Reducing overconsumption and misuse of antibiotics, and the broad-spectrum types in particular, is crucial for limiting the prevalence of antibiotic-resistant microbes. Broad-spectrum antibiotics should only be used in cases of severe illness (1, 2).
When seen in an international context, the situation in Norway is relatively favourable (3), but resistance to antibiotics is nevertheless on the rise (1).
One of the goals in the Action plan against antibiotics resistance in the health services is to reduce the consumption of broad-spectrum antibiotics in hospitals by 30 % before 2020 compared to 2012 levels (2). The sales of broad-spectrum antibiotics to hospitals were reduced by 6 % between 2012 and 2015 (1). This may indicate a higher awareness related to the use of these drugs in Norwegian hospitals. The national guidelines for the use of antibiotics in hospitals (hereafter referred to as «the guidelines»), published in June 2013, may have contributed to this reduction (4).
Sales figures provide an overview of antibiotics procured, while prevalence surveys of antibiotic use provide an overview of drugs that have been prescribed and the indications for prescribing them. This enables an assessment of whether the prescribing practices were in accordance with the guidelines. The prevalence surveys of antibiotic use are organised by the Norwegian Institute of Public Health semi-annually (5), and have been mandatory since May 2015.
The objective of this paper was to describe how the broad-spectrum antibiotics referred to in the action plan were used in Norwegian hospitals. We also wanted to assess whether this use complied with the guidelines, and whether there was potential for further reductions in antibiotic use.
Material and method
We used data from the third nationwide mandatory prevalence survey. All ongoing systemic courses of antibiotics (except tuberculostatics) at 08: 00 on 11 May 2016 were registered according to a template found on the website of the Norwegian Institute of Public Health (6). The total number of hospitalised patients was recorded.
When antibiotics were prescribed, the registration included the patients’ gender and age group, indication for the prescription, whether the drug was administered for purposes of treatment or prophylaxis, and the prescribing doctor’s specialty. The registration also included whether antibiotics were administered against healthcare-associated infections – defined as infections that occurred two days or later after admission to hospital – or post-operative infections. All other infections were registered as community-acquired infections. The duration of the courses of antibiotics and comorbidity were not registered.
Forms were completed by a doctor or pharmacist in each hospital department (6). Quality control was performed by an infection control practitioner (doctor or nurse) in each hospital, before data were submitted electronically to the Norwegian Institute of Public Health.
Broad-spectrum antibiotics have been defined in accordance with the action plan as piperacillin with enzyme inhibitor, second and third generation cephalosporins, quinolones and carbapenems (2).
The data were collected under the Regulations for the Norwegian surveillance system for antibiotic use and healthcare-associated infections (5). No further approvals were required.
The diagrams and estimates were made with the aid of Excel 2013 MSO (Microsoft Corp., Washington, USA).
Results
All antibiotics prescribed
For the survey in May 2016, all health institutions in Norway submitted data. Results from 58 hospitals were included. A total of 10 085 patients were registered as hospitalised on the day of the survey, of whom 2 780 (28 %) were given 3407 courses of systemic antibiotics (Figure 1). A total of 674 prescriptions for treatment of healthcare-associated infections were issued for 526 patients, implying a prevalence of 5.2 %.
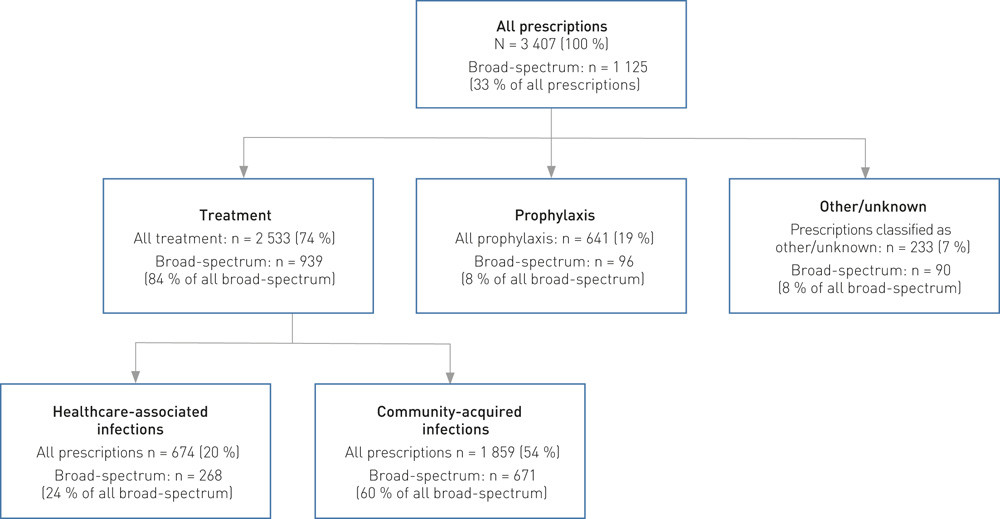
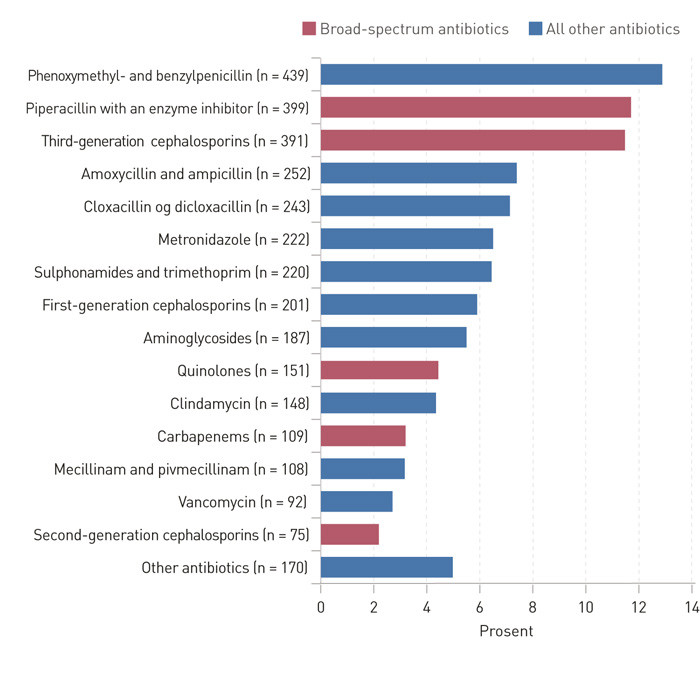
Broad-spectrum antibiotics accounted for 33 % of all prescriptions of antibiotic drugs (Figure 1). Out of 1 125 prescriptions of broad-spectrum drugs, 939 (84 %) were administered as treatment and 96 (8 %) as prophylaxis, whereas 90 (8 %) were classified as unknown/other (Figure 1). Of the broad-spectrum antibiotics, piperacillin with an enzyme inhibitor and third-generation cephalosporins were most frequently prescribed, in total accounting for 23 % of all prescriptions.
Treatment with broad-spectrum antibiotics
A total of 939 prescriptions of broad-spectrum antibiotics were issued for 916 patients. Of these, 893 received one broad-spectrum drug, alone or in combination with narrow spectrum antibiotics, while 23 patients received a combination of two broad-spectrum antibiotics (Table 1).
Table 1
Treatment (not including prophylaxis) for various indications in Norwegian hospitals on 11 May 2016 (n = 2 533 prescriptions/2 097 patients)
| Indication |
Number of prescriptions |
Number of patients treated |
Broad-spectrum antibiotics, number of patients (%)1 |
Narrow-spectrum antibiotics recommended in the guidelines2, number of patients (%) |
Other narrow-spectrum treatment3, number of patients (%) |
| Community-acquired lower respiratory tract infection |
583 |
515 |
185 (36) |
171 (33)4 30 (6)5 |
129 (25) |
| Healthcare-associated lower respiratory tract infection |
129 |
117 |
85 (73) |
17 (14)4 3 (3)5 |
12 (10) |
| Intra-abdominal infection |
214 |
171 |
136 (80) |
23 (13)6 |
12 (7) |
| Bloodstream infection, septicaemia and neutropenic fever |
303 |
225 |
121 (54) |
43 (19)5 |
61 (27) |
| Infection with an uncertain focus without septicaemia/unknown |
193 |
180 |
96 (53) |
84 (47) |
|
| Surgical-site infection |
242 |
164 |
79 (48) |
85 (52) |
|
| Lower urinary tract infection |
232 |
220 |
55 (25) |
112 (51)7 9 (4)8 |
44 (20) |
| Upper urinary tract infection |
117 |
98 |
53 (54) |
13 (13)7 13 (13)8 |
19 (20) |
| Skin and soft-tissue infection |
223 |
193 |
49 (26) |
97 (50)9 |
47 (24) |
| Other infections10 |
297 |
214 |
57 (27) |
157 (73) |
|
| Total |
2 533 |
2 097 |
916 (44) |
531 (25) |
650 (31) |
| [i] | |||||
[i] 1 Either alone or in combination with other antibiotics
2 Recommended in the guidelines for uncomplicated infections with an unknown microbe
3 Monotherapy or combination therapy
4 Penicillin or amoxycillin/ampicillin alone
5 Penicillin or amoxycillin/ampicillin in combination with an aminoglycoside
6 Ampicillin, aminoglycoside, metronidazole, trimethoprim-sulfa – two or more of these in combination
7 Piv-/mecillinam, trimethoprim/trimethoprim-sulfa
8 Ampicillin and aminoglycoside in combination
9 Penicillin, clindamycin, cloxa-/dicloxacillin, either alone or in combination
10 Other infections: Infections in bones and joints (19 broad-spectrum prescriptions), infections in the central nervous system (n = 17), infections in the cardiovascular system (n = 11), infections in the eye, ear, nose, throat or mouth (n = 8), gastroenteritis except antibiotic-associated diarrhoea (n = 2), obstetric or gynaecological infection, including sexually transmitted infections in women (n = 1), prostatitis, epididymitis, including sexually transmitted infections in men (n = 1)
Broad-spectrum antibiotics accounted for 268 of 674 prescriptions (40 %) for treatment of healthcare-associated infections. Treatment of community-acquired infections accounted for 671 of a total of 1 859 prescriptions (36 %) (Figure 1).
Both the total number of prescriptions and the proportion of broad-spectrum antibiotics increased with increasing age of the patient. In children and adolescents (0 – 19 years), the broad-spectrum antibiotics accounted for 52 out of 246 prescriptions (21 %), in the age group 20 – 29 years they accounted for 27 out of 101 prescriptions (27 %). In patients aged 30 years or older, this applied to 859 out of 2 192 prescriptions (39 %). Altogether 458 (49 %) of all courses of broad-spectrum antibiotics were administered to patients who were 70 years or older. For six prescriptions the age of the patient was not recorded, including one for broad-spectrum antibiotics.
Among men, 542 out of 1 388 antibiotic treatments (35 %) involved broad-spectrum drugs, while in women this applied to 397 out of 1 145 prescriptions (30 %).
Internal medicine specialists prescribed 526 of the broad-spectrum antibiotics (56 %), surgeons 289 (31 %) and other specialists 124 (13 %). Doctors at internal medicine departments prescribed third-generation cephalosporins most frequently – 239 prescriptions, 72 % of all prescriptions of this group of antibiotics. Doctors in surgical departments mostly prescribed piperacillin with an en-zyme inhibitor – 166 prescriptions, 49 % of all prescriptions of this drug.
Indications for treatment with broad-spectrum antibiotics
Among patients with community-acquired lower respiratory tract infections, altogether 185 out of 515 (36 %) were treated with at least one broad-spectrum antibiotic. For healthcare-associated lower respiratory tract infections, this proportion amounted to 85 out of 117 (73 %) (Table 1). For all lower respiratory tract infections combined, 282 out of 712 prescriptions (40 %) involved broad-spectrum antibiotics. These 282 prescriptions accounted for 30 % of all broad-spectrum antibiotic treatment. The most frequently used drugs were third-generation cephalosporins and piperacillin with an enzyme inhibitor, which accounted for 49 % and 33 % respectively of the prescriptions of broad-spectrum drugs for this indication.
Altogether 136 out of 171 patients (80 %) were treated with broad-spectrum antibiotics for intra-abdominal infections (Table 1). Piperacillin with an enzyme inhibitor accounted for 74 % of these prescriptions.
Broad-spectrum antibiotics were administered to 121 out of 225 patients (54 %) with septicaemia, bloodstream infections or neutropenic fever (Table 1). Here, third-generation cephalosporins accounted for 43 % and piperacillin with an enzyme inhibitor for 27 % of the broad-spectrum prescriptions.
Broad-spectrum antibiotics were administered for treatment of 108 patients with urinary tract infections. Approximately the same number were treated for lower (n = 55) and upper (n = 53) urinary tract infections. These accounted for 25 % and 54 % respectively of all patients with these two indications. Here, third-generation cephalosporins accounted for 50 out of 109 (46 %) prescriptions of broad-spectrum drugs.
Microbiological testing
A microbiological sample was collected prior to 678 out of 939 prescriptions (72 %) of broad-spectrum antibiotics, and prior to 1 237 out of 1 594 prescriptions (78 %) of other antibiotics.
This proportion was highest for cases of septicaemia, bloodstream infections and neutropenic fever, for which a microbiological sample was collected prior to 93 % of the prescriptions of broad-spectrum antibiotics, and 87 % of the prescriptions of other antibiotics. The lowest proportion was for cases of intra-abdominal infection, with 50 % and 52 % of the prescriptions respectively. For the remaining indications, the proportion varied between 65 % and 80 %.
Prophylaxis
A total of 96 out of 641 prophylactic prescriptions (15 %) involved broad-spectrum antibiotics. Altogether 77 of these were administered as surgical prophylaxis, whereof 36 (47 %) involved piperacillin with an enzyme inhibitor. There were 19 prescriptions of broad-spectrum antibiotics for purposes of medical prophylaxis, whereof 12 (63 %) involved third-generation cephalosporins.
Discussion
The proportion of patients who received antibiotics and the proportion who received broad-spectrum antibiotics in our survey were similar to findings in other Scandinavian countries (7, 8). In most other countries, both of these proportions were significantly higher (9) – (11).
Differences in terms of the occurrence of resistant bacteria and varying prescribing practices make it difficult to compare antibiotic use in Norway with practices outside of Scandinavia (3). In all studies, however, lower respiratory tract infections were the most frequent indication for treatment with antibiotics (7) – (11).
Treatment of lower respiratory tract infections
The guidelines recommend penicillin as the drug of choice for non-severe community-acquired pneumonia. For severe pneumonia, penicillin in combination with an aminoglycoside or cefotaxime are recommended.
In cases of exacerbation of COPD, amoxycillin and ampicillin are also recommended (4). Despite this, in this survey we found a considerable prescribing of third-generation cephalosporins and piperacillin with an enzyme inhibitor for treatment of community-acquired lower respiratory tract infections. The frequent prescribing of third-generation cephalosporins may be due to the fact that the guidelines can be perceived as recommending cefotaxime as equivalent to penicillin in combination with aminoglycosides as the drugs of choice for severe pneumonia.
The proportion of patients with severe pneumonia in this survey is unknown. In a Norwegian study from 2016, the proportion of patients with severe pneumonia amounted to 8 – 10 % (12). This is significantly lower than the proportion of patients who received broad-spectrum antibiotics as treatment for a lower respiratory tract infection in this survey.
The guidelines recommend piperacillin with an enzyme inhibitor as an alternative, and not as a first choice, for treatment of ventilator-associated pneumonia, severe aspiration pneumonia or suspicion of multidrug-resistant bacteria (4). The proportion of patients with these indications in this study is unknown, but they commonly account for a small minority of all patients with infections in the lower respiratory tract (2, 12).
For healthcare-associated pneumonia, either penicillin (in combination with aminoglycoside) or cefotaxime are recommended. The proportion of patients who received broad-spectrum antibiotics was significantly higher in the treatment of lower respiratory tract infections that were healthcare-related, rather than community-acquired. Accordingly, the use of broad-spectrum antibiotics may be reduced by preventing infections in the lower respiratory tract. One study has claimed that at least 20 % of all healthcare-associated infections are likely to be preventable (13).
This analysis indicates that the use of broad-spectrum antibiotics against infections in the lower respiratory tract in Norwegian hospitals can be reduced.
Treatment of intra-abdominal infections
The frequent prescribing of piperacillin with an enzyme inhibitor to patients with an intra-abdominal infection may be due to the fact that the guidelines define this as a drug of choice, equivalent to ampicillin/aminoglycosides/trimethoprim-sulfamethoxazole, optionally in combination with metronidazole (4).
The proportion of prescriptions for broad-spectrum antibiotics is, however, significantly higher than the proportion for the recommended narrow-spectrum antibiotics for this indication. This may indicate that broad-spectrum drugs are preferred over the narrow-spectrum alternatives when these are ranked equally in the guidelines.
Treatment of septicaemia, bloodstream infections and neutropenic fever
In cases of septicaemia, bloodstream infections and neutropenic fever, penicillin in combination with an aminoglycoside is the recommended treatment. When there is suspicion of an origin in the urinary tract, ampicillin and an aminoglycoside are recommended; when there is suspicion of an origin in the abdomen, the addition of metronidazole is recommended (4). In spite of this, both third-generation cephalosporins and piperacillin with an enzyme inhibitor were relatively frequently prescribed for treatment of these indications.
According to the guidelines, cefotaxime, piperacillin with an enzyme inhibitor and ciprofloxacin can be administered to patients with these indications in cases of severe renal failure (4, 14, 15). It is difficult to assess whether the use of broad-spectrum antibiotics in our study complied with the guidelines, since the patients’ renal function was unknown.
Treatment of urinary tract infections
Similarly, it is difficult to conclude whether there is a potential for reduced use of broad-spectrum antibiotics in treatment of lower urinary tract infections and whether the guidelines are being complied with, even though none of the broad-spectrum drugs is recommended as a drug of choice in the guidelines (4).
The fact that the highest proportion of broad-spectrum drugs is for cases of upper urinary tract infections seems to be in compliance with the guidelines, since cefotaxime is recommended as equivalent to ampicillin in combination with an aminoglycoside.
Microbiological testing
Microbiological testing occurred least frequently for indications where collecting a sample at the site of the infection was difficult or impossible, as in intra-abdominal infections. It is nevertheless desirable to collect a relevant microbiological sample as often as possible before initiation of antibiotic treatment.
Prophylaxis
This survey identified instances where broad-spectrum antibiotics were used as prophylaxis, even though the guidelines do not envisage such use. For prophylactic use, the recommendations specify antibiotics other than those used for treatment, in order to avoid development of resistance against antibiotic drugs that are essential for treatment.
Methodological considerations
This point prevalence survey provided an overview of the indications for use of antibiotics, which enabled us to assess their use in relation to the guidelines. We do not know, however, what criteria the doctors have applied upon registration of the indication. This survey does not allow assessment of the occurrence of unnecessary prescribing of antibiotics; we have only examined the types of antibiotics used once the decision to initiate treatment had been taken.
One disadvantage of point prevalence surveys is that random events on the day of the survey may have an impact on the results. This methodological weakness presents a challenge when the number of registrations is low. The number of patients included in this study and the number of prescriptions for antibiotics were both high. Our findings are also corroborated by findings in previous prevalence surveys of antibiotic use in Norwegian hospitals (6). Other equivalent prevalence surveys have also produced robust results (7) – (10).
This study may indicate that broad-spectrum antibiotics are preferred above narrow-spectrum drugs wherever these are ranked equally, even though the guidelines in general recommend the use of narrow-spectrum rather than broad-spectrum drugs (4). A Cochrane review from 2013 and three recent Scandinavian studies showed that the use of antibiotics, including broad-spectrum drugs, can be reduced without any negative consequences for the patients (12, 16) – (18). This could encourage a revision of the guidelines with a clearer prioritisation of narrow-spectrum antibiotics over broad-spectrum drugs.
MAIN MESSAGE
This point prevalence survey from spring 2016 identified that broad-spectrum antibiotics highlighted in the Action plan against antibiotics resistance in the health services accounted for 33 % of all antibiotics prescribed in Norwegian hospitals.
Lower respiratory tract infection was the most frequent indication for treatment with broad-spectrum antibiotics, and accounted for 30 % of all treatment with broad-spectrum antibiotics.
The results indicate that a reduction in the use of broad spectrum antibiotics is achievable, including in the treatment of lower respiratory tract infection as well as for prophylactic purposes.
- 1.
NORM/NORM-VET-årsrapport. 2015. Usage of Antimicrobial Agents and Occurrence of Antimicrobial Resistance in Norway. Oslo/Tromsø: Norsk overvåkningssystem for antibiotikaresistens hos mikrober (NORM), Veterinærinstituttet, Folkehelseinstituttet, 2016. https://unn.no/Documents/Kompetansetjenester,%20-sentre%20og%20fagr%C3%A5d/NORM%20-%20Norsk%20overv%C3%A5kingssystem%20for%20antibiotikaresistens%20hos%20mikrober/Rapporter/NORM_NORM-VET-2015.pdf (1.2.2017).
- 2.
Handlingsplan mot antibiotikaresistens i helsetjenesten. Oslo: Helse- og omsorgsdepartementet, 2015. https://www.regjeringen.no/contentassets/915655269bc04a47928fce917e4b25f5/handlingsplan-antibiotikaresistens.pdf (30.9.2016).
- 3.
EARS-Net. Antimicrobial resistance interactive database. ECDC – European Centre for Disease Prevention and Control. http://atlas.ecdc.europa.eu/public/index.aspx?Instance=GeneralAtlas (30.9.2016).
- 4.
Nasjonal faglig retningslinje for bruk av antibiotika i sykehus. Oslo: Helsedirektoratet, 2013. https://helsedirektoratet.no/retningslinjer/antibiotika-i-sykehus (30.9.2016).
- 5.
Forskrift om Norsk overvåkingssystem for antibiotikabruk og helsetjenesteassosierte infeksjoner (NOIS-registerforskriften). Oslo: Helse- og omsorgsdepartementet, 2005. https://lovdata.no/dokument/SF/forskrift/2005- 06-17-611 (30.9.2016).
- 6.
Folkehelseinstituttet. Sykehus: Prevalensundersøkelsene av helsetjenesteassosierte infeksjoner og antibiotikabruk i sykehus NOIS-PIAH. https://www.fhi.no/hn/helseregistre-og-biobanker/nois/om-prevalensundersokelsene-av-helse/ (30.9.2016).
- 14.
Lindemann PC, Haldorsen BC, Smith I et al. Aminoglykosider bør fortsatt brukes i empirisk sepsisbehandling. Tidsskr Nor Legeforen 2013; 133: 1054 – 5. [PubMed]
- 15.
Torfoss D, Høiby EA, Holte H et al. Penicillin og aminoglykosid ved febril nøytropeni. Tidsskr Nor Legeforen 2008; 128: 2738 – 40. [PubMed]
- 16.
Davey P, Brown E, Charani E et al. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst Rev 2013; 4: CD003543. [PubMed]