fibrillation and previous cerebral stroke as well as emergency treatment were registered. The variables were registered from the patient record and entered into a separate registry.
All patients underwent a CT scan of the head upon admission. The location of the haemorrhage and any breakthrough of the haemorrhage to the ventricular system were identified, and the volume of haemorrhage was measured in the CT images as A × B × C/2 cm3, in which A is the largest diameter of the haemorrhage, B is the diameter perpendicular to A, and C is the number of sections with visible haemorrhage multiplied by the thickness of the section (5).
As quickly as possible after the elapse of the three-month period after symptom onset, the patients underwent an outpatient check or were contacted by telephone. If the patient could not be contacted directly, information was provided by next of kin or nursing-home personnel. The time of registration was set at 90 days after the haemorrhage.
Registrations were made on the modified Rankin scale (mRS) (6). This is a measure of neurological outcomes after the haemorrhage, where 0 denotes «no symptoms at all» and 5 means «requiring constant nursing care and attention». Barthel ADL index scores (activities of daily living) were also registered (7). This provides a measure of the extent to which the patient is self-reliant in daily activities. The scale ranges from 1 to 20, where 1 is «fully reliant on care» and 20 is «fully independent». The use of nursing resources in the form of hospitalisation days, rehabilitation institutions, nursing homes and involvement of home nursing services during the first 90 days was registered.
The results were processed with the aid of IBM SPSS Statistics 21 (IBM Corp., USA). The chi-square test was used to compare categorical variables, the Mann-Whitney U‑test was used for comparison of non-parametric variables and the independent t-test for comparison of continuous variables. The statistical significance level was set at 5 %.
Population data were retrieved from Statistics Norway (8). To estimate incidence we used the average population in Vestfold county in the study period, altogether 236 200 people. We created ten-year age groups, except for the youngest patients (< 50 years), who were categorised in a single group, because haemorrhages occur very rarely in these (2). We then estimated the incidence in each age group for men, women and all patients, based on the same population data. We subsequently adjusted the incidence to the European standard population from both 1976 and 2010 in accordance with population data from Eurostat (9) using the direct method of standardisation (10). For all rates, we estimated a 95 % confidence interval (CI) from the chi-square approach to the Poisson distribution.
The project was submitted to the Regional Committee on Medical and Health Research Ethics for South-Eastern Norway, which has deemed it to constitute a project for quality assurance of an established treatment programme (REK reference: 2010/1035a). The project has been approved by the Norwegian Centre for Research Data (NSD, formerly the Norwegian Social Science Data Services) (ref. 24461). Only data that are registered in normal clinical practice were collected. Information was provided to patients orally upon admission, whenever possible. During the 90-day check, oral information was again provided about the study.
Results
A total of 201 patients were admitted with an acute intracerebral haemorrhage during the registration period. This is equivalent to an incidence of this condition of 22.7/100 000/year in Vestfold county. Nineteen admissions were caused by a recurring haemorrhage in patients who previously had sustained one or more. Altogether 182 patients (102 men) had a first-time intracerebral haemorrhage. This is equivalent to a total incidence of first-time haemorrhages of 20.5/100 000/year. The following results refer only to patients with a first-time haemorrhage.
The age distribution and incidence for men and women in the different age groups are shown in Table 1. The average age was 72.8 years (range: 20 – 101 years). There was a marked increase in the frequency of intracerebral haemorrhage after the age of 60. In all age groups, the incidence was higher among men. The age-adjusted incidence, adjusted for the European standard population in 1976 amounted to 13.7/100 000/year, while the incidence adjusted for the European standard population in 2010 (the EU-27 + EFTA) amounted to 22.8/100 000/year.
Table 1
Annual age-specific incidence for first-time spontaneous intracerebral haemorrhage in Vestfold county in the period 1 September 2010 – 31 May 2014, by gender and age. The bottom row shows also the incidence after adjustment to the European standard population in a: 1976 and b: 2010 (9)
| Total |
Men |
Women |
|||||||||
| Age (years) |
Number |
Incidence |
(95 % CI) |
Number |
Incidence |
(95 % CI) |
Number |
Incidence |
(95 % CI) |
||
| 0 – 49 |
13 |
2.3 |
(0.2 – 7.2) |
10 |
3.5 |
(0.8 – 9.5) |
3 |
1.1 |
(0.0 – 5.6) |
||
| 50 – 59 |
14 |
11.7 |
(5.8 – 20.3) |
8 |
13.5 |
(7.3 – 22.9) |
6 |
10.0 |
(4.8 – 18.4) |
||
| 60 – 69 |
35 |
34.2 |
(23.5 – 47.5) |
22 |
43.6 |
(31.5 – 58.5) |
13 |
25.1 |
(16.2 – 36.9) |
||
| 70 – 79 |
52 |
86.9 |
(69.2 – 106.8) |
33 |
121.8 |
(100.9 – 145.1) |
19 |
58.0 |
(44.0 – 75.0) |
||
| 80 – 89 |
57 |
166.2 |
(141.7 – 193.3) |
26 |
193.8 |
(167.2 – 222.8) |
31 |
148.4 |
(125.1 – 173.9) |
||
| > 90 |
11 |
145.6 |
(122.8 – 171.2) |
3 |
154.4 |
(130.6 – 180.3) |
8 |
142.5 |
(120.1 – 167.9) |
||
| All ages |
182 |
20.5 a: 13.7 b: 22.8 |
(12.6 – 31.5) |
102 |
23.2 |
(14.6 – 34.5) |
80 |
17.9 |
(10.3 – 27.8) |
||
Table 2 shows the distribution of risk factors among the patients. Altogether 71 patients (39.0 %) used no form of anticoagulant upon admission, whereas 43 patients (23.6 %) used warfarin, alone or in combination with a platelet inhibitor or low-dosage heparin. Atrial fibrillation was the most common indication for the use of warfarin.
Table 2
Distribution of risk factors for patients admitted with first-time spontaneous intracerebral haemorrhage at Vestfold Hospital in the period 1 September 2010 – 31 May 2014. Number (%) unless otherwise specified.
|
|
Total (N = 182) |
Men (n = 102) |
Women (n = 80) |
| Age (years), average ± SD |
72.8 ± 13.6 |
70.3 ± 14.1 |
75.9 ± 12.4 |
| Hypertension |
96 (52.7) |
55 (53.9) |
41 (51.3) |
| Atrial fibrillation |
49 (26.9) |
29 (28.4) |
20 (25.0) |
| Previous brain infarction |
33 (18.1) |
17 (16.7) |
16 (20.0) |
| Diabetes mellitus |
31 (17.0) |
21 (20.6) |
10 (12.5) |
| Use of plate inhibitors |
70 (38.5) |
42 (41.2) |
28 (35.0) |
| Use of warfarin |
43 (23.6) |
28 (27.5) |
15 (18.3) |
The median haemorrhaged volume after administration of warfarin amounted to 20 ml, and with no warfarin use to 15 ml (p = 0.123). The average international normalised ratio (INR) for warfarin users upon admission was 3.1 (range: 1.0 – 7.5). Altogether 67 % of the warfarin users had an INR value within the therapeutic area (2 < INR < 3). We found no significant difference in haemorrhaged volumes for patients with an INR value ≥ 3 when compared to those with an INR value < 3 (p = 0.567). Novel oral anticoagulants (NOACs) were introduced during the last part of the inclusion period, and only four patients used such drugs. Altogether 63 patients (34.6 %) used a platelet inhibitor exclusively.
Two days after symptom onset, mortality in this material amounted to 23 %, seven-day mortality was 30.1 %, thirty-day mortality was 39.6 % and 90-day mortality amounted to 40.7 %. Altogether 71 patients died from the cerebral haemorrhage itself, while three died from other causes. Those who died were significantly older (75.6 years on average) than those who survived the haemorrhage (70.1 years on average) (p = 0.021).
Warfarin use was associated with a significantly elevated 90-day mortality (60.5 %) when compared to no use of warfarin (34.5 %) (p = 0.002). Treatment with platelet inhibitors was also associated with an elevated 90-day mortality (41.3 %) when compared to no use of any form of anticoagulant treatment (28.2 %), but this difference was not statistically significant (p = 0.111). There was no significant difference in 90-day mortality in terms of whether the haemorrhage had occurred in the right or left brain hemisphere.
A total of 161 patients (89.9 %) were admitted during the first 24 hours after symptom onset and 171 patients (95.5 %) within the first 48 hours. The average hospitalisation time for these patients amounted to 9.4 days (range: 0 – 67 days), with a median hospitalisation time of 7.0 days.
Most of the patients received no specific treatment targeting the haemorrhage. Altogether 29 patients who were using warfarin received emergency treatment to normalise their INR value, in the form of the vitamin K compound phytomenadione (Konakion), prothrombin complex concentrate (Octaplex/Prothromplex), alone or in combination with phytomenadione, or coagulation active plasma (Octaplas) in combination with phytomenadione, plasma or thrombocytes (Table 3).
Table 3
Treatment in association with acute admission for spontaneous intracerebral haemorrhage at Vestfold Hospital in the period 1 September 2010 – 31 May 2014 (n = 182)
| Treatment |
Number |
| Phytomenadione |
11 |
| Phytomenadione + Octaplex/Prothromplex |
13 |
| Octaplas + phytomenadione/plasma/thrombocytes |
5 |
| Surgical treatment |
16 |
| Symptomatic treatment only |
137 |
Sixteen patients underwent surgery in the context of the emergency admission, most often in the form of external drainage or evacuation of the haematoma.
Ten patients (5.5 %) had an arteriovenous malformation as the likely cause of their haemorrhage. Four of these underwent emergency surgery for evacuation of the haematoma and the malformation. Three patients with arteriovenous malformation died at the acute stage without undergoing surgery.
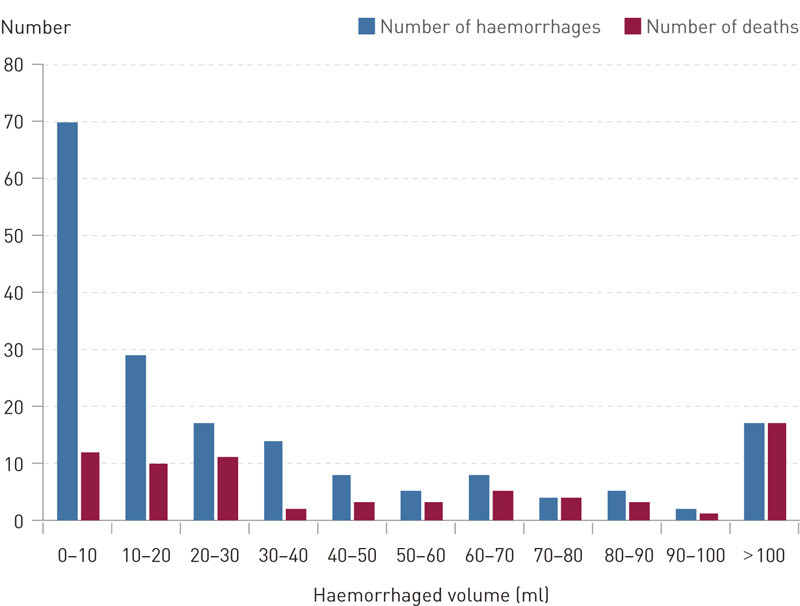
Figure 1 shows the distribution of the haemorrhaged volumes measured upon admission. The smallest haemorrhages occurred most frequently. Mortality increased in pace with increasing haemorrhaged volume, and at a haemorrhaged volume exceeding 100 ml, all patients died. A total of 72 patients (39.6 %) suffered from an intracerebral haemorrhage with a breakthrough to the ventricular system. The presence of a breakthrough haemorrhage entailed a significantly increased mortality, and after 90 days, altogether 44 (61.1 %) of these patients had died (p = 0.001). Three patients suffered only from intraventricular haemorrhage, all of whom died.
Of the 108 patients who survived their cerebral haemorrhage, before its onset altogether 103 lived in their own home. Three months later, 73 patients (67.6 % of the survivors) were back in their own homes. Altogether 23 of these were under supervision by home nursing services, amounting to a total of 450 visits after the patients had been discharged from the hospital. For those who survived the hospitalisation period, a total of 140 days had been spent in a municipal or other rehabilitation institution. Table 4 shows the patients’ place of residence before, and three months after their haemorrhage.
Table 4
Place of residence before and three months after a first-time spontaneous intracerebral haemorrhage for survivors admitted to Vestfold Hospital in the period 1 September 2010 – 31 May 2014 (n = 108)
| Before the haemorrhage |
Three months after the haemorrhage |
|||||
| Number |
(%) |
Number |
(%) |
|||
| Own home without home nursing |
86 |
(79.6) |
50 |
(46.3) |
||
| Own home with home nursing |
17 |
(15.7) |
23 |
(21.3) |
||
| Rehabilitation institution |
0 |
9 |
(8.3) |
|||
| Sheltered housing |
1 |
(0.9) |
1 |
(0.9) |
||
| Nursing home |
4 |
(3.7) |
24 |
(22.2) |
||
| No information |
0 |
(0) |
1 |
(0.9) |
||
| Total |
108 |
108 |
||||
The average score on Barthel ADL index for the survivors was 16.1 (range: 1 – 20). Altogether 50.0 % of the survivors functioned very well, with a Barthel ADL index value of 19 or 20 three months after their haemorrhage. Those who lived at home (n = 73) scored on average 18.8 on the Barthel ADL index (range: 12 – 20), while the nursing-home patients (n = 24) scored 9.5 on average (range: 1 – 17).
The average mRS value for all of the surviving patients was 2.15 (0 – 5). Those who lived at home had an average value of 1.36, while the nursing-home patients had an average value of 4.17. Among those who lived at home after three months there were none who had an mRS value of 5, and three had an mRS value of 4.
Those who lived at home had sustained a significantly smaller haemorrhaged volume (14 ml) on average than those who stayed in an institution (20 ml) (p = 0.040).
Discussion
Our material from Vestfold country included 182 patients with a first-time spontaneous intracerebral haemorrhage. This is the largest prospective study of brain haemorrhages undertaken in Norway. We found a total incidence for first-time intracerebral haemorrhages of 20.5/100 000/year. This is likely to be a minimum incidence, since some patients from Vestfold county may have sustained a brain haemorrhage while outside the county without being transferred to Vestfold Hospital. Some patients with small haemorrhages may also have not been admitted to hospital.
Few Norwegian studies have been made of the incidence of intracranial haemorrhages. A study from the Innherred district from the years 1994 – 96 reported a total incidence of intracerebral haemorrhage of 32/100 000/year (3). This study also included patients with recurring haemorrhages. A large Norwegian study of intracerebral haemorrhages undertaken in the country’s southernmost region in 134 patients found a total incidence of 17.6/100 000/year for first-time haemorrhages (4). Haemorrhages from vascular malformations were not excluded in this study, but three patients with an isolated intraventricular haemorrhage were excluded. Age-adjusted to the standard European population from 1976 we found a total incidence of 13.7/100 000/year, which is consistent with the study from Norway’s southernmost region (12.5/100 000/year). In general, the European population has aged since 1976, and we found an age-adjusted incidence for the standard European population in 2010 of 22.8/100 000/year (9).
An international meta-analysis found an incidence of spontaneous intracerebral haemorrhages of 24.6/100 000/year (2). There were major differences between countries. Many of the studies included also encompassed patients with undefined cerebral stroke, as well as patients who had not undergone any radiological examination. A large, prospective study from southern Sweden found an incidence of primary intracerebral haemorrhage of 30.0/100 000/year (11).
Spontaneous intracerebral haemorrhage affects primarily the oldest part of the population, and the incidence increases with age. In the age group older than 90 years we found a somewhat lower incidence than in the age group 80 – 89 years. There are few patients in this group and the confidence intervals are overlapping, so this finding might not be genuine. However, the study from southern Sweden also showed a non-significant, reduced incidence of intracerebral haemorrhages in men in the oldest age group (≥ 85 years) (11).
The prognosis is poorest for the oldest patients. Those who died from their haemorrhage were significantly older than those who survived. A large haemorrhage volume upon admission was associated with a poor prognosis. It is known that a haematoma may expand somewhat during the first 24 hours after the onset of bleeding (12). We have only measured the volume of the haematoma at the admission stage.
Spontaneous or primary intracerebral haemorrhage is not an unambiguous designation. Most incidence studies have excluded patients with traumatic brain haemorrhage, haemorrhage in a tumour, bleeding after thrombolytic treatment, subdural haematoma and haemorrhage from aneurysms, as we also have done. Some studies have also excluded patients with arteriovenous malformations. We have included patients with acute haemorrhages from such malformations to facilitate comparisons with the other Norwegian incidence studies.
While the frequency of ischaemic stroke has declined during the last decades, it remains uncertain whether this also applies to cerebral haemorrhages (2, 13, 14). This may be caused by the increasing use of anticoagulant treatment during the same period (13). International data differ substantially, however. A Finnish study shows a reduction in warfarin-related haemorrhages and reduced mortality, despite the increasing use of warfarin over time (15).
Most often, the symptoms of a brain haemorrhage occur acutely and alarmingly. Most patients are therefore brought quickly to hospital. In our material, altogether 89.9 % of the patients were admitted within the first 24 hours after symptom onset. The average number of hospitalisation days for patients with a spontaneous intracerebral haemorrhage amounted to 9.4 days. This is a longer hospitalisation period than for other types of cerebral stroke. In 2014, the average number of hospitalisation days for patients with all types of brain strokes at Vestfold Hospital amounted to 5.7 days (1).
The use of anticoagulants gives rise to an increased risk of intracerebral haemorrhage. In our study, the use of warfarin was associated with a significantly increased mortality. Other studies have found the same (4). The novel oral anticoagulants were introduced towards the end of the inclusion period, and we have therefore no relevant data for these drugs. Studies have shown that they are likely to involve a lower risk of intracerebral haemorrhage than warfarin (16).
The use of platelet inhibitors also entails a higher risk of intracerebral haemorrhage (17). We found that those who used platelet inhibitors had a non-significant, increased mortality when compared to patients who did not undergo any anticoagulant treatment.
Hypertension is a key risk factor for both ischaemic stroke and cerebral haemorrhage. More than half of our patients suffered from hypertension.
Over a number of decades, the prognosis for stroke patients has improved (18). This mainly applies to ischaemic stroke. Spontaneous cerebral haemorrhage remains a serious condition with high mortality. In our material, the 30-day mortality amounted to 39.6 %. This concurs with the other Norwegian studies from the Innherred district (37.8 %) (3) and the country’s southernmost region (36.6 %) (4).In an international meta-analysis, median mortality after 30 days amounted to 40.4 % (2). In another review article, 30-day mortality amounted to 42 % (19). In a large retrospective Finnish study, altogether 51 % of the patients had died after 28 days (20). It might seem as though the mortality from spontaneous brain haemorrhage has remained fairly constant over several decades (2).
A previous material from a large international study showed that patients with a haemorrhage in the right brain hemisphere had a higher risk of death within 90 days than those with a haemorrhage in the left hemisphere (21). No such difference was found in our material.
Most patients with a spontaneous intracerebral haemorrhage are treated conservatively (22). A markedly elevated blood pressure may cause increased growth of haematomas and ought to be lowered. According to guidelines from the European Stroke Association (ESO) from 2014, it is safe to lower the systolic blood pressure to < 140 mm Hg within six hours after symptom onset, and this is possibly better than a systolic target of < 180 mm Hg (23). However, this is a tentative recommendation.
In warfarin-associated haemorrhages, the anticoagulant treatment should be reversed and the INR values normalised wherever possible. For haemorrhages associated with the use of dabigatran an antidote is now available – idaruzisumab.
Surgical treatment is recommended for haemorrhages in the cerebellum and from arteriovenous malformations (24). External drainage is also relevant in cases of threatening hydrocephalus. The benefits of evacuating supratentorial haematomas are uncertain (25). A large randomised study showed no definitive effect of early surgery of supratentorial haematomas when compared to conservative treatment (25). Nor did a meta-analysis show any better effect of surgery when compared to conservative treatment of such haematomas (26).
Approximately half of those who survived their brain haemorrhage had few sequelae and returned to their homes without any need of extra help or supervision. These had a Barthel ADL index score of 19 or 20, meaning that they were self-reliant in their daily activities. Most of those who were living at home had an mRS value ≤ 2, which is deemed a good outcome after a brain stroke.
Conclusion
We found an incidence of spontaneous first-time intracerebral haemorrhage in Vestfold county of 20.5/100 000/year for the period 1 September 2010 – 31 May 2014. A large proportion of the patients received anticoagulant treatment or used platelet inhibitors prior to admission, and the mortality among these was higher than among those who had received no such treatment.
MAIN MESSAGE
The incidence of first-time intracerebral haemorrhages in Vestfold county amounted to 20.5/100 000/year during the period 1 September 2010 – 31 May 2014
Two out of five patients died within 30 days
The use of anticoagulation treatment upon admission was associated with a significantly increased 90-day mortality
- 1.
Norsk hjerneslagregister. Årsrapport 2014. https://stolav.no/norskhjerneslagregister (2.3.2017).
- 7.
Mahoney FI, Barthel DW. Functional evaluation: the Barthel index. Md State Med J 1965; 14: 61 – 5. [PubMed]
- 8.
Statistisk sentralbyrå. Folkemengde og befolkningsendringar. https://www.ssb.no/statistikkbanken/SelectVarVal/Define.asp?MainTable=Rd0002AaX5&KortNavnWeb=folkemengde&PLanguage=0&checked=true (15.1.2017).
- 9.
Eurostat task force. Revision of the European Standard Population. http://ec.europa.eu/eurostat/documents/3859598/5926869/KS-RA-13 – 028-EN.PDF (15.1.2017).
- 18.
Ellekjaer H, Selmer R. Hjerneslag–like mange rammes, men prognosen er bedre. Tidsskr Nor Lægeforen 2007; 127: 740 – 3. [PubMed]
- 24.
Salvesen R, Ingebrigtsen T. Spontan intracerebral blødning. Tidsskr Nor Lægeforen 2007; 127: 1064 – 8. [PubMed]
- 25.
Mendelow AD, Gregson BA, Fernandes HM et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the International Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 2005; 365: 387 – 97. [PubMed] [CrossRef]