Oesophageal cancer is a relatively rare form of cancer, but the incidence is increasing in Norway and other Western countries. In 2014, 289 new cases (220 men) were registered in Norway. The incidence varies considerably from country to country, and in Norway (men 9.2, women 2.5 per 100 000) it is approximately double the age-adjusted incidence worldwide (men 4.7, women 1.3 per 100 000) (1).
The two main forms are adenocarcinoma and squamous cell carcinoma. The proportion of the former is increasing and the proportion of the latter is decreasing (2). The disease has a very high mortality, and is the sixth most frequent cause of cancer-related mortality worldwide, and the ninth for men in Norway (1, 3).
Curative treatment for the disease is primarily surgery, and about 20 % of patients who receive the diagnosis undergo surgery (4, 5). Supplementary neoadjuvant chemotherapy or radiochemotherapy increase survival (6, 7). There are complications associated with the surgical procedure, and a not insignificant post-operative mortality, defined as death within 30 days or during the same in-hospital period.
Previous studies have reported complication rates of 27 – 70 % (8) – (14). The complications reported for this type of surgery are pneumonia in 2 – 39 %, anastomotic leakage in 0 – 35 % and recurrent paresis in 0 – 31 % (15). Post-operative mortality also varies from one study to the next, and ranges from 0 – 15 % (15, 16).
Five-year survival after surgery is 80 % if the primary tumour has not infiltrated the submucosa, but falls to 5 – 10 % in the event of lymph node metastases (17). In several datasets from Western countries that include different stages of the cancer, five-year overall survival after resection varies from 21 % to 47 % (18, 19). In Norwegian datasets, five-year survival after curative resection has been between 8 % and 30 % (20) – (23).
Oesophageal resection requires access to both the abdominal and the thoracic cavity, and in the past open surgery has been used. Over the past decade, this intervention has increasingly been performed by means of minimally invasive oesophageal resection, which entails less loss of blood, fewer pulmonary complications and wound infections, shorter stays in hospital and a better quality of life (24) – (26). In the period November 2007 to June 2013, oesophageal resections at Oslo University Hospital, Ullevål, were performed using a hybrid approach, with laparoscopy and open thoracotomy. Since 2013, oesophageal resections have been performed using a minimally invasive approach with both thoracoscopy and laparoscopy.
So far, minimally invasive procedures have not been proven to affect long-term survival (26, 27). Nevertheless, long-term survival following surgery for oesophageal cancer has increased because of the change in oncotherapy regimens, increasing operation volume per surgeon and the possibility of better visualisation, with more radical lymph node dissection associated with minimally invasive techniques (4, 28) – (30).
The purpose of this study was to map post-operative complications, mortality and survival following hybrid oesophageal resection in the period November 2007 to June 2013.
Material and method
Patient dataset
We conducted a retrospective cohort study that included patients who had undergone hybrid oesophageal resection at Oslo University Hospital, Ullevål, from November 2007 to June 2013. The inclusion criteria were malignant neoplasm of oesophagus (ICD-10 C15.3 – 5 and C15.8 – 9) and the gastro-oesophageal junction (C16.0), in cases of planned hybrid oesophagectomy with curative intention.
During this period, eight patients had open surgery and they are therefore not included in the dataset. The reaons for open surgery were lack of laparoscopic expertise on the part of the operator (n = 5), previous laparotomies with adhesions (n = 2) and previous gastrectomy for oesophageal cancer of the gastro-oesophageal junction (n = 1). Exclusion criteria were inoperability due to spreading to surrounding organs and remote metastases.
Diagnosis and stage were established in accordance with national guidelines, with gastroscopy with biopsy and CT scan of thorax and abdomen. MR thorax, whole body PET/CT and endoscopic ultrasound examination with possible cytological examination of lymph nodes were used selectively. Staging was carried out pursuant to the 6th edition of TNM classification from 2002 (31). Stages I, IIa, IIb and III correspond to TIN0M0, T2-3N0M0, T1-2N1M0 and T3N1M0/T4N0-1M0.
The surgical technique used in the thoracic and abdominal cavities is in principle the same for hybrid and open surgery. A general trend, irrespective of approach, is that lymph node dissection has become more radical than previously. In this study of hybrid surgery with laparoscopy and open thoracotomy, oesophageal resection was performed, as well as partial gastric resection and lymphadenectomy of the upper abdomen and mediastinum up to and including the tracheal bifurcation. Lymphadenectomy included lymph nodes perigastrically, at stations 7 and 9, perioesophageally and along both main bronchi and the tracheal bifurcation. A tube-shaped substitute for the oesophagus was made from the residual stomach. During this period, the hybrid procedures were performed by one of two principal surgeons.
A number of the patients received cytostatics and/or radiotherapy pre- and/or post-operatively. Patients with stages IIa, IIb and III, and as a general rule those aged under 75 without severe comorbidity (heart, lung or kidney disease) were assessed for neoadjuvant or perioperative oncotherapy. Patients with microscopically positive resection margins (R1) after resection were assessed for adjuvant therapy.
Ethics and acquisition of information
The study is an internal quality assurance study with legal basis in section 26 of the Health Personnel Act, and publication has been cleared with the data protection officer at Oslo University Hospital.
We used as our starting point an existing prospectively acquired quality assurance database at the Department of Gastrointestinal and Paediatric Surgery at Oslo University Hospital, Ullevål. Demographic data, extent of disease and tumour characteristics, assessment, forms of treatment, per- and post-operative complications, hospital stay, observation time and survival data were recorded there.
In order to be able to classify the complications on the Clavien-Dindo scale, data on treatment of some complications were acquired retrospectively. The most recent update of the survival figures was carried out in December 2015.
Outcomes of the study
The complications examined in this study were recorded prospectively. They were classified according to conventional criteria based on clinical observations and endoscopic, radiological and microbiological findings. The post-operative period was defined as the time from the operation to discharge from Oslo University Hospital, Ullevål.
The Clavien-Dindo classification was used to classify post-operative complications according to their severity (32). It is based on the degree of treatment required by complications, and ranks them in five sub-categories. Grade I is any deviation from the normal post-operative course without a need for treatment. Grade II requires total parenteral nutrition, blood transfusion or medication other than anti-emetics, anti-pyretics and analgesics. Grade III means a need for radiological, endoscopic and surgical intervention without (IIIa) and with general anaesthesia (IIIb). Grade IV means life-threatening complications that require ICU management because of failure of one (IVa) or more organs (IVb). Grade V implies patient death.
Pathological assessment of the resectates formed the basis for determining how radical the surgery had been. Microscopic radicality with a tumour-free resection margin is defined as R0. With R1 there is microscopic, and with R2 macroscopic residual tumour. Examples of R2 are tumour perforation, residual non-resectable tumour and/or metastasis-infiltrated lymph nodes.
Survival was registered by reviewing the patients in the electronic patient records system, which is linked up to the National Population Registry Office. The last registration was made in December 2015.
Statistical analyses
Survival was estimated by means of the Kaplan-Meier method. Log-rank tests were performed to analyse the difference in survival between R0 and R1-2, adeno- and squamous cell carcinoma and stages I–III, and p values below 0.05 were considered significant. Descriptive analyses were used on demographic data. IBM SPSS Statistics version 22 was used for all calculations.
Results
In the course of the study period, 122 patients underwent hybrid oesophageal resection operations with radical intention. Thirteen of them were found peroperatively to be inoperable, and were therefore not included in the study. A total of 109 patients were included in the study.
The median age of all those included was 65 years (range 40 – 79 years), and there was a clear preponderance of men (79 %). The majority of the patients had ASA classification 2 (58.7 %) or 3 (33.0 %). Sixty-five patients (59.6 %) received neoadjuvant and planned perioperative therapy, consisting of neoadjuvant chemoradiotherapy for 33, chemoradiotherapy for ten, radiation for one and perioperative chemotherapy for 21. Five patients (4.6 %) received adjuvant treatment as a result of R1 resection. Age, BMI, smoking details and ASA group are shown in Table 1.
Table 1
Patient characteristics
| Number |
(%) |
|
| Age (years) |
||
| < 60 |
40 |
(36.7) |
| 60 – 70 |
46 |
(42.2) |
| > 70 |
23 |
(21.1) |
| Body mass index (BMI) |
||
| Underweight |
9 |
(8.3) |
| Normal weight |
53 |
(48.6) |
| Overweight |
33 |
(30.3) |
| Obese |
14 |
(12.8) |
| Smoking |
||
| Smoker |
41 |
(37.6) |
| Ex-smoker |
15 |
(13.8) |
| Never smoked |
49 |
(45.0) |
| Unknown |
4 |
(3.7) |
| ASA Class |
||
| 1 |
8 |
(7.3) |
| 2 |
64 |
(58.7) |
| 3 |
36 |
(33.0) |
| 4 |
1 |
(0.9) |
For five patients (4.6 %), laparoscopy was converted to laparotomy because of prolonged procedures (n = 2), perforation of the small intestine (n = 1), haemorrhaging (n = 1) or tight anatomical conditions at the oesophageal hiatus (n = 1). The median operation time was 390 minutes (range 225 – 660 minutes). Twenty-seven patients (24.8 %) lost more than 500 ml of blood. One patient required post-operative transfusion. The median hospital stay after the operation was 16 days (range 9 – 88 days). On discharge, 21 patients (19.2 %) were transferred to local hospitals for further follow-up. The remainder were discharged to home.
Tumour-free resection margins (R0) were achieved in 100 of the patients (91.7 %), 60 patients (55.0 %) had infiltration of one or more lymph nodes. Adenocarcinoma was found in 87 patients (79.8 %), and squamous cell carcinoma in 22 (20.2 %). Tumours were located above and by the carina in three (2.8 %), in the distal oesophagus in 41 (37.6 %) and at the gastro-oesophageal junction in 65 (59.6 %) of the patients. The TNM stage was I in 13 of the patients (11.9 %), IIa in 34 (31.2 %), IIb in 11 (10.1 %) and III in 51 (46.8 %).
Post-operative morbidity and mortality
A total of 118 complications were recorded (Table 2) affecting 70 of the patients (64.2 %). Thirty-one (28.4 %) had more than one complication (median 2, range 2 – 6). The most common were pneumonia (33.0 %) and arrhythmia (12.8 %). Pleural fluid was drained from 12.8 %. Most patients (Table 3) had grade II (29.4 %) as the most serious complication, followed by grade IIIa (19.3 %). Five patients (4.6 %) developed anastomotic leakage, treated with drain (n = 1), stent (n = 3) or stent and clip (n = 1). Five underwent reoperation – for chylothorax (n = 2), haemorrhaging (n = 2) and air leakage from the right lower lobe (lobectomy) (n = 1).
Table 2
Number of complications (n = 118) in 70 patients (64.2 %) classified according to the Clavien-Dindo classification
| Complication |
Number |
| Grade I (n = 8) |
|
| Pneumothorax (no treatment) |
1 |
| Recurrent paresis (speech therapist) |
2 |
| Wound infection (no treatment/flushed) |
4 |
| Muscular necrosis (not treated) |
1 |
| Grade II (n = 68) |
|
| Pneumonia (antibiotics) |
27 |
| Arrhythmia (antiarrhythmics) |
13 |
| Urine retention (clean intermittent catheterisation, (CIC) |
2 |
| Wound infection (antibiotics, drained if necessary) |
4 |
| Post-operative delirium (psychotropic drugs) |
6 |
| Deep vein thrombosis (low-molecular heparin) |
2 |
| Urinary tract infection (antibiotics) |
8 |
| Oral candidiasis (antimycotics) |
1 |
| Heart failure (diuretics) |
2 |
| Haemorrhaging (blood products, fluid) |
1 |
| Gastric retention (total parenteral nutrition, TPN) |
2 |
| Grade IIIa (n = 26) |
|
| Empyema (drained, antibiotics) |
3 |
| Pleural fluid (drained) |
14 |
| Anastomotic leakage (drain/stent) |
5 |
| Pericardial fluid (drain) |
1 |
| Failure external sutures (replaced, sutured) |
1 |
| Anastomosis ulcer (stented) |
1 |
| Arrhythmia (implantable cardiac defibrillator, ICD) |
1 |
| Grade IIIb (n = 4) |
|
| Chylothorax (re-operation) |
2 |
| Haemorrhaging (re-operation) |
2 |
| Grade IVa (n = 10) |
|
| Respiratory failure (respirator) |
10 |
| Grade IVb (n = 2) |
|
| Multi-organ failure (≥ 2 organ systems) |
2 |
Table 3
Distribution of degree of complication using Clavien-Dindo classification for patients (n = 109)
| Highest degree of complication |
Number |
(%) |
| I |
2 |
(1.8) |
| II |
32 |
(29.4) |
| IIIa |
21 |
(19.3) |
| IIIb |
3 |
(2.8) |
| IVa |
10 |
(9.2) |
| IVb |
2 |
(1.8) |
| V |
0 |
(0) |
None died prior to discharge from either Oslo University Hospital or a local hospital.
Survival
The median follow-up time was 51 months, and 26 patients (23.9 %) were monitored for over five years. Forty-nine of the patients (45.0 %) died in the course of the follow-up period, and the cause of 40 of the deaths (81.6 %) was cancer-related. The median survival for all patients (R0-2) was 55 months. Estimated five-year survival was 48 % (95 % CI 36 – 60 %) (Fig. 1). Estimated five-year survival for adenocarcinoma cases was 45 % (95 % CI 31 – 59 %) and for squamous cell carcinoma 65 % (95 % CI 42 – 87 %). Estimated five-year survival for the group with R0 resection (n = 100) was 51 % (95 % CI 38 – 63 %), with median survival of 55 months. Estimated median survival for R1-2 resection (n = 9) was ten months (95 % CI 0 – 28 months).
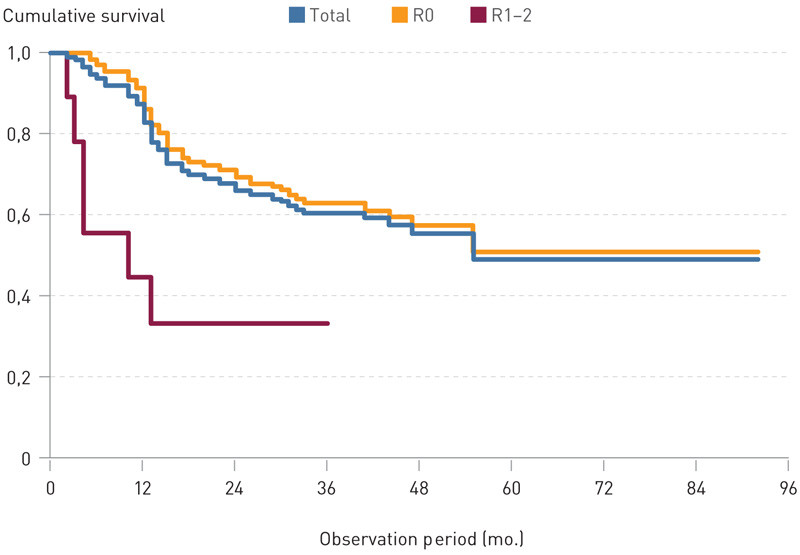
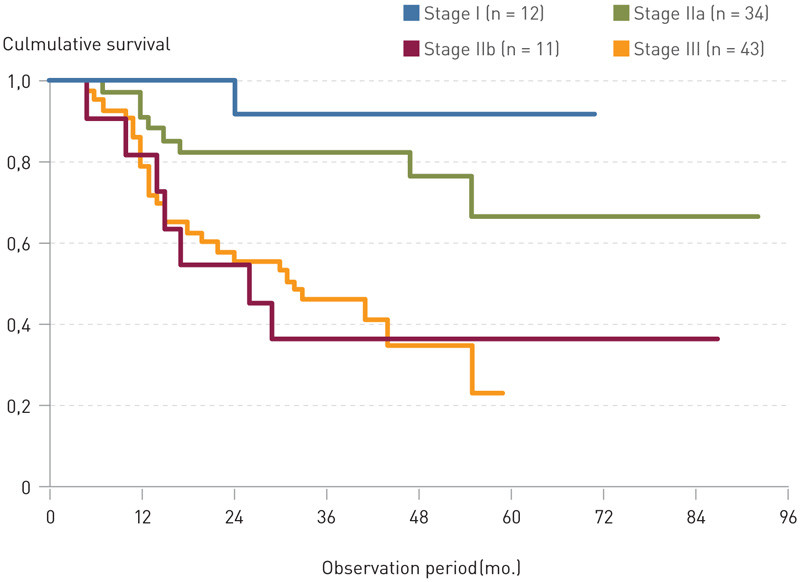
There was a significant difference in survival between the groups R0 and R1-2 (p value 0.005). The patients in the R1-2 group made up a small percentage of the total dataset, so that the results from this group had little effect on survival in the dataset as a whole. In the R0 group there was a significant difference in survival (p value < 0.001) between stages I, IIa, IIb and III (Fig. 2). Cancer-specific survival for the R0 group was 59 % (95 % CI 47 – 71 %).
Discussion
Comparing the incidence of post-operative complications and mortality after oesophageal resection in different studies is challenging because standardised complication definitions and grades are not used (15, 16).
In three studies from France (33), Scandinavia (34) and the Netherlands (14), percentage morbidity according to the Clavien-Dindo classification was found to be 46.5 %, 48.1 % and 70.3 %, respectively, as compared to 64.2 % in the present study. Hospital mortality figures were 4.3 %, 3.9 % and 3.6 %, respectively, compared with 0 %. The patient populations in these three studies resemble the Ullevål dataset in respect of age, gender and BMI distribution, but the operations were largely performed by means of laparotomy and thoracotomy.
Pulmonary complications in particular, but also the total number of complications, are reported to have been reduced through the use of hybrid resection compared with open oesophageal resection (35). In the present study of hybrid surgery, the proportion of minor complications (grades I–IIIa) was higher than in previous studies (14, 33, 34). However, the incidence of more serious complications (grades IIIb-V) was lower. This is attributable to few anastomotic leakages and reoperations and no hospital mortality in our patient dataset.
In this study, estimated five-year survival for resection (R0-2) was estimated at 48 %. This is higher than previous Norwegian figures (8 – 30 %) and on a par with the results for patients who received neoadjuvant radiochemotherapy in a Dutch multi-centre study (47 %) (19) – (23). Survival was also higher than in an English study (27 %) and a Swedish study (21 %) (30, 10). The proportion who received neoadjuvant therapy in these two studies was lower than the proportion in the Ullevål dataset (17 % and 5 % compared with 60 %). There was also a larger proportion with TNM stage IV (13 % and 11 % compared with 0 %) and a lower proportion with tumour-free resection margins (not specified in the English study, 84 % in the Swedish, 92 % in this study). In the Dutch study, however, the distributions of the TNM stage and tumour-free resection margins were comparable to those in the Ullevål study.
These results indicate that a high degree of neoadjuvant treatment, a high proportion of R0 resections and a low stage result in higher overall survival in patients who undergo resection for oesophageal cancer. These factors, in conjunction with the high operation volume per surgeon and more radical lymph node dissection than previously, may have contributed to the high survival in this study.
Survival figures in this study are taken from patient records. The data might have been even more reliable if the information had been obtained from the Cause of Death Register. Another weakness of the study was that in order to be able to use the Clavien-Dindo classification, data on treatment of some complications had to be retrieved retrospectively by going through patient records. There is also a possibility that minor complications that arose after discharge to the local hospital are not recorded.
The dataset in this study is too small to permit evaluation of the survival effect of the oncotherapy, because of the different therapy regimens and therefore limited number of patients for each type of such therapy. On the other hand/However, this study, with a relatively large dataset, is useful as a basis for comparison of survival after resection for oesophageal cancer using open, hybrid or a total minimally invasive approach (thoracoscopy and laparoscopy).
The prognosis after resection for oesophageal cancer in Norway has improved in the last decade (20) – (22). Increasing use of total minimally invasive surgery results in less severe complications, a shorter hospital stay and a better quality of life than open surgery, but it has not been documented whether survival increases (24) – (26). Total minimal invasive surgery also reduces the number of complications more than hybrid access. The proportion of total thoracolaparoscopy resection is an estimated 15 – 30 % worldwide, and this has been our standard method since June 2013 (24).
MAIN POINTS
Outcomes for hybrid oesophageal section were no post-operative mortality, a low proportion of serious complications and good five-year survival
There is a growing tendency for minimally invasive procedures in the surgical treatment of oesophageal cancer
- 1.
Cancer Registry of Norway. Cancer in Norway 2014 – Cancer incidence, mortality, survival and prevalence in Norway. Oslo: Cancer Registry, 2015.
- 2.
Demeester SR. Epidemiology and biology of esophageal cancer. Gastrointest Cancer Res 2009; 3 (suppl): S2 – 5. [PubMed]
- 3.
Ferlay J, Soerjomataram I, Ervik M et al. GLOBOCAN 2012 v1.0. Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. Lyon: International Agency for Reasearch on Cancer, 2013. http://globocan.iarc.fr (3.2.2015).
- 5.
Viste A. Kirurgi ved spiserørskreft. Kirurgen 2011; nr. 1: 10 – 2.
- 20.
Mala T, Løtveit T. Kirurgisk behandling av kreft i spiserøret. Tidsskr Nor Lægeforen 2001; 121: 2815 – 7. [PubMed]
- 22.
Goscinski MA, Larsen SG, Warloe T et al. Adenocarcinomas on the rise–does it influence survival from oesophageal cancer? Scand J Surg 2009; 98: 214 – 20. [PubMed]
- 23.
Johnson E, Gjerlaug BE, Holck-Steen A et al. Kirurgi og stenting for cancer oesophagi. Tidsskr Nor Lægeforen 2005; 125: 286 – 8. [PubMed]
- 24.
D'Journo XB, Thomas PA. Current management of esophageal cancer. J Thorac Dis 2014; 6 (suppl 2): S253 – 64. [PubMed]
- 27.
Petropoulos K, Macheras A, Liakakos T et al; -. Minimally invasive esophagectomy for esophageal cancer: techniques and outcomes. Chirurgia (Bucur) 2015; 110: 99 – 108. [PubMed]
- 31.
Sobin LH, Wittekind CH. International Union Against Cancer (UICC): TNM classification of malignant tumors. 6. utg. New York: Wiley, 2002.