In 2014, there were approximately 220 000 persons with known diabetes mellitus in Norway (1). Of these, 80 – 90 % had type 2 diabetes. The vast majority are followed up in the primary health service (2). From 1988 to 2004, the Norwegian College of General Practice published a treatment programme for diabetes in general practice (3). In 2009, this work was continued in the form of national professional guidelines from the Directorate of Health on prevention, diagnosis and treatment of the disease (4). The guidelines emphasise the importance of a change in diet, smoking cessation, and physical activity, as well as monitoring blood sugar level, blood pressure, blood lipids and weight, and regular check-ups are recommended every 2 – 6 months (4).
Use of paper-based or electronic forms for systematic recording of clinical data may contribute to improved follow-up of patients with chronic illnesses (5). Forms used in the follow-up of diabetes may include everything from simple paper forms to more advanced solutions whereby the data are processed and feedback provided that is tailored to individual patients, for example suggestions for changes in treatment or follow-up.
Some forms are used to collect data for central registries, and can provide a basis for automated feedback on whether patients belonging to one GP or one GP surgery are achieving the therapeutic objectives compared to the national or regional average.
An electronic form has been developed for use in the GP surgery for follow-up of patients with diabetes (the NOKLUS diabetes form). This is intended to be both a clinical tool for the GP and a data-collection tool for the Norwegian Diabetes Registry (6).
Several systematic overviews are available on the effects of various measures with regard to diabetes follow-up, but none deals specifically with the use of forms for recording clinical data (7). Following a request from the Norwegian College of General Practice, we have conducted a systematic review of the possible health benefits of using forms for the follow-up of diabetes in general practice.
Material and method
We performed systematic searches for studies in the MEDLINE (Ovid), EMBASE (Ovid), ISI Web of Science, Cochrane CENTRAL and PubMed databases, from initiation of the databases until June 2015. In our search we combined text words and key words that describe the population and the intervention, for example (diabet*) AND (structured care OR benchmarking OR decision support systems OR structured assessment OR standardized patient record).
The search was further limited using search terms for relevant study designs, parts of which were limited using search terms for the primary health service (for example, primary care OR community care) (Larun appendix 1). We also searched for relevant studies by reviewing reference lists in included articles.
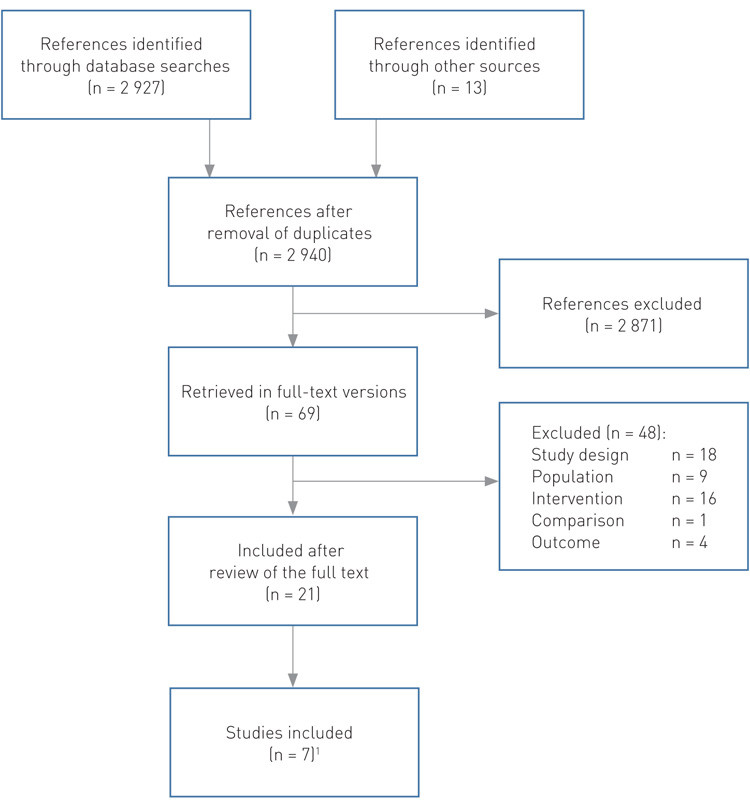
The search resulted in 2 940 titles and abstracts, which we evaluated against pre-defined selection criteria (Box 1). We included a total of seven randomised, controlled studies (Figure 1). One of the authors (LL) extracted data from the studies, while another (KGB) checked that the data retrieved were correct. The same two authors (LL and KGB) reviewed all the studies included and assessed the risk of bias in relation to patient selection, performance, detection, attrition and reporting (8). Randomisation procedure, blinding of participants and personnel, and analyses of attrition are important in this assessment.
Criteria for selection of studies
Study design
Randomised, controlled studies
Patient
Adults > 18 years with diabetes followed up by the primary health service
Intervention
Use of a diabetes form, with or without individual feedback
Control
Follow-up without use of a diabetes form, e.g. normal use of electronic patient records
Endpoint
Primary: Mortality, coronary disease, stroke and complications such as renal failure, peripheral neuropathy or retinopathy
Secondary: Long-term blood sugar (HbA1c), blood pressure, weight, blood lipids or microalbuminuria
Exclusion: Studies that report only goal achievement, i.e. the proportion being followed up in accordance with prevailing guidelines
Follow-up period
More than 12 months after the first completion of the for
When cluster-level randomisation is undertaken, for example when a GP surgery is randomised rather than individual patients, this must be accounted for in the calculation of confidence intervals and p-values. Our analyses are therefore based on results with adjustment for cluster effects. Where possible, we have used effect estimates that are also adjusted for potential differences between the groups at the start of the study.
We report dichotomous endpoints such as the hazard ratio (HR), which is a time-to-event outcome, or odds ratio (OR), which is an outcome representing the odds of an event at a defined point in time. We report continuous endpoints as mean differences (MD). Meta-analyses were performed with Review Manager Software (RevMan) (9) using a random effect model that takes account of possible variation in the true effect between single studies. The main emphasis was placed on hard endpoints, such as risk of mortality, coronary disease and stroke. We collated the results for primary and secondary endpoints (Box 1).
The quality of the evidence was assessed using the GRADE scale (Grading of Recommendations Assessment, Development and Evaluation) (10). This article is co-published with a report from the Norwegian Knowledge Centre for the Health Services/Norwegian Institute of Public Health (11). The project plan for this work was published in November 2014 (12).
Results
We included seven randomised controlled studies published between 2001 and 2011 (Table 1 (13) – (19). Three studies were conducted in the USA (13) – (15), one in the Netherlands (16), one in the UK (17), one in Canada (18) and one in Denmark (19). The number of patients varied from 335 to 7 412, and the average age was approximately 60 years. Good randomisation procedures with concealed allocation of the participants are described in three of the studies, and we consider that these studies have a low risk of bias (13, 18, 19). The remaining studies were considered to have a high risk of bias, primarily because the randomisation and blinding procedures were not clearly described (8).
Table 1
Description of the included randomised, controlled studies (n = 7)
| First author, year, country, (reference) | Number of participants | Duration of intervention | Was the form integrated in the electronic patient records? | What patient data were recorded? | What report did the responsible doctor receive? |
| Bebb, 2007, UK (17) | 1 534 | 1 year | Unclear | Blood pressure | Not reported |
| Cleveringa, 2008, the Netherlands (16) | 3 391 | 1 year | Yes | HbA1c, blood pressure, blood lipids, weight, smoking | Automatic call-in for check-ups Report on whether the patient is achieving therapeutic objectives |
| Holbrook, 2009, Canada (18) | 511 | 6 months | Yes | HbA1c, blood pressure, blood lipids, albuminuria, weight, smoking | Monthly automatic telephone reminder to the patient on medication, booking an appointment and blood tests |
| MacLean, 2004, USA (13) | 7 412 | 3 years | Yes | HbA1c, blood pressure, blood lipids, weight | Reminder if laboratory tests exceed the threshold and no follow-up appointment booked The doctor receives a report on status of his/her patients relative to the study population |
| O’Connor, 2009, USA (14) | 3 703¹ | 3 years | Yes² | HbA1c, blood pressure, blood lipids | Data relevant to diabetes treatment sent every 4 months Proposes change in medication, frequency of testing and consultation The doctor receives a report on status of his/her patients relative to the study population |
| O’Connor, 2011, USA (15) | 2 556 | 8 months | Yes² | HbA1c, blood pressure, blood lipids | Completed form at top of patient records sent to doctor Proposes change in medication, frequency of testing and consultation |
| Olivarius, 2001, Denmark (19) | 1 263 | 6 years | No | HbA1c, blood pressure, blood lipids, microalbuminuria, weight | Questionnaire sent one month before consultation Yearly reporting on risk factors, medication, complications and therapeutic objectives for individual patients Encouraged to set target figures |
| [i] | |||||
[i] ¹ Distributed over one control group and three different intervention arms
² Indirect: need to follow a link within records to open additional module
The doctors had used a diabetes form in all the studies, and in collaboration with nurses in two of them (16, 17). Time spent on training was around one hour where this was reported. The form was integrated in the patient records in the majority of the studies (Table 1). The opportunities for processing of recorded patient data varied, and could be functions of automated reminders to the patient (18), reminders of appointment call-in (13, 16, 18), suggestions for changes in medication (14, 18, 20) and feedback on recorded data for the patient or the GP practice compared with others (13, 14, 19).
Primary endpoints
Only one study (19) reported results for our primary endpoints (Box 1). This Danish study included 1 263 participants. The results showed no obvious effect on mortality resulting from the use of forms (HR 0.91; 95 % CI 0.72 – 1.14). For the other endpoints, the estimated effect varied from an odds ratio of 0.65 (95 % CI 0.31 – 1.35) for non-fatal myocardial infarction to an odds ratio of 0.90 (95 % KI 0.53 – 1.52) for retinopathy.
For all the endpoints studied, there were positive tendencies in favour of the use of forms, but the effect size estimates were too imprecise to enable any definite conclusions. We considered the quality of the documentation for these endpoints to be generally low, because it was based on only one study, and due to wide confidence intervals for the effect estimates (Table 2). Results for renal failure were not reported in any of the studies.
Table 2
GRADE table for mortality, cardiovascular disease, retinopathy and peripheral neuropathy
| Calculation of absolute effect (95 % CI) | ||||||
| Endpoint | Risk with normal follow-up | Risk with structured follow-up | Relative effect (HR¹/OR²) (95 % CI) | Number of participants (number of studies) | Quality of documentation (GRADE) | |
| Mortality, per 1 000 | 339 | 314 (258 – 377) | HR 0.91 (0.72 – 1.14) | 1 262 (1) | ⊕⊕◯◯ Low3, 4 | |
| Angina pectoris, per 1 000 | 67 | 61 (34 – 107) | OR 0.90 (0.49 – 1.66) | 714 (1) | ⊕⊕◯◯ Low3, 4 | |
| Myocardial infarction (non-fatal), per 1 000 | 46 | 30 (15 – 61) | OR 0.65 (0.31 – 1.35) | 830 (1) | ⊕⊕◯◯ Low3, 4 | |
| Stroke (non-fatal), per 1 000 | 40 | 35 (16 – 76) | OR 0.89 (0.39 – 2.01) | 851 (1) | ⊕⊕◯◯ Low3, 4 | |
| Newly discovered peripheral neuropathy, per 1 000 | 210 | 186 (131 – 255) | OR 0.86 (0.57 – 1.29) | 704 (1) | ⊕⊕◯◯ Low3, 4 | |
| Newly discovered retinopathy, per 1 000 | 136 | 124 (77 – 194) | OR 0.90 (0.53 – 1.52) | 679 (1) | ⊕⊕◯◯ Low3, 4 | |
| [i] | ||||||
[i] ¹ HR: hazard ratio
² OR: odds ratio
³ Based on one study with limited number of events
⁴ Very imprecise estimate (wide confidence interval)
Secondary endpoints
In all the studies that were included, the effect of using the form was reported for at least one secondary endpoint (Box 1), and it was possible to perform meta-analyses for several of these (Larun appendix 2). Use of the form had little or no effect on body weight. However, we saw some effect on total cholesterol level and blood pressure (Figure 2). For example, we found an average change in systolic blood pressure of –2.99 mm Hg (95 % CI – 4.38 to – 2.59), based on the results from five studies (Figure 2).
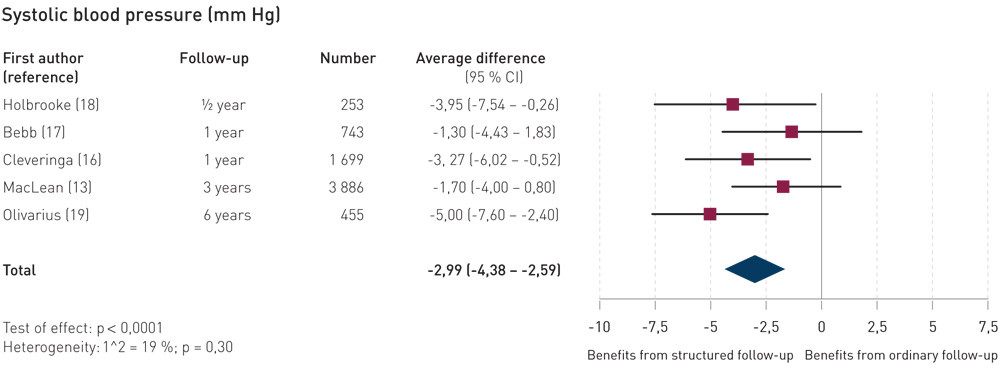
For long-term blood sugar level and urine microalbumin level, the results were more uncertain. The quality of the documentation varied between high and low for different endpoints. Low confidence in the effect estimates is largely attributable to inconsistent and/or imprecise results (wide confidence intervals) (Table 3).
Table 3
GRADE table for HbA1c, blood pressure, weight, blood lipids and microalbuminuria
| Calculated absolute effect (95 % CI) | ||||
| Endpoint | Average with normal follow-up | Average effect of structured follow-up | Number of participants (number of studies) | Quality of the documentation (GRADE) |
| HbA1c [%] | 6.9 – 7.3 | –0.05 (–0.20; 0.09) | 11 314 (3)¹ | ⊕⊕◯◯ Low2, 3 |
| Systolic blood pressure [mm Hg] | 135 – 147 | –2.99 (–4.38; –1.59) | 13 598 (5) | ⊕⊕⊕◯ Medium² |
| Diastolic blood pressure [mm Hg] | 75 – 82 | –0.98 (–1.97; 0.02) | 13 598 (5) | ⊕⊕⊕◯ Medium² |
| Body mass index [kg/m²] | 31.9 – 33.7 | –0.09 (–0.47; 0.29) | 7 923 (2)⁴ | ⊕⊕⊕⊕ High |
| Total cholesterol [mmol/l] | 4.8 – 4.9 | –0.18 (–0.26; –0.10) | 4 248 (2) | ⊕⊕⊕◯ Medium² |
| HDL cholesterol [mmol/l] | 1.33 | 0.01 (–0.01; 0.04) | 3 391 (1) | ⊕⊕◯◯ Low2, 5 |
| LDL cholesterol [mmol/l] | 2.4 – 2.6 | –0.05 (–0.16; 0.06) | 11 314 (3) | ⊕⊕◯◯ Low2, 3 |
| Albumin/creatinine ratio [mg/mmol] | 5.95 | 0.65 (–1.11; 2.41) | 272 (1)⁶ | ⊕⊕◯◯ Low⁵ |
| [i] | ||||
[i] ¹ The study by Olivarius and colleagues (n = 858) also shows a statistically significant reduction in HbA1c of approx. 0.9 per cent (95 % CI 0.83 – 0.93) (19)
² Risk of bias due to methodological limitations in one or more studies
³ Inconsistent results across available studies
⁴ The study by Olivarius and colleagues (n = 852) also shows little or no change in weight: –0.83 kg (–1.75; 0.09)
⁵ Very imprecise estimates
⁶ The study by Olivarius and colleagues (n = 483) also shows a reduction in the number of participants with urine albumin ≥ 15 mg/l with use of the form (OR 0.63; 95 % CI 0.41 – 0.98) with no change in serum creatinine measured.
Discussion
In this systematic review of the research literature we identified only one study on the association between the use of a diabetes form in which effects on hard endpoints such as mortality and coronary disease had been registered (19). The results gave positive indications, but as yet the documentation is insufficient to confirm or disprove a possible effect.
We identified another six studies on the efect of diabetes forms on secondary endpoints such as blood pressure and cholesterol levels (13) – (18). We found that the use of a form most likely leads to lower levels in patients, but the size of this effect is minor. The use of a form is unlikely to have an effect on the patients’ body weight, and the effect on long-term blood sugar levels and urine microalbumin level is uncertain. The fact that only few studies were found is interesting in itself, highlighting the fact that we have no data to draw any specific conclusions as to whether the use of structured forms is a cost-effective measure. The NOKLUS form also belongs in this category (6).
On the whole, measures to improve the quality of clinical practice have a small to moderate effect across interventions and clinical areas (21). Systematic reviews that are not restricted to diabetes patients have shown that computer-based systems for clinical decision support may help bring about changes in practice among health personnel and a possible health gain for patients (22, 23).
Tricco and collaborators summarised findings from 142 randomised studies of various measures to improve the follow-up of persons with diabetes, but their report does not refer to the effect of diabetes forms specifically (24). They found clear effects on process measures such as prescribing acetylsalicylic acid and anti-hypertension drugs, but the results were less convincing with regard to measures of clinical outcomes, such as blood pressure or cholesterol levels. They reported no hard clinical endpoints. In light of the results from other systematic reviews, our finding of uncertain and small effects from using various types of diabetes forms in clinical practice was approximately as expected (24).
The objective of the study was to investigate whether the use of forms in diabetes follow-up reduces the risk of serious complications in patients. We chose to summarise studies that had investigated this association directly, but this should not be taken to mean that our summary provides a complete overview of all studies that are relevant for assessing the advantages and disadvantages of using forms. Despite our comprehensive searches in five central medical databases we cannot exclude the possibility that some relevant studies have escaped our attention.
We should also acknowledge that among the studies that failed to fulfil our inclusion criteria, some may nevertheless be relevant. For example, we chose to exclude studies that reported whether the use of forms promoted compliance with treatment guidelines. Nor did we search for studies that used a qualitative design, which may have provided an answer to how the use of forms affects the consultations or how it is perceived by patients and general practitioners.
In addition to limitations in their research questions and methodologies, the included studies also have certain limitations. There was considerable variation in the ways in which the interventions were implemented, how the forms were designed, the kinds of feedback that were given and whether the form was integrated in the electronic patient records or not. The way in which the control group was followed up varied as well. Ideally, we should have undertaken sensitivity and sub-group analyses to explore differences between the different variables, but the available data material is too small to make such analyses meaningful. A further challenge was that in most studies the follow-up period was restricted to one year. We would have liked to see more studies with longer follow-up periods.
The studies in our systematic review had been conducted in Western countries, with patients and healthcare systems that can be regarded as similar to Norway’s – perhaps with the USA as an exception. As described above, the use of differing types of forms and differing types of feedback represents a challenge. This notwithstanding, our general assessment is that the available research literature provides knowledge that elucidates possible effects of a more frequent use of diabetes forms in Norwegian general practice.
An assessment of the benefits of a diabetes form must include a number of concerns. The diabetes form from NOKLUS is intended as a clinical tool for the general practitioner as well as a data collection tool for the Norwegian Diabetes Registry. There are obvious advantages to be gained from having a national registry as a database for research that may benefit patients.
With regard to the use of structured forms in consultations with patients, there is the added concern that the consultation has an allotted time and that questions from the patients require attention. Being able to discuss emotional and practical issues involved in life with a chronic disease is important. Counselling in questions of lifestyle is time-consuming, but necessary. The time that the doctor spends on completing forms may come into conflict with the total time available for direct contact with the patient.
The use of the reimbursement rate for annual check-ups of diabetes may elucidate how extensively the NOKLUS diabetes form is used in general practice. According to HELFO, use of the rate has increased from a total of 12 500 in 2012 to 17 800 in 2014. This means that on average, each general practitioner uses this rate approximately three times per year (T. Nydal, HELFO, personal communication), corresponding to annual check-ups of only 10 % of all patients with type 2 diabetes.
Although the absolute values for effects on individual measurements are small and hardly of any major clinical importance, these small effects may nevertheless help identify patients who are presumed to have poorly controlled diabetes. These patients are known to have elevated morbidity and mortality. Reducing the proportion of patients with a high total risk is an independent objective in practice. Identification of poorly controlled patients is also important when doctors are to use data to compare their own practice with those of their colleagues.
The Directorate of Health has explicitly recommended more frequent use of the NOKLUS diabetes form (25). Our systematic review shows that the clinical effect of using the form is uncertain. An improvement in the follow-up of diabetes patients has been registered for a number of years, including before the NOKLUS diabetes form was introduced (2). It is questionable whether the form complies with the criteria for an appropriate and cost-effective measure, and it remains to be seen whether an increased use of structured forms is the proper medicine to achieve further improvements.
One should consider assessing the benefits of the NOKLUS diabetes form and measure its effects on mortality and morbidity, how the use of the form is perceived by general practitioners and patients, and whether processing of the collected registry data may help improve the quality of the follow-up that general practitioners provide to this patient group.
We wish to thank Ola Morten Rygh and Ingvild Kirkehei, who helped us in the data search and collection and with quality assurance. We also wish to thank Gro Jamtvedt, Liv Merete Reinar and peer reviewers for their input. The working hours spent on producing this article were funded by the Norwegian Institute of Public Health.
Appendices at tidsskriftet.no/larun1engappendiks og tidsskriftet.no/larun2engappendiks
MAIN MESSAGE
No documented evidence is available to indicate that the use of forms for follow-up of diabetes reduces the risk of mortality, myocardial infarction, stroke, peripheral nerve injury or retinopathy
Use of forms has little, or very limited effect on HbA1c levels, blood pressure, weight, blood lipids and microalbuminuria
- 1.
Folkehelseinstituttet. Forekomst av diabetes: fakta og helsestatistikk om diabetes. Oslo: Folkehelseinstituttet, 2014. www.fhi.no/eway/default.aspx?Main_6157=6263:0:25,5862&MainContent_6263=6464:0:25,5863&List_6212=6218:0:25,5872:1:0:0:0:0 (1.6.2014).
- 2.
Claudi T, Ingskog W, Cooper JG et al. Kvaliteten på diabetesbehandlingen i allmennpraksis. Tidsskr Nor Legeforen 2008; 128: 2570 – 4. [PubMed]
- 3.
Claudi T, Cooper J, Midtjell K et al. NSAMs handlingsprogram for diabetes i allmennpraksis. Oslo: Norsk selskap for allmennmedisin, Norges Diabetesforbund, Statens helsetilsyn, Statens institutt for folkehelse, Den norske legeforening, 2005.
- 4.
Claudi T, Abrahamsen R, Andersen S et al. Diabetes: Forebygging, diagnostikk og behandling. Oslo: Helsedirektoratet, 2009.
- 5.
Krüger K. Elektroniske pasientjournaler bør være strukturerte. Tidsskr Nor Lægeforen 2007; 127: 2090 – 3. [PubMed]
- 8.
Higgins JPT, Altman DG, Sterne JAC. Chapter 8: Assessing risk of bias in included studies. I: Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0. København: The Cochrane Collaboration, 2011. http://handbook.cochrane.org/chapter_8/8_assessing_risk_of_bias_in_included_studies.htm (30.10.2015).
- 9.
Cochrane. Review Manager software (RevMan). RevMan 5.2. København: Cochrane Informatics & Knowledge Management Department, 2012.
- 10.
Brozek J, Oxman A, Schünemann H. GRADEpro. GDT: The Grading of Recommendations Assessment, Development and Evaluation (short GRADE) Working Group 2008. http://gradepro.org/ (30.10.2015).
- 11.
Larun L, Kirkehei I, Rygh OM et al. Bruk av strukturerte skjema i diabeteskontrollen i primærhelsetjenesten. Rapport. Oslo: Kunnskapssenteret i Folkehelsetjenesten, 2016.
- 12.
Larun L, Brurberg KG, Kirkehei I et al. Bruk av strukturerte skjema i diabeteskontrollen i primærhelsetjenesten – prosjektplan. Oslo: Nasjonalt kunnskapssenter for helsetjenesten, 2014. www.kunnskapssenteret.no/prosjekter/prosjektplan-for-bruk-av-strukturerte-skjema-i-diabeteskontrollen-i-primaerhelsetjenesten (30.10.2015).
- 17.
Bebb C, Kendrick D, Coupland C et al. A cluster randomised controlled trial of the effect of a treatment algorithm for hypertension in patients with type 2 diabetes. Br J Gen Pract 2007; 57: 136 – 43. [PubMed]
- 21.
Fretheim A, Flottorp S, Oxman AD. Effekt av tiltak for implementering av kliniske retningslinjer. Rapport. Oslo: Nasjonalt kunnskapssenter for helsetjenesten, 2015.
- 25.
Helsedirektoratet. Gjennomføre årskontroller og rutinekontroller ved diabetes 2015. https://helsedirektoratet.no/diabetes/gjennomfore-arskontroller-og-rutinekontroller-ved-diabetes (9.1.2015).
Etter en henvendelse fra Norsk forening for allmennmedisin har Kunnskapssenteret i Tidsskriftet nr.5/2016 publisert en systematisk oversikt av effekten av bruk av skjema i oppfølging av diabetes i allmennpraksis (1). Vi er enige i forfatternes fortolkning av studien; det er tendens til at bruk av diabetesskjema kan bidra til lavere dødelighet og sykelighet. Den vitenskapelige evidensen for dette er lav. Seks av de syv inkluderte studiene var av så kort varighet at man ikke kan forvente noen effekt på hjerte- og karsykdom eller dødelighet. Den lengste studien viste imidlertid tendens til en gunstig effekt. Ingen av studiene undersøkte effekt på prosessindikatorer eller andel som var dårlig regulert, og bare fire ga tilbakemelding til legene. Forfatterne refererer til at andre systematiske undersøkelser har vist at databaserte systemer for klinisk beslutningsstøtte kan medføre praksisendring og helsegevinst. På populasjonsnivå er en gjennomsnittlig reduksjon i systolisk blodtrykk på 3 mmHg betydelig. Mange deltakere og lang observasjonstid må til for å påvise en reduksjon i komplikasjoner og død.
«Noklus diabetesskjema» er et beslutningsstøtteprogram og en registreringsmodul for «Norsk diabetesregister for voksne (NDRV)». Innrapportering av data til registeret gir årlig tilbakemelding til fastlegen om kvaliteten på diabetesbehandlingen i praksisen. Data fra registeret kan gi viktig informasjon om status for diabetes oppfølging og behandling i Norge. Lokale rapporter gir fastlegen mulighet til ekstra oppfølging av pasientene med dårlig kontroll. Så vidt vi forstår skiller dette seg vesentlig fra de systemene som er benyttet i de inkluderte studiene. Disse studiene er derfor lite representative for å vurdere effekten av «Noklus diabetesskjema».
Data fra norsk allmennpraksis viser et stort forbedringspotensiale for oppfølging og behandling av diabetes. Undersøkelse av føtter, U-albumin og øyelege som anbefalt i de kliniske retningslinjer var kun dokumentert hos 25-70 % (2). For allmennpraktikere kan hjelpemidler som "Noklus diabeteskjema" fungere som viktig påminnelse, huskeliste og arbeidsverktøy som også kan bidra til redusert tidsbruk. Vårt inntrykk, som bekreftes ved upubliserte undersøkelser fra allmennpraksis i Salten og sykehusdata fra "NDRV", tyder på at bruk av strukturerte diabetesskjema/journaler fører til en bedring av kvaliteten på behandlingen. Vi er imidlertid enige med forfatterne at det er få gode undersøkelser om effekten av bruk av skjema, og at det er ønskelig med studier som kan avklare dette nærmere. Den pågående ROSA-4 undersøkelsen som inkluderer diabetespasienter fra allmennpraksis vil gi nyere data om dette.
Det er vanskelig å bli bedre hvis man ikke vet hva man gjør. Vi oppfordrer allmennleger til fortsatt bruk av "Noklus diabetesskjema" både for å få systematikk i diabeteskontrollene og for å levere data til diabetesregisteret.
Litteratur
1. Larun L, Bjørner T, Fretheim A et al. Bruk av skjema i oppfølgingen av diabetes i allmennpraksis. Tidsskr Nor Legeforen 2016; 136:417 – 22
2. Claudi T, Cooper JG, Hausken MF et al. Kvaliteten på diabetesbehandlingen i allmennpraksis. Tidsskr Nor Legeforen 2008; 22: 2570-4.