Myeloproliferative neoplasms are a group of disorders characterised by clonal growth in one or more haematopoietic cell lines. The group includes chronic myelogenic leukaemia, polycythaemia vera, essential thrombocytosis and primary myelofibrosis. Chronic myelogenic leukaemia has a different aetiology from the other three, and is associated with a reciprocal translocation between chromosome 9 and chromosome 22 which leads to the formation of a Philadelphia chromosome. Thus we distinguish between Philadelphia chromosome-positive and Philadelphia chromosome-negative myeloproliferative neoplasms.
The annual incidence of the two combined is about 2 – 3/100 000 (1) – (3). As polycythaemia vera and essential thrombocytosis have a low malignancy, their prevalence is higher than their incidence, at around 50/100 000 for both. Myelofibrosis is a more highly malignant disease with a poorer prognosis and a prevalence of around 5/100 000 (1) – (4).
The diseases are characterised by overproduction of mature, functional blood cells, from one or more of the myeloid cell lines in the bone marrow, often also in the liver and spleen. The clinical course is long, with a risk of thrombosis and haemorrhage and a tendency to the development of fibrosis and in rarer cases transformation into more highly malignant blood diseases such as myelodysplastic syndrome and acute myelogenic leukaemia (5).
The kinship among the Philadelphia chromosome-negative myeloproliferative diseases is also reflected in their molecular pathogenesis. The JAK2 gene is located in position 24 on chromosome 9 and codes for a tyrosine kinase that signals downstream to activate cytokine receptors (Fig. 1). Knowledge of the role of JAK2 in signalling pathways related to haematopoiesis led to the hypothesis that a mutation in this gene could be linked to pathogenesis.
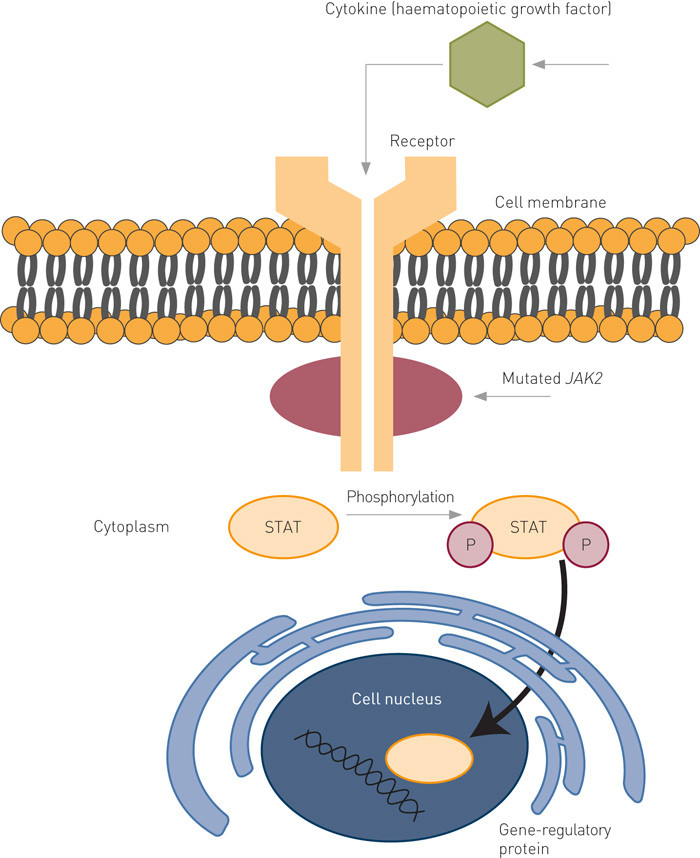
In 2005, four independent research groups found a somatic mutation in the JAK2 gene that caused a valine-to-phenylalanine substitution in codon 617, and hence increased tyrosine phosphorylation and activity through intracellular signalling pathways (6) – (10). The nomenclature is therefore written JAK2V617F, hereafter called the JAK2 mutation. The mutation makes haematopoietic cells more sensitive to growth factors such as erythropoietin and thrombopoietin, resulting in increased proliferation of myeloid cell lines. Detection of the JAK2 mutation was one of the World Health Organization (WHO)’s diagnostic criteria for myeloproliferative neoplasms in 2008 (11, 12).
Patients with a JAK2-positive myeloproliferative disorder have previously been found to have different phenotypic characteristics from patients with JAK2-negative disease (13) – (15). JAK2 mutation analysis was available in Norway in 2006. The purpose of the present study was to examine the prevalence of the mutation in a cohort of Norwegian patients who were assessed for myeloproliferative neoplasia, and to investigate whether detection of the mutation was characterised by particular biochemical or clinical factors.
Material and method
Since 2006, the Laboratory for Clinical Biochemistry, Haukeland University Hospital, has offered JAK2 mutation analysis, performed on DNA isolated from leukocytes in whole blood (16). The method is based on a real time polymerase chain reaction (PCR) (16), and the results are reported as allele burden (amount of mutated JAK2 as a percentage of total JAK2). The detection cut-off limit is 0.1 % and patients with results lower than this are defined as JAK2-negative. A low allele burden is defined as percentage ratio 0.1 < ∈ < 25, a high allele burden as percentage ratio 25 < ∈ < 100. In the period 2006 – 2008, the JAK2-mutation analysis was performed using a different PCR method, with a detection cut-off of 2 % (17).
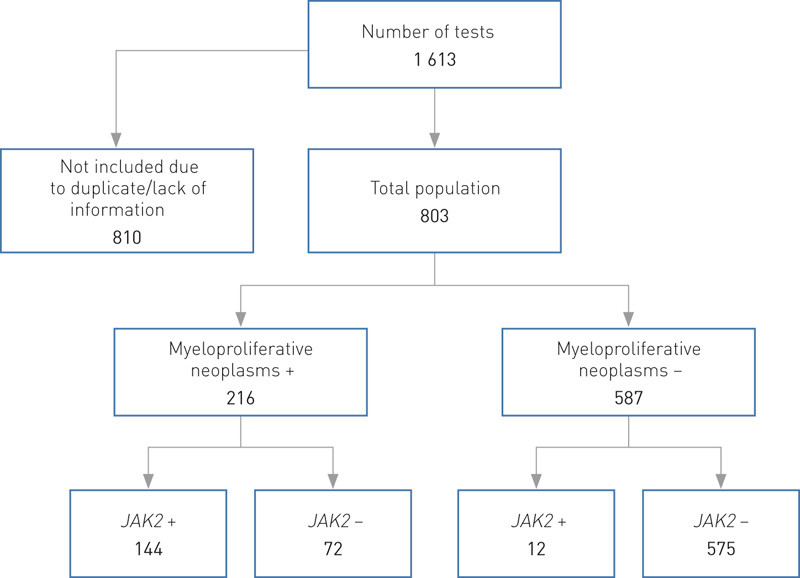
With the approval of the Regional Committee for Medical and Health Research Ethics (REC West, reference number 2011/2278) we gathered clinical information about patients for whom the JAK2-mutation analyses were performed in the period 2006 – 2012. Duplicates (same patient) and tests from individuals who were not patients of the Bergen Hospital Trust were not included in the 1 613 registered tests, as clinical information was only available from the electronic patient record system (DIPS) belonging to the Bergen Hospital Trust. The total study population was 803 patients.
Clinical data collected consisted of diagnoses made by the doctor providing treatment and the following variables at the time of diagnosis: age, mutation status, allele burden, haematological blood tests, smoking status and complications. A bone marrow biopsy and if relevant further diagnostic assessment were performed where the doctor providing treatment believed it was indicated.
The statistics software SPSS15.0 was used for calculations and graphical presentation. The chi-squared test, Fisher’s exact test and the t-test were used for statistical analyses. P-values of < 0.05 were regarded as statistically significant.
Results
Diagnosis of myeloproliferative neoplasms
Of the 803 patients, 156 (19.4 %) were found to have the JAK2 mutation. The number diagnosed on the basis of data from electronic patient records as having a myeloproliferative disorder was 216. Of these, 144 (66.7 %) were JAK2V617F-positive and 72 (33.3 %) were JAK2V617F-negative (Fig. 2).
Diagnostic distribution
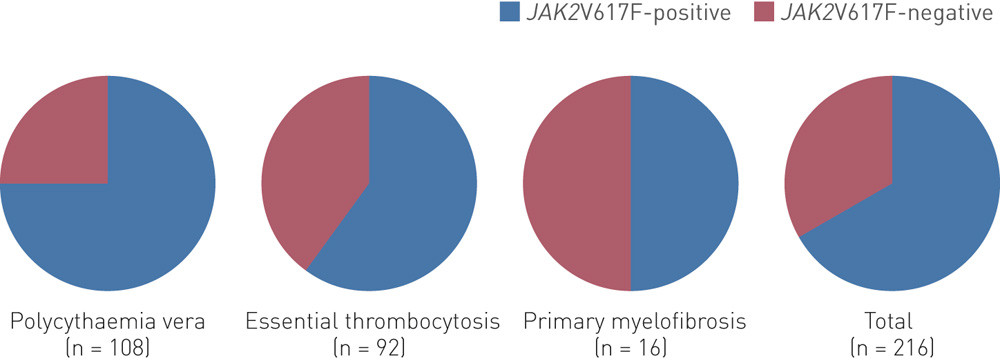
The following diagnoses were registered for the 216 patients diagnosed as having a myeloproliferative disorder: 108 polycythaemia vera (50.0 %), 92 essential thrombocytosis (42.6 %) and 16 myelofibrosis (7.4 %).
The proportion of patients with a JAK2 mutation differed among the various diagnoses (Fig. 3). Eighty-one of the 108 patients with polycythaemia vera (75.0 %), 55 of the 92 with essential thrombocytosis (59.9 %) and eight of the 16 with myelofibrosis (50.0 %) were found to have the mutation. As few patients had been diagnosed with myelofibrosis, further analyses were only conducted on the other patient data.
Biochemical and clinical variables associated with polycythaemia vera
The average thrombocyte count of patients with polycythaemia vera at the time of diagnosis was more than twice as high in mutation-positive as in mutation-negative patients (Table 1). Leukocyte and lactate dehydrogenase levels were also significantly higher in mutation-positive patients. Age at the time of diagnosis was distinctly lower in mutation-negative patients, and they were also more often smokers.
Table 1
Differences in haematological variables and age for JAK2V617F-positive and JAK2V617F-negative patients with polycythaemia vera and essential thrombocytosis. P-values based on t-test (two-tailed). The frequency varies substantially due to incomplete patient data
| Polycythaemia vera, JAK2 V617F-positive |
Polycythaemia vera, JAK2 V617F-negative |
Essential thrombocytosis, JAK2 V617F-positive |
Essential thrombocytosis, JAK2 V617F-negative |
||||||||||
|
|
Average (SD) |
Number |
Average (SD) |
Number |
P-value |
Average (SD) |
Number |
Average (SD) |
Number |
P-value |
|||
| Haemoglobin (g/100 ml) |
17.0 (2.5) |
65 |
17.5 (1.6) |
18 |
0.43 |
14.4 (1.6) |
48 |
13.1 (1.3) |
34 |
< 0.01 |
|||
| EVF |
0.54 (0.08) |
54 |
0.52 (0.05) |
14 |
0.64 |
0.43 (0.06) |
23 |
0.39 (0.04) |
24 |
0.01 |
|||
| Leukocytes (· 109/l) |
12.9 (7.5) |
62 |
8.6 (3.1) |
17 |
0.02 |
11.8 (5.8) |
48 |
8.8 (3.7) |
34 |
< 0.01 |
|||
| Thrombocytes, (· 109/l) |
551 (290) |
63 |
268 (87) |
17 |
< 0.01 |
842 (273) |
50 |
859 (329) |
35 |
0.79 |
|||
| Lactate dehydrogenase (U/l) |
289 (130) |
34 |
198 (48) |
13 |
0.02 |
235 (73) |
26 |
244 (80) |
25 |
0.68 |
|||
| Age (years) |
66.6 (15.0) |
72 |
55.0 (14.8) |
23 |
< 0.01 |
66.0 (13.8) |
51 |
61.3 (19.5) |
35 |
0.23 |
|||
There was a preponderance of mutation-positive women, and a slight majority of mutation-negative men. There was no clear difference between the mutation groups with respect to haemoglobin and erythrocyte volume fraction (EVF). No correlation was found between mutation status and thrombosis prevalence (Table 2).
Table 2
Differences in gender, smoking habits and detected venous and arterial thrombosis between JAK2V617F-positive and JAK2V617F-negative patients with polycythaemia vera and essential thrombocytosis in numbers and percentages. P-values based on chi-squared test and Fisher’s exact test (two-tailed). The frequency (n) varies substantially due to incomplete patient data
| Polycythaemia vera |
Essential thrombocytosis |
||||||||||||
| Variable |
JAK2 V617F-positive |
JAK2 V617F-negative |
P-value |
JAK2 V617F-positive |
JAK2 V617F-negative |
P-value |
|||||||
| Gender |
0.12 |
0.16 |
|||||||||||
| Male |
34 |
(42) |
16 |
(59) |
16 |
(29) |
16 |
(43) |
|||||
| Female |
47 |
(58) |
11 |
(41) |
39 |
(71) |
21 |
(57) |
|||||
| Smoking |
0.01 |
0.31 |
|||||||||||
| Yes |
23 |
(38) |
17 |
(68) |
26 |
(55) |
14 |
(44) |
|||||
| No |
38 |
(62) |
8 |
(32) |
21 |
(44) |
18 |
(56) |
|||||
| Thrombosis |
0.55 |
0.10 |
|||||||||||
| None |
34 |
(42) |
11 |
(41) |
22 |
(41) |
21 |
(57) |
|||||
| Arterial |
33 |
(41) |
11 |
(41) |
27 |
(51) |
13 |
(35) |
|||||
| Venous |
6 |
(7) |
4 |
(15) |
1 |
(2) |
3 |
(8) |
|||||
| Arterial and venous |
8 |
(10) |
1 |
(4) |
3 |
(6) |
0 |
– |
|||||
Biochemical and clinical variables associated with essential thrombocytosis
On average, the haemoglobin level at the time of diagnosis was higher in the mutation-positive than in the mutation-negative group. Significantly higher values were also found for erythrocyte volume fraction and leukocytes in the mutation-positive group (Table 1). There was a higher prevalence of arterial thrombosis in the mutation-positive group, but the finding was not statistically significant (Table 2).
There were no significant differences between the subgroups for the remaining variables.
High allele burden and higher risk of complications
Data on the allele burden of 143 patients were registered. Thirty-nine (27 %) had a high allele burden, while that of the other 104 (73 %) was low.
We found no significant difference between these groups in the incidence of thromboses, but there was a higher incidence of transformation to a higher grade cancer among those with a high allele burden compared with those with a low burden: 13.9 % and 4.0 %, respectively (p = 0.046).
Discussion
In this study we have looked at the prevalence of the JAK2 mutation in a large patient cohort examined for suspected myeloproliferative neoplasm. The mutation was detected in around a fifth of the patients. This does not necessarily mean that none of those with negative results had a myeloproliferative disease. In addition to the classic JAK2 mutation, JAK2V617F, a number of other mutations related to these disorders have been discovered in recent years.
Previous studies have found mutation in exon 12 of the JAK2 gene in about 3 % of patients with polycythaemia vera, while mutations in the gene that codes for the thrombopoietin receptor (MPL) (19) have been found in about 3 % of those with essential thrombocytosis and in 10 % of those with primary myelofibrosis. Nonetheless, the situation before 2013 was that no causal explanation had been found for 30 – 45 % of patients with essential thrombocytosis and primary myelofibrosis. In 2016, two research teams using exon sequencing have found somatic mutations in the CALR gene in 25 – 35 % of these patients (20, 21).
On the basis of these results it is therefore possible today to explain up to 97 % of all cases of myeloproliferative neoplasm with mutations in the JAK2, MPL or CALR genes (20). Analysis of mutations in the MPL and CALR genes is now available at Haukeland University Hospital, but no results have been included in this study.
The possibility that an alternative distinction, between JAK2-mutation-positive and JAK2-mutation-negative myeloproliferative neoplasms, might be more appropriate has been discussed. The reason for this is that mutation-negative status has a distinctly different phenotype. Another idea is that mutation-positive myeloproliferative neoplasms are distributed along a continuum according to allele burden. Tests from animal models have previously shown that the level of expression of the JAK2 mutation has a bearing on the development of a particular disease (22, 23).
As JAK2 mutation analysis has been established as an integral part of the assessment of patients with suspected myeloproliferative neoplasms (11), all those suspected of having these disorders in the Bergen Hospital Trust between the period February 2006 and February 2012 should have undergone this analysis.
It is striking that as many as 25 % of those with polycythaemia vera were mutation-negative, as this was inconsistent with findings in the international literature, where < 10 % was reported (20, 24). It is unlikely that the discrepancy is due to the analytical method, errors in previous papers or an epidemiology peculiar to Norway. A more likely explanation is a certain amount of overdiagnosis of polycythaemia vera, and that patients with other types of polycythaemia were incorrectly given this diagnosis.
Polycythaemia is a collective designation for disorders with elevated haematocrit, and a distinction is made between absolute and relative polycythaemias (25). In absolute polycythaemia, the total erythrocyte volume is pathologically high, as in polycythaemia vera, while in relative polycythaemias the erythrocyte volume is normal, but haematocrit is still elevated. Secondary polycythaemias are absolute polycythaemias that are often due to hypoxia related to underlying heart and lung disease. Relative polycythaemia is associated with smoking, and heavy smoking may be a cause of secondary polycythaemia (25).
The fact that there are almost twice as many smokers among those with JAK2 mutation-negative polycythaemia vera (38 % compared with 68 %) and a lower degree of leuko- or thrombocytosis in the JAK2 mutation-negative group provides support for the overdiagnosis theory. Patients with polycythaemia vera also often have a certain degree of thrombocytosis and leukocytosis, in contrast to those with other types of polycythaemia (24). On the other hand, those who are positive for the JAK2 exon 12 mutation often have isolated erythrocytosis (18, 26).
As further support, the PCR analyses performed in the initial period (2006 – 2008) had a higher detection threshold for the JAK2 mutation (17), which may have led to some patients with a low allele burden not being classified as JAK2 mutation-positive up to 2008. Other limitations implicit in the study are that the diagnosis applied is based on the judgement of the doctor providing treatment and is not tested further.
Previous research found a higher haemoglobin level, lower erythropoietin level and higher leukocytes for JAK2 mutation-positive patients than for CALR-positive patients with essential thrombocytosis (27). MPL and CALR mutations were not investigated in our patient cohort, but we assume that a large proportion of the JAK2 mutation-negative patients with essential thrombocytosis or primary myelofibrosis would test positive for these markers. Our data show significantly elevated levels of haemoglobin, haematocrit and leukocytes in the JAK2 mutation-positive patient cohort. This is consistent with the belief that the JAK2V617F mutation is related to a more aggressive subgroup of essential thrombocytosis than other mutations (28).
Since 2005, the JAK2 mutation has proved to be an important marker in the diagnosis of myeloproliferative neoplasms. The previous WHO diagnostic criteria did not include MPL or CALR mutations (12), but these are now incorporated in the new WHO classification (24, 29) and implemented in the diagnostic criteria in the Norwegian national action programme with guidelines for the diagnosis, treatment and follow-up of malignant blood diseases (30). Monitoring of the JAK2 mutation allele burden may also be indicated after an allogeneic bone marrow graft to treat primary myelofibrosis, and in the future may also play a part by identifying patients with a higher risk of relapse (4, 31).
JAK2V617F analyses together with other mutation analyses – for JAK2 exon 12, MPL and CALR – are central markers in the assessment of patients suspected of having myeloproliferative neoplasms. Our study has shown that there has probably been overdiagnosis of polycythaemia vera, with the result that not all those who have previously received the diagnosis would have met the present criteria. JAK2 mutation-positive myeloproliferative neoplasms have clinical features that distinguish them from mutation-negative, and future risk classification and treatment algorithms may take account of this.
The authors received support for their research from the Western Norway Regional Health Authority, the Norwegian Cancer Society, the Caroline Musæus Aarsvold Fund and the Eivind Møllbach Pedersen Fund.
MAIN POINTS
Myeloproliferative neoplasms are characterised by increased production of one or more myeloid cell lines, often associated with a point mutation in the JAK2 gene on chromosome 9
This mutation was found in 19 % of a cohort of 803 patients examined at Haukeland University Hospital in the period 2006 – 2012
JAK2V617F-positive and JAK2V617F-negative myeloid neoplasms had different clinical and biochemical characteristics
The mutation was found to be less prevalent in polycythaemia vera cases than previously reported, which makes it likely that not all the patients would have met the present day diagnostic criterion for the disease
- 12.
WHO. World Health Organization Classification of Tumors of Haematopoietic and Lymphoid Tissues. 4. utg. Lyon: International Agency for Cancer, 2008.
- 14.
Kiladjian JJ. The spectrum of JAK2-positive myeloproliferative neoplasms. Hematology Am Soc Hematol Educ Program 2012; 2012: 561 – 6.
- 25.
Berentsen S. Sekundære og relative polycytemier: Indikasjon for venesectio? Indremedisineren 2015; 1: 16 – 9.
- 30.
Norwegian Directorate of Health. National guidelines for diagnostic workup and treatment of malignant hematological disorders. http://legeforeningenno/Fagmed/Norsk-selskap-for-hematologi/Handlingsprogrammer/ (12.9.2016).