Paroxysmal nocturnal haemoglobinuria at Oslo University Hospital 2000–2010
The rare disease paroxysmal nocturnal haemoglobinuria (PNH) has an estimated incidence of 1.3 cases per million population per year (1). Patients are classified as either classic PNH (without other bone marrow disease), secondary PNH (associated with bone marrow diseases such as aplastic anaemia and myelodysplastic syndrome (MDS)) or subclinical PNH (associated with bone marrow disease) (2).
PNH is caused by a mutation in the gene phosphatidylinositol glycan, class A (PIG-A) in haematopoietic stem cells. The mutation leads to defective synthesis of the glycosylphosphatidylinositol (GPI) anchor. PNH cells lack proteins that normally bind to the membrane using this anchor, including CD55 and CD59. These two proteins protect cells against complement attack. Absence of the GPI anchor may give PNH cells a survival advantage by rendering them resistant to apoptosis and immunotherapy, thereby enabling PNH clones to become established in the bone marrow and cause disease (3). Complement normally undergoes continual activation via the alternative activation pathway. In healthy individuals this is of no consequence, but in patients with PNH, complement activation leads to haemolysis. The haemoglobin that is released binds nitric oxide (NO), thereby affecting components such as platelets and smooth muscle (4).
Classic PNH is a chronic systemic disease with high mortality (approximately 35 % 5‑year mortality), but spontaneous recovery after several years of illness is seen in about 15 % of cases (5). The disease affects all age groups. The main clinical symptoms are the result of unrestrained complement attack on the patient's own blood cells (6, 7). Classic PNH manifests clinically with chronic intravascular haemolysis, pancytopenia and recurrent venous thrombosis. The condition is often neither paroxysmal nor nocturnal, but is characterised by continual haemolysis – even when symptoms are absent (8). Haemoglobinuria is not always present (2).
PNH has been featured previously in the Journal of the Norwegian Medical Association (9, 10). Until around 2000, the disease was diagnosed by means of the sucrose haemolysis test and Ham’s test on the basis of complement activation by low ionic strength and low pH, respectively. However, these erythrocyte-based tests can give false negatives after extensive haemolysis, after transfusion and when the dominant PNH population consists of leucocytes. Diagnostic flow cytometry is sensitive and can identify pathological PNH populations more effectively than the aforementioned tests (10, 11). Flow cytometry detects deficient expression of CD55 and CD59 on the surface of erythrocytes, monocytes and granulocytes, and can reveal PNH clones that constitute only 0.3 % of the total cells (10). The use of flow cytometry will also reveal cases of secondary PNH with aplastic anaemia and myelodysplastic syndrome, often in patients who do not have signs of haemolysis (subclinical PNH) (3). In Norway, this method was recommended over the other tests as early as 1999 (10), and it has been used preferentially ever since. In recent years, diagnostics have been further improved through the combination of antibodies with a fluorochrome-labelled molecule (FLAER) that binds to the GPI anchor (12).
The treatment of PNH has been symptomatic – with hydration, pain relief and transfusions in haemolytic crises, and otherwise with iron replacement and anticoagulation therapy. Immunosuppressive therapy is used in secondary PNH (9). Allogeneic stem cell transplantation can provide a permanent cure, but is not first-line therapy because of significant treatment-related morbidity and mortality (13). A humanised monoclonal antibody (eculizumab) against complement protein C5, which prevents formation of the terminal complement complex, has shown good results (8). Eculizumab reduces haemolysis, transfusion requirements, thromboembolic events and mortality, and is also well-tolerated (14, 15). The recently published Norwegian guidelines for diagnosis and treatment of PNH (16) recommend eculizumab for selected patients.
In this article, we describe subtypes, course and treatment of PNH based on a review of the medical records of all patients who were tested for the disease using flow cytometry at the Department of Immunology and Transfusion Medicine, Section for Immunohaematology, Oslo University Hospital over a 10-year period.
Material and method
Patients
The dataset consists of patients who were tested for PNH using flow cytometry between February 2000 and September 2010, when we were responsible for PNH testing in the Southern and Eastern Norway Regional Health Authority.
Five of the patients are from other health authorities because Oslo University Hospital is responsible for stem cell transplantations across several health regions. Three of the patients have been described previously (9).
Diagnostic method
Flow cytometry was used for diagnosis throughout the entire period and has been described in detail previously (10). A test was interpreted as positive if the PNH clone(s) made up 1 % or more of the cell population examined, with absence of expression of CD55 and/or CD59 in at least two cell populations (Figure 1). We adjusted the clonal size for erythrocytes if the patient had received an erythrocyte transfusion in the last three months.
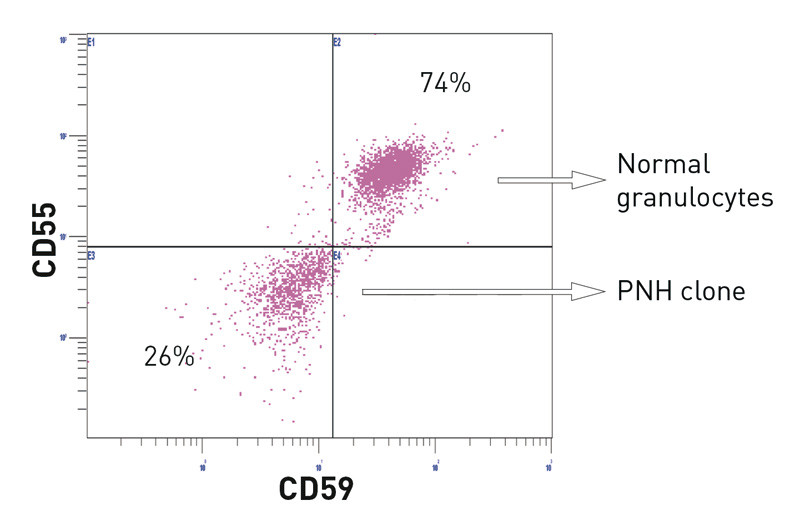
Variables
Medical and transfusion records were obtained between February 2012 and February 2013 by contacting the referring doctor and blood banks at each of the hospitals. We reviewed the records and noted the referral diagnosis, relevant blood-test results at the time of diagnosis and subsequently, comorbidities and disease course. We also recorded treatment, outcome, follow-up and the results of post-treatment blood tests. Data from the blood banks was recorded as the number of transfused erythrocyte concentrates. All data entry from the medical records was performed by a single individual (LSHNM).
On the basis of flow cytometric analysis and clinical data from the medical records, patients were classified (LSHNM) as classic PNH (chronic haemolysis and pancytopenia with no other bone marrow disease), secondary PNH associated with bone marrow disease, and subclinical PNH (without haemolysis) associated with bone marrow disease (2). For three patients, medical records were incomplete. Two of these did not have PNH. The third patient completed treatment in 2002 and has been included in the dataset with observational data up to 2006.
Data were processed using Excel software (Microsoft Office 2007).
Ethics
The study was approved by the Data Protection Officer at Oslo University Hospital (2011/20340).
Results
Indications
A total of 28 patients, 14 women and 14 men, were tested for PNH using flow cytometry. Indication for testing/referral diagnosis is shown in Table 1.
Table 1
Indication for requisition of flow cytometric PNH-analysis in 28 patients tested for paroxysmal nocturnal haemoglobinuria using flow cytometry in the Department of Immunology and Transfusion Medicine, Oslo University Hospital, Ullevål, 2000 – 2010. The table shows referral/principal diagnosis. DAT = direct antiglobulin test
| Indication |
Number of patients |
Number with confirmed paroxysmal nocturnal haemoglobinuria |
| Aplastic anaemia/myelodysplastic syndrome without haemolysis |
10 |
7 |
| Aplastic anaemia/myelodysplastic syndrome with haemolysis |
8 |
7 |
| Thrombosis |
4 |
3 |
| DAT-negative haemolytic anaemia |
3 |
2 |
| Chronic myeloproliferative disease |
2 |
2 |
| Paroxysmal nocturnal haemoglobinuria diagnosed with Ham’s test |
1 |
1 |
| Total |
28 |
22 |
Diagnostics
We received 41 blood samples (1 – 5 samples per patient). In all, 23 patients were from the Southern and Eastern Norway Regional Health Authority and five were from other regions. Flow cytometry revealed PNH in 22 of 28 patients, 10 men and 12 women. Four patients were diagnosed as classic PNH, 12 as secondary PNH and six as subclinical PNH. Median age at diagnosis was 50.5 years (14 – 79 years), median observation time after diagnosis is 52.5 months (1 – 365 months).
The diagnosis was disproved in four patients, while two were excluded from the dataset due to uncertainty around the diagnosis. Of the 22 patients with PNH, four had previously been investigated with Ham’s test, and three of them had tested negative.
Complications
Thromboembolic episodes were recorded in five patients, including two with classic PNH and three with secondary PNH. There were reports of venous thrombosis of the lower extremities and in the splanchnic area/«stomach area», portal vein thrombosis/Budd-Chiari syndrome, pulmonary embolism and splenic vein thrombosis. Haemolytic crises (abdominal pain and signs of massive intravascular haemolysis) were described in one patient, while two others showed renal failure and pulmonary hypertension, respectively.
Transfusion requirements
The number of transfusions received after diagnosis varied greatly (median 74, range 1 – 445), but there was also marked variation in the length of the observation period (1 – 365 months). Of 21 patients who were transfused, eight received > 100 erythrocyte concentrates. One patient (with classic PNH) did not require transfusion.
Treatment
Patients with classic PNH received antithrombotic therapy (warfarin, low-molecular-weight heparin, aspirin. One patient also received iron supplements and granulocyte colony-stimulating factor, while another received erythropoietin. Iron chelation therapy (deferasirox) was used in only one individual with classic disease. Another patient showed clinical improvement after prednisolone.
A total of ten patients (six with secondary and four with subclinical PNH) were treated with anti-thymocyte globulin (ATG) for bone marrow hypoplasia. Five went into complete remission, three died in the course of treatment, one still needed transfusions after treatment and another developed increasing haemolytic anaemia after a couple of years. Five patients with secondary or subclinical PNH underwent stem cell transplantation, one of whom had previously received ATG. Two of the five who underwent stem cell transplants were cured of their primary disease and PNH, whereas three died.
Six patients were treated with eculizumab, two following ATG. Median duration of eculizumab treatment at the end of data collection was 11 months (range < 1 month to 3.5 years). In three of the patients, treatment was discontinued due to transformation to myelodysplastic syndrome/acute myeloid leukaemia (AML) and bleeding complications. At the time of writing, three patients are being treated with eculizumab. Two have secondary PNH – one no longer has anaemia that requires transfusions, the other has reduced transfusion requirements. The third (with classical disease) was about to begin the treatment when the data were collected, but is now asymptomatic (G.E. Tjønnfjord, personal communication).
In all, 11 of the patients (two of whom had classic PNH) were deceased when the dataset was analysed. Seven died in the course of treatment (ATG or stem cell transplantation), three died after general deterioration/immune failure and one as a result of gastrointestinal bleeding.
Pregnancies
Four patients, one with classic and three with secondary PNH, have had a total of 11 pregnancies, eight before and three after diagnosis. The latter three pregnancies resulted in one live birth. In two patients suspicion of PNH arose during investigation of anaemia in pregnancy.
Discussion
Between 2000 and 2010, a total of 28 patients were tested for PNH using flow cytometry in our department, of whom 22 were found to have the disease. Venous thrombosis was described in five of 16 patients with symptomatic disease (31 %). This is comparable with other datasets, in which 33 – 50 % of patients were found to have thrombosis (5, 7), as were three of the nine patients in our previous study (9). Two patients with PNH were diagnosed with chronic myeloproliferative disease (myelofibrosis). PNH associated with chronic myeloproliferative disease is rare, but has been described previously (17).
PNH itself is a rare disease. Despite centralised diagnosis and treatment at a regional level, our dataset is small and firm conclusions cannot be drawn. Our main aim is to remind others of the disease so that as many patients as possible can be identified.
Whereas nine patients with suspected PNH were referred to the Department of Haematology, Oslo University Hospital in the 20 years prior to 2001 (9), in the course of a decade (2000 – 2010) we received samples from 28 patients and detected PNH clones in 22 of them. The threshold for requesting PNH analysis has probably become lower, and flow cytometry is far more sensitive than Ham’s test (11). Nevertheless, it is surprising that so many patients (79 %) tested positive. This may be an indication that too few tests are being requested because PNH is rarely suspected.
Since 2010, flow cytometry-based diagnosis of PNH has been improved further by combining even more antibodies with FLAER, a fluorochrome-conjugated (Alexa 488) non-lysing mutated proaerolysin, which binds specifically to GPI in the cell membrane (12).
Only six patients were treated with eculizumab, a monoclonal antibody against complement factor C5. In three patients, the drug probably contributed to improvement, in the other three further treatment was not possible because eculizumab was no longer indicated. It is a weakness of the study that we do not have long-term follow-up data from more patients who have been treated with eculizumab.
New and better methods for the diagnosis and treatment of PNH have improved the prognosis for this population (15). The Norwegian guidelines for diagnosis and treatment of PNH have recently been updated (16), and devote significant space to the use of eculizumab. International studies show that the drug reduces transfusion requirements and improves life expectancy among the most severely affected, and is also well tolerated (14, 15).
C5 deficiency is associated with increased risk of meningitis (Neisseria meningitidis) (18). Patients who are to receive eculizumab should therefore be vaccinated against meningococcal disease before the start of treatment. They should also be given phenoxymethylpenicillin for self-treatment and/or a safety information card stating that they should be treated without delay on suspicion of meningococcal disease (19). In Norway, eculizumab is indicated for symptomatic PNH (classic and secondary) with anaemia-related fatigue that impairs social functioning, frequent episodes of abdominal pain, thromboembolism and organ involvement (16).
Pregnancy in these patients entails a high risk of complications for both mother and foetus. Treatment of pregnant women with eculizumab has shown promising results (20). Monitoring of these patients requires close collaboration between the haematologist and specialists in foetal medicine with specific experience.
We have previously called for increased awareness of PNH with direct antiglobulin test (DAT)-negative haemolytic anaemias, aplastic anaemia and Budd-Chiari syndrome of unknown aetiology (9). In our dataset, aplastic anaemia/myelodysplastic syndrome with or without haemolysis was the indication for flow cytometric analysis in 18 of 28 patients, showing that previous efforts probably have contributed to increased awareness of the disease. We continue to urge general practitioners, specialists in internal medicine and haematologists to keep this condition in mind. Although PNH is very rare, and knowledge and experience of it is limited among health professionals, it is important to be aware of the disease because correct treatment improves survival (15). Moreover, the presence of a PNH clone predicts a favourable response to immunosuppressive therapy in aplastic anaemia (21).
More sensitive diagnostic methods that also reveal secondary/subclinical PNH associated with bone marrow disease have transformed the previous view of the disease as a relatively benign condition (9). The dataset presented here illustrates that this is a serious disease, as larger datasets have shown (5), but also a condition with extensive variation in symptoms and severity.
We wish to thank Knut Liseth at Haukeland University Hospital, Birgitte D. Eiken at Østfold Hospital Fredrikstad and Marit Rinde at Akershus University Hospital for help in obtaining patient records. We are also grateful to the blood banks for data on transfusions and to Annette Vetlesen for transfusion data from the Blood Bank of Oslo.
MAIN MESSAGE
Over the period 2000 – 2010, we analysed 28 blood samples from patients with suspected paroxysmal nocturnal haemoglobinuria (PNH) using flow cytometry
PNH was detected in 22 samples
In 18 of the patients, PNH was secondary to another bone marrow disease
- 1.
Borowitz MJ, Craig FE, Digiuseppe JA et al. Guidelines for the diagnosis and monitoring of paroxysmal nocturnal hemoglobinuria and related disorders by flow cytometry. Cytometry B Clin Cytom 2010; 78: 211 – 30. [PubMed]
- 9.
Dalgaard J, Brinch L, Tjønnfjord GE. Paroksystisk nattlig hemoglobinuri – en sjelden sykdom med mange ansikter. Tidsskr Nor Lægeforen 2002; 122: 374 – 8. [PubMed]
- 10.
Vetlesen A, Kjeldsen-Kragh J, Tjønnfjord GE. Paroksystisk nattlig hemoglobinuri – diagnostikk ved hjelp av væskestrømscytometri. Tidsskr Nor Lægeforen 1999; 119: 3909 – 13. [PubMed]
- 16.
Tjønnfjord EB, Tjonnfjord GE. Norske retningslinjer for diagnostikk og behandling av pasienter med paroksystisk nattlig hemoglobinuri (PNH). 2013. http://legeforeningen.no/Fagmed/Norsk-selskap-for-hematologi/Handlingsprogrammer/ (15.3.2014).