Parkinson’s disease is a chronic progressive neurodegenerative disorder characterised by motor impairments in the form of tremor, rigidity and akinesia/bradykinesia (1, 2). The incidence of Parkinson’s disease in Norway is 12.6 per 100 000 inhabitants per year, similar to that reported in other Western European countries and North America (3). We therefore have no reason to believe that there are any particular differences in incidence across Norway, but this has never been specifically examined.
The incidence of Parkinson’s disease increases with age, as shown by a German study that found a prevalence of 1.7 % in those over 65 (4). Loss of dopaminergic neurons in the substantia nigra and the subsequent reduction in dopamine release in the striatum are central to the disease pathogenesis (5).
In the symptomatic treatment of Parkinson’s disease, efforts are made to correct this dopamine deficiency (1, 6). Oral levodopa and/or dopamine agonists are effective in early stages of the disease. After a couple of years of levodopa therapy, however, many patients develop motor fluctuations in the form of varying motor function, and/or dyskinesias, i.e. involuntary movements (7). Recent Norwegian figures show that more than 50 % of patients develop motor fluctuations within five years of diagnosis (8). This occurs despite the use of multiple drugs to counteract fluctuations in efficacy (6).
The reason that many patients nevertheless experience fluctuations is in part because the dopamine storage capacity of the brain gradually declines as the disease progresses, which means that the effects of levodopa are shorter-lived (9). This can be countered to some degree by using smaller but more frequent doses of levodopa, to avoid large fluctuations in drug concentration. But as the disease progresses and the therapeutic window becomes narrower, many patients experience disabling motor fluctuations despite frequent dosing. It is in these cases that advanced therapies are appropriate.
There are three such therapies available for advanced Parkinson’s disease: continuous infusion therapy with a levodopa-carbidopa intestinal gel (Duodopa), continuous infusion therapy with the dopamine agonist apomorphine, and deep brain stimulation. In levodopa-carbidopa infusion therapy, a medicated gel is introduced into the small intestine via a percutaneous endoscopic gastrostomy with a jejunal extension tube (PEG-J) (10). Apomorphine is infused subcutaneously, usually at an abdominal site (11). Deep brain stimulation involves implanting electrodes in the brain, usually bilaterally in the subthalamic nuclei, for low voltage, high frequency electrical stimulation (12). The electrodes are connected to an external programmable neurostimulator positioned subcutaneously on the chest wall (12).
All three methods are effective in reducing motor fluctuations (13) – (16). Treatment helps to maintain patients in a stable «on» phase (16). Deep brain stimulation may also be effective against drug-resistant tremor (13) – (16).
Both Norwegian and international treatment guidelines recommend that advanced therapy be considered when patients with Parkinson’s disease experience disabling motor fluctuations despite optimised oral medication (6, 13, 14). This may be, for example, if they experience fluctuations despite more than five daily doses of levodopa (15).
Which form of advanced therapy to choose is less clear, however, and there have been no randomised controlled trials comparing all three options. In a number of cases there may be contraindications for one or more of the advanced therapies; these may include advanced age, dementia, psychiatric disorders, and cerebrovascular or bowel disease, but often any of the three forms would be possible (14) – (16).
Since 2012, deep brain stimulation has been defined in Norway as a multi-regional or national service, available at Oslo University Hospital, Rikshospitalet and St. Olavs Hospital (17). Prior to 2012, deep brain stimulation was also performed at Haukeland University Hospital, and in an earlier period at Stavanger University Hospital.
By contrast, infusion therapy with levodopa-carbidopa intestinal gel or apomorphine is not geographically regulated in Norway. Many departments of neurology in Norway offer the former, whereas infusion therapy with apomorphine has been little used due to subcutaneous side effects resulting from apomorphine cytotoxicity (11). However, use of apomorphine infusion does appear to have increased somewhat in Norway following changes to the infusion system and improvements in the treatment of subcutaneous side effects.
Differences in how the various advanced therapies for Parkinson’s disease are regulated has led to uncertainty over whether patients in different parts of the country receive the same treatment options. We therefore wished to determine whether there were geographical differences at county level in the proportion of patients receiving either of the two standard forms of advanced therapy: deep brain stimulation and levodopa-carbidopa infusion therapy.
Apomorphine therapy was not included in our study since fewer than ten patients were estimated to have started apomorphine infusion therapy during the study period. Moreover, it is not possible to identify these patients as the Norwegian Prescription Database does not distinguish between apomorphine used for infusion therapy and apomorphine used in subcutaneous injections, and the pump suppliers do not have a suitable register either.
To the best of our knowledge, there have been no previous studies of geographical differences in advanced therapy for Parkinson’s disease in Norway or elsewhere.
Results
In the study period, a total of 262 patients with Parkinson’s disease began treatment with deep brain stimulation (146 patients) or infusion therapy with levodopa-carbidopa intestinal gel (116 patients). The distribution of these patients across counties is shown in Table 1 and Figure 1.
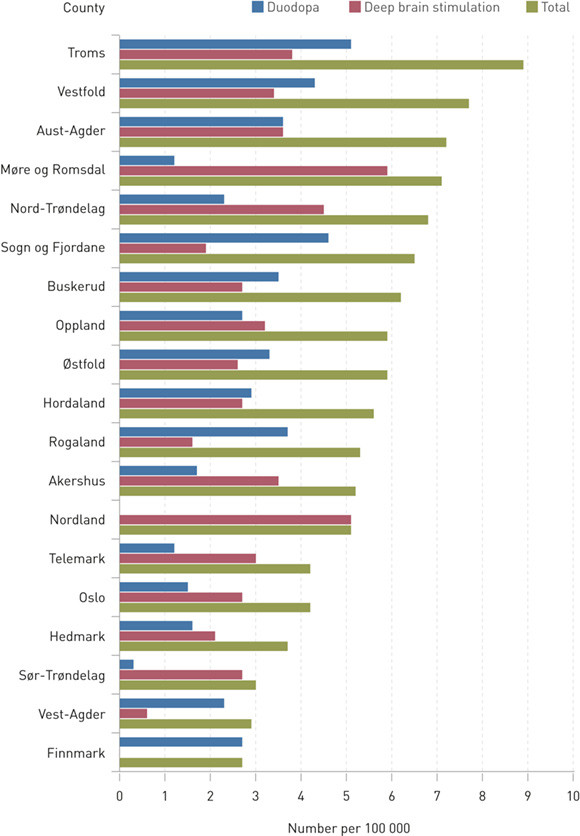
Table 1
The proportion of patients per county who received deep brain stimulation as a percentage of new patients who received deep brain stimulation or infusion therapy with levodopa-carbidopa intestinal gel (Duodopa) in the period 2009 – 2013
| County |
Percentage deep brain stimulation (%) |
| Finnmark |
0 |
| Troms |
43 |
| Nordland |
1001 |
| Nord-Trøndelag |
66 |
| Sør-Trøndelag |
901 |
| Møre og Romsdal |
831 |
| Sogn og Fjordane |
29 |
| Hordaland |
48 |
| Rogaland |
302 |
| Vest-Agder |
21 |
| Aust-Agder |
50 |
| Telemark |
71 |
| Vestfold |
44 |
| Buskerud |
44 |
| Akershus |
67 |
| Oslo |
64 |
| Oppland |
54 |
| Hedmark |
57 |
| Østfold |
44 |
1 Indicates significantly greater proportion of deep brain stimulation than in other counties
2 Indicates significantly greater proportion of Duodopa
Deep brain stimulation. Of 146 patients, 92 (63 %) underwent surgery at Oslo University Hospital, 38 (26 %) at St. Olavs Hospital and 16 (11 %) at Haukeland University Hospital. Haukeland University Hospital ceased performing deep brain stimulation when the multi-regional treatment service was established in 2012; the last operation took place in May 2012. The distribution of target areas for electrode placement was as follows: subthalamic nucleus in the mesencephalon (STN) 84.2 %, VIM nucleus of the thalamus (ventral intermediate nucleus) 11.6 % and GPi (internal segment of the globus pallidus) 4.1 %.
The number of operated patients per 100 000 inhabitants in the study period ranged from 5.9 (Møre og Romsdal) to 0 (Finnmark), with an average across counties of 2.9. Finnmark was the only county in which no new patients with Parkinson’s disease received deep brain stimulation during the study period.
Levodopa-carbidopa intestinal gel. The number of patients receiving this type of infusion therapy varied from 5.1 per 100 000 inhabitants (Troms) to 0 (Nordland). The average number of patients treated was 2.6 per 100 000 inhabitants.
Advanced therapies combined. During the study period, the number of patients with Parkinson’s disease who received either of these two forms of advanced therapy ranged from 8.9 per 100 000 inhabitants in Troms to 2.7 per 100 000 inhabitants in Finnmark. The average for the whole of Norway was 5.5 patients per 100 000, or 1.1 per 100 000 inhabitants per year.
Statistical analyses. We found a significant difference in the county-wise distribution of deep brain stimulation and infusion therapy with levodopa-carbidopa intestinal gel (p = 0.00078 by exact test). The counties with an uneven distribution were Møre og Romsdal, Nordland, Sør-Trøndelag and Rogaland. In the first three counties, usage was skewed towards more deep brain stimulation. In Rogaland, the opposite was true; that is, relatively more patients received infusion therapy (Table 1).
Since we do not have data for infusion therapy with apomorphine, we do not know exactly how many patients with Parkinson’s disease received one of the three types of advanced therapy over the course of the study period. This means that although the figures apparently show a difference between counties, we cannot present statistics on any county-wise differences in the overall provision of advanced therapy.
Material and method
We included all patients with Parkinson’s disease who began advanced therapy in the form of levodopa-carbidopa intestinal gel or deep brain stimulation in the period 2009 – 2013: a total of 262 persons. The study was approved by the Norwegian Data Protection Authority.
County of residence was recorded on the basis of residential address at the start of treatment. Lists of all those who underwent initial electrode implantation for deep brain stimulation in the study period at Oslo University Hospital, St. Olavs Hospital and Haukeland University Hospital were obtained from the respective hospitals’ episode statistics and then anonymised. Anonymised data on the total number of patients with Parkinson’s disease who began infusion therapy with levodopa-carbidopa intestinal gel in each county in the same period were obtained from the Norwegian Institute of Public Health’s Prescription Database. The figures reveal where the patients lived and are independent of where the probe insertion procedure was carried out.
We used population statistics from Statistics Norway for 2011 to calculate the number of patients treated per 100 000 inhabitants in each county. According to Statistics Norway, the sex ratio did not differ across counties in the study period and differences in age distribution were relatively small (18). We therefore opted not to calculate standardised population rates.
We examined the relative use of deep brain stimulation and levodopa-carbidopa infusion therapy in each county. Statistical analyses were performed using IBM SPSS statistics 22 for Mac. The null hypothesis was that there were no systematic differences between the counties in the relative proportion of patients who began infusion therapy versus deep brain stimulation, and that any observed variation can be explained by random fluctuations.
This hypothesis was tested using an exact test with 100 000 Monte Carlo permutations, where the significance level was set to p < 0.05. The test generates an empirical p‑value for the observed distribution of therapies in a 19 × 2 table (counties × therapies). If the null hypothesis is rejected at the given significance level, the test also identifies the largest possible cluster of cells in the 19 × 2 table that together show a distribution consistent with the null hypothesis. The remaining cells, characterised by outliers that fall outside this distribution, reveal those counties that differ significantly from the rest.
Discussion
While there would appear to be substantial variation between counties in the number of patients per 100 000 inhabitants who, over the course of the study period, began advanced therapy with either deep brain stimulation or levodopa-carbidopa infusion (from 8.9 in Troms to 2.7 in Finnmark), the number of patients is small. Moreover, we lack data for infusion therapy with apomorphine. We therefore cannot say for certain that some counties offer advanced therapy to fewer patients than others. It is nevertheless noteworthy that Finnmark, the only county in Norway without its own department of neurology, had the lowest proportion of patients receiving advanced therapy.
The differences in the provision of deep brain stimulation and levodopa-carbidopa infusion therapy are another matter. According to Statistics Norway, any differences in age distribution between the various counties were relatively small during the study period, so this should not have affected the results. Nor do we have any evidence to suggest that there are marked differences between counties in the prevalence of Parkinson’s disease, or in the prevalence of patients in need of advanced therapy in Norway. Our findings therefore suggest that the two forms of advanced therapy investigated are used to differing degrees across counties.
It is important that patients throughout the country receive equal treatment, and observations from our study can hopefully contribute to this by detecting and highlighting that there are in fact differences in the provision of treatment at present.
County size should not affect the choice of treatment, but small sample sizes in terms of number of inhabitants and of treated patients did affect our calculations and make it difficult to achieve statistical significance. This is illustrated by the fact that the relative use of deep brain stimulation and levodopa-carbidopa infusion therapy in Finnmark was not significantly different from that in other counties, despite the fact that no patients from Finnmark received deep brain stimulation during the study period.
There are no clear guidelines on when to offer advanced therapy or on what form of advanced therapy should be preferred in individual cases (14) – (16). Both deep brain stimulation and infusion therapy are often started later than is desirable, and the general view now is that advanced therapy should be offered as soon as there is an indication for it (15, 19).
In many cases, all three forms of advanced therapy are valid options. Many different factors may then influence which type of therapy is offered, including patient preference, clinical judgement, local availability, knowledge and expertise, the doctor’s personal experiences and preferences, price, and political guidelines.
Cost analyses have shown that deep brain stimulation requires fewer health resources and is a less expensive option than infusion therapy (20), but it is unclear to what extent this affects choice of therapy in Norway, where both options are reimbursed. The reason why a significantly greater proportion of patients receive deep brain stimulation in Møre og Romsdal, Nordland and Sør-Trøndelag, and levodopa-carbidopa infusion therapy in Rogaland, remains unclear. Stavanger University Hospital used to perform deep brain stimulation prior to the study period, and one possibility is that closure of this facility may have had an impact on the choice of advanced therapy for patients from Rogaland.
Although Sør-Trøndelag has a relatively high proportion of patients receiving deep brain stimulation and is also one of two counties in which this treatment is administered, there is no evidence that this multi-regional service is skewed in favour of the counties or regional health authorities that contain the treatment centres. The number of patients with Parkinson’s disease who underwent deep brain stimulation in the study period was 2.7 per 100 000 inhabitants in both Sør-Trøndelag and in Oslo, and 2.7 was also the median for all Norwegian counties. Hordaland, which performed this type of surgery throughout much of the study period, had a median of 2.7 as well. We accordingly have no grounds to suspect that differences in waiting times between surgical centres have any appreciable impact on choice of treatment.
It is also unlikely that surgical centres differ greatly in their assessment of the indication for deep brain stimulation, although the proportion of patients that have received deep brain stimulation in Sør-Trøndelag is high. Patients from Møre og Romsdal and from Nordland who received deep brain stimulation were treated in both Trondheim and Oslo.
Differences in choice of therapy may possibly reflect regional differences in the information given to patients. When advanced therapy is required, it is the patient who should have the final say in the choice of method (15). A Swedish study found that patients with advanced Parkinson’s disease often had limited knowledge, and moreover had received inadequate information from their doctor about the various types of advanced therapy (21), so that they had insufficient grounds on which to make an independent choice. But irrespective of knowledge, it is the patient who must make the final decision, and it is therefore very important that all affected patients receive the best possible information about the various options.
Almost all counties administered both treatments during the study period. The exceptions are Nordland, where only deep brain stimulation was used, and Finnmark, where patients received only levodopa-carbidopa infusion therapy. However, the numbers from each county are small, and this does not mean that patients from these two counties were not given the opportunity to choose the other therapy type.
We still do not know what proportion of patients with Parkinson’s disease should be offered advanced therapy, or what would constitute the optimal relative usage of the various therapy types, clinically or socioeconomically (15). Therefore, we cannot judge which counties provide optimal treatment. But it is important that patients – before making their choice – receive a thorough introduction to all three methods and their respective advantages and disadvantages (6, 15).
Parts of this study are based on an embargoed student dissertation completed by the first author.
MAIN POINTS
We found clear geographical differences in the type of advanced therapy used in cases of Parkinson's disease
The use of deep brain stimulation and levodopa-carbidopa intestinal gel infusion therapy varied across counties
We found no evidence to suggest that national centres for deep brain stimulation prioritised patients from their own region.
- 1.
Larsen JP, Beiske AG, Bekkelund SI et al. Motoriske symptomer ved Parkinsons sykdom. Tidsskr Nor Legeforen 2008; 128: 2068 – 71. [PubMed]
- 6.
Nasjonalt kompetansetjeneste for bevegelsesforstyrrelser. Veiledende retningslinjer for diagnostisering og behandling ved Parkinsons sykdom. Revidert 2010. https://www.helse-stavanger.no/no/OmOss/Avdelinger/nasjonalt-kompetansesenter-for-bevegelsesforstyrrelser/Documents/Behandlingsplaner/PSK%20-%20Retningslinjer%20for%20diagnostisering%20og%20behandling%20ved%20Parkinsons%20sykdom.pdf (17.4.2016).
- 9.
Sage JI, Mark MH. Basic mechanisms of motor fluctuations. Neurology 1994; 44 (suppl 6): S10 – 4. [PubMed]
- 12.
Toft M, Lilleeng B, Ramm-Pettersen J et al. Behandling av bevegelsesforstyrrelser med dyp hjernestimulering. Tidsskr Nor Legeforen 2008; 128: 1972 – 6. [PubMed]
- 15.
Odin P, Ray Chaudhuri K, Slevin JT et al. Collective physician perspectives on non-oral medication approaches for the management of clinically relevant unresolved issues in Parkinson's disease: Consensus from an international survey and discussion program. Parkinsonism Relat Disord 2015; 21: 1133 – 44. [PubMed] [CrossRef]
- 17.
Regjeringen.no. Nasjonale tjenester i spesialisthelsetjenesten. Veileder til Forskrift nr. 1706 av 17. Desember 2010 om godkjenning av sykehus, bruk av betegnelsen universitetssykehus og nasjonale tjenester i spesialisthelsetjenesten. 2015. https://www.regjeringen.no/contentassets/3b2da09c60e9471c9975fe566416017d/veileder_nasjonale_tjenester_for_innspill.pdf (17.4.2016).
- 18.
Statistisk sentralbyrå. Folke- og boligtellingen, hovedtall, 2011. https://www.ssb.no/befolkning/statistikker/fobhoved (14. 2.2017).