Lung cancer is the second most prevalent form of cancer in Norway for men and the third for women (1 p. 26), and the type of cancer that claims most lives (2). Five-year survival in Norway is approximately 12 – 16 % (1 pp. 68 – 69). In 2011, 2 842 new cases of lung cancer were diagnosed in Norway (1 p.25). Non-small cell lung cancer accounted for about 80 % of these. Approximately 20 % of patients with this type of cancer undergo surgery (3), while 4 % (about 120 patients per year) receive radiotherapy with a curative intent. Surgery is the most important type of treatment for achieving a cure, and almost all long-term survivors (> 5 years) have undergone surgery (4). In a dataset from the Cancer Registry of Norway, Strand et al. found that two-year survival was 75 % for operated patients and 23 % for all lung cancer patients during the same period (3). Correct selection for surgery is crucial, both in order to identify patients who have an operable tumour, and to prevent patients with locally advanced or metastatic disease being subjected to a futile operation.
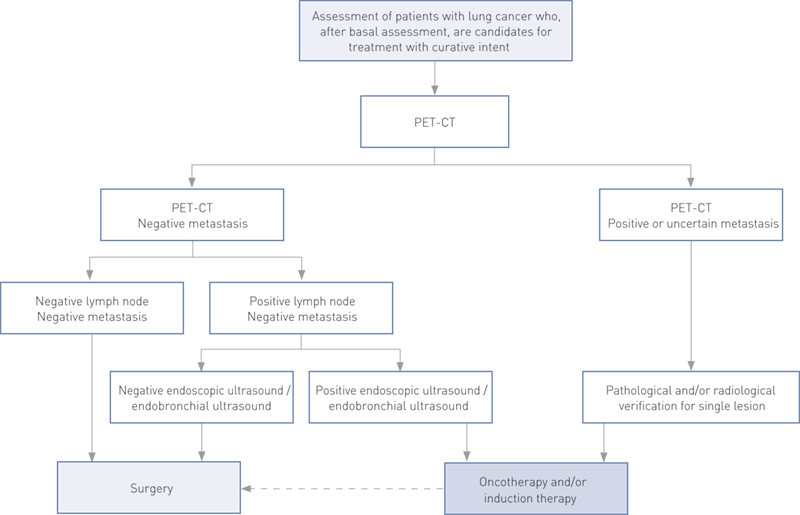
Both international (5) and national guidelines recommend that the majority of patients with non-small cell lung cancer who are found after a primary assessment to have a curable disease, undergo 18F-fluoro-2-deoxyglucose (FDG) positron emission tomography (PET) combined with computed tomography (CT) prior to surgery (7). Hereafter in the article, when we write PET-CT, it is FDG-PET-CT that is meant. Patients found to have spreading to lymph nodes in the mediastinum (N2 or N3 disease) and/or remote metastases, are generally regarded as inoperable and referred for oncotherapy. It is assumed in these cases that PET-CT findings arousing suspicion of malignancy will be verified by invasive tests (fine-needle biopsy) or by MRI of the suspected skeletal metastases.
Use of PET-CT commenced at Radiumhospitalet in 2005 and at Rikshospitalet in 2006. The aim of this article is to report on the results of using this method with patients who had a PET-CT performed as part of an assessment of their potentially operable non-small cell lung cancer at Rikshospitalet.
Material and method
Patients
In the period 2007 – 2011, 651 patients with potentially operable non-small cell lung cancer were assessed and treated at Rikshospitalet. PET-CT scans were conducted on 533 patients as part of the TNM classification prior to possible surgery (Fig. 1, Table 1). The calculations in this study are restricted to these 533 patients. We calculated sensitivity, specificity and positive and negative predictive value for spreading to mediastinal lymph nodes. Histological or cytological diagnosis, based on preoperative fine-needle biopsy, aspiration cytology or examination of a post-operative tissue specimen, was used as the gold standard. In some cases we had no material from mediastinal lymph nodes. In these cases, we took findings of metastases verified by biopsy or MRI of the lesion in question as evidence of spreading to mediastinal lymph nodes.
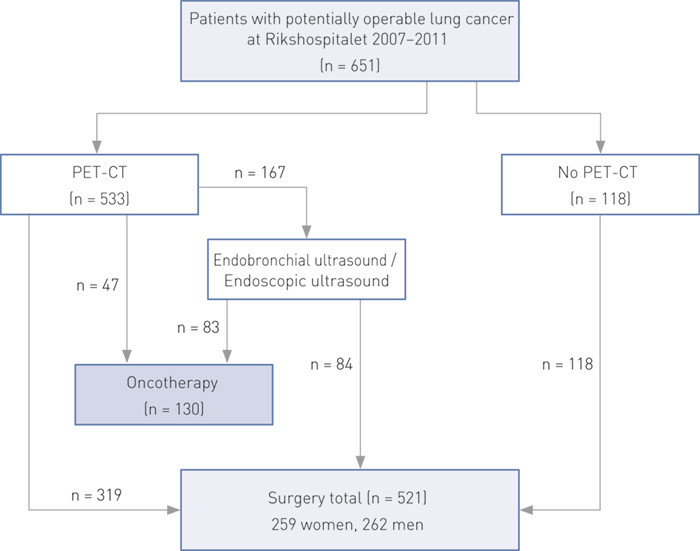
Table 1a
Patients with potentially operable non-small cell lung cancer examined with PET-CT at Rikshospitalet 2007 – 2011. Number (%) unless otherwise specified
| Examined with PET-CT (n = 533) |
||
| Age, years; mean, (SD) |
66 |
(9) |
| Women |
250 |
(47) |
| Histology |
|
|
| Adenocarcinoma |
296 |
(56) |
| Squamous cell carcinoma |
122 |
(23) |
| Carcinoid |
43 |
(8) |
| Non-small cell lung cancer, unclassified |
29 |
(5) |
| Large cell lung cancer |
23 |
(4) |
| Bronchioalveolar cell carcinoma |
20 |
(4) |
Table 1b
Patients with non-small cell lung cancer operated upon at Rikshospitalet 2007 – 2011. Number (%) unless otherwise specified
| Examined with PET-CT (n = 403) |
Not examined with PET-CT (n = 118) |
||||
| Age [years; mean, (SD)] |
66 |
(9) |
62 |
(13) |
|
| Women |
195 |
(48) |
63 |
(54) |
|
| Histology |
|||||
| Adenocarcinoma |
230 |
(57) |
58 |
(49) |
|
| Squamous cell carcinoma |
94 |
(23) |
25 |
(21) |
|
| Carcinoid |
19 |
(5) |
23 |
(20) |
|
| Non-small cell lung cancer |
|||||
| Unclassified |
26 |
(6) |
3 |
(3) |
|
| Large cell lung cancer |
19 |
(5) |
4 |
(3) |
|
| Bronchioalveolar cell carcinoma |
15 |
(4) |
5 |
(4) |
|
Patients were referred from hospitals in the former Southern Norway Health Region after undergoing primary assessment locally. Such assessment takes place in accordance with national guidelines (6) and includes clinical examination including assessment of co-morbidity, exploratory blood tests, bronchoscopy and diagnostic CT of the thorax and upper abdomen. Since 2005, all thoracic surgery in the former Southern Norway Health Region has been centralised at Rikshospitalet. Patients who are found after examination to have a potentially operable tumour, and who do not have co-morbidity that contraindicates surgery, are referred by their respective hospitals to the weekly regional thoracic surgery meeting at Rikshospitalet (video conference). It is here that the final decision is made as to whether the patient should be offered surgery or referred for oncotherapy.
Unfortunately, the majority of patients found to have lung cancer have already reached an advanced stage of the disease and are unsuitable for surgery. These patients are generally not referred to Rikshospitalet, but receive oncotherapy at their local hospital or at Radiumhospitalet. Our dataset therefore does not consist of a representative selection of all lung cancer patients in the region, but of patients initially intended to undergo surgery. We originally wanted to refer all patients with a potentially technically operable tumour directly to PET-CT (Fig. 2). However, PET-CT capacity was limited, and the number of patients varied. In addition, a waiting period of > 14 days and/or postponement of the operation date while waiting for a PET-CT for patients who were otherwise fully assessed, was regarded as unacceptable. A total of 118 patients with a potentially operable tumour (20 – 30 per year) underwent operation without a PET-CT being carried out (Fig. 1). Where there was a need to prioritise, an assessment was made, and in line with the national (6) and US guidelines (5), stage 1A patients underwent surgery without a prior PET-CT (n = 49). PET-CT was also omitted for a number of patients with carcinoids (n = 23), since the examination does not always result in a clear positive in this patient group. These patients can to advantage also be assessed with octreotide scintigraphy (8). A further 46 patients had surgery without prior PET-CT on the basis of discretionary assessments.
Data was consecutively recorded in the cancer database (Medinsight) of the Department of Respiratory Medicine. Written consent for entry in the database was obtained from all registered patients. We do not have an overview of how many did not consent to registration, but our impression is that they were few. The study is part of a larger protocol that was approved by the Regional Ethics Committee (S-07130a) and the Rikshospitalet’s Data Protection Officer. Our study is not described in detail in the protocol, and is therefore retrospective. For the same reason, it is not registered as a clinical trial in a public register.
Staging and classification
Lung cancer may metastasize via the bloodstream to virtually every organ in the body, but most frequently to another lobe of the lungs, the suprarenal glands, skeleton, brain or liver. Usually spreading takes place first via the internal lymph drainage of the lung and on to the mediastinal lymph nodes (9).
When patients with lung cancer are assessed in Norway, the 7th international TNM classification (10) is used to describe the preoperative extent of the tumour (clinical TNM = cTNM) as well as a histological examination of the post-operative tissue specimen (pathological TNM = pTNM) (Table 2, Table 3).
Table 2
TNM classification of size and extension of lung cancer pursuant to ref. (10)
| Tumour/T stage | ||
| T1 |
Tumour largest diameter ≤ 3 cm surrounded by lung parenchyma or visceral pleura, distal to main bronchus |
|
| T1a |
Tumour largest diameter ≤ 2 |
|
| T1b |
Tumour largest diameter > 2 cm, but ≤ 3 cm |
|
| T2 |
Tumour largest diameter > 3 cm, but ≤ 7 cm Or tumour that affects main bronchus ≥ 2 cm from carina, which is invading visceral pleura, or tumour causing partial atelectasis/obstructive pneumonitis |
|
| T2a |
|
|
| T2b |
|
|
| T3 |
Tumour > 7cm or tumour (irrespective of size) with one of the following properties:
|
|
| T4 |
Tumour irrespective of size with following properties:
|
|
| Regional lymph nodes/N stage | ||
| N0 |
No detectable pathological regional lymph nodes |
|
| N1 |
Metastases to ipsilateral, peribronchial and/or ipsilateral hilar lymph nodes, and intrapulmonary lymph nodes including lymph nodes in direct relationship with primary tumour |
|
| N2 |
Metastases to ipsilateral, mediastinal and/or subcarinal lymph nodes |
|
| N3 |
Metastases to contralateral, mediastinal, contralateral hilar, ipsilateral or contralateral scalene or supraclavicular lymph nodes |
|
| Remote metastases/M stage | ||
| M0 |
No remote metastases detected |
|
| M1 |
Remote metastases present |
|
| M1a |
|
|
| M1b |
Remote metastases |
|
Table 3
Classification of lung cancer into stages according to (10). The column assessment is based on national (6) and international guidelines (8)
| Stage |
Classification |
Assessment |
| Ia |
T1a-b, N0, M0 |
Operable |
| Ib |
T2a, N0, M0 |
Operable |
| IIa |
T2b, N0, M0 |
Operable |
| T1a-b, N1, M0 |
Operable |
|
| T2a, N1, M0 |
Operable |
|
| IIb |
T2b, N1, M0 |
Operable |
|
|
T3, N0, M0 |
Operable |
| IIIa |
T1a-T2b, N2, M0 |
Rarely operable |
| T3, N1-N2, M0 |
Possibly operable |
|
| T4, N0-N1, M0 |
Inoperable |
|
| IIIb |
T4, N2, M0 |
Inoperable |
|
|
T1a-T4, N3, M0 |
Inoperable |
| IV |
T1a-T4, N0 – 1, M1 |
Inoperable |
Integrated PET-CT
Diagnostic multidetector computed tomography (MDCT) with intravenous contrast medium is the standard examination for assessing lung cancer (11). The examination provides accurate information about the extent of the primary tumour (T stage), but has limitations regarding the distinction between malignant and benign lymph nodes (N stage). During the past ten years, it has been found that PET with the glucose analogue FDG combined with CT in the same session is the best imaging technique for revealing both spreading to lymph nodes and extrathoracic metastases (12). PET is a functional diagnostic imaging procedure based on detection of radioactive isotopes that emit positrons. FDG is the radiopharmacon (tracer) most frequently used in PET. FDG is taken up by cells in the same way as ordinary glucose, and reflects the energy needs of the cells. Its absorption is physiologically high in organs with a high glucose metabolism (heart, brain, liver, kidneys), in inflammatory processes and in malignant tumours. Visualisation of the absorption takes place by means of a PET scanner, which images and measures the distribution of the radioactive substance in the body (13). Integrated PET-CT scanners which automatically fuse images are used today. CT is used for both anatomical localisation and attenuation correction of PET scans in order to optimise the images (Fig. 3a-e).
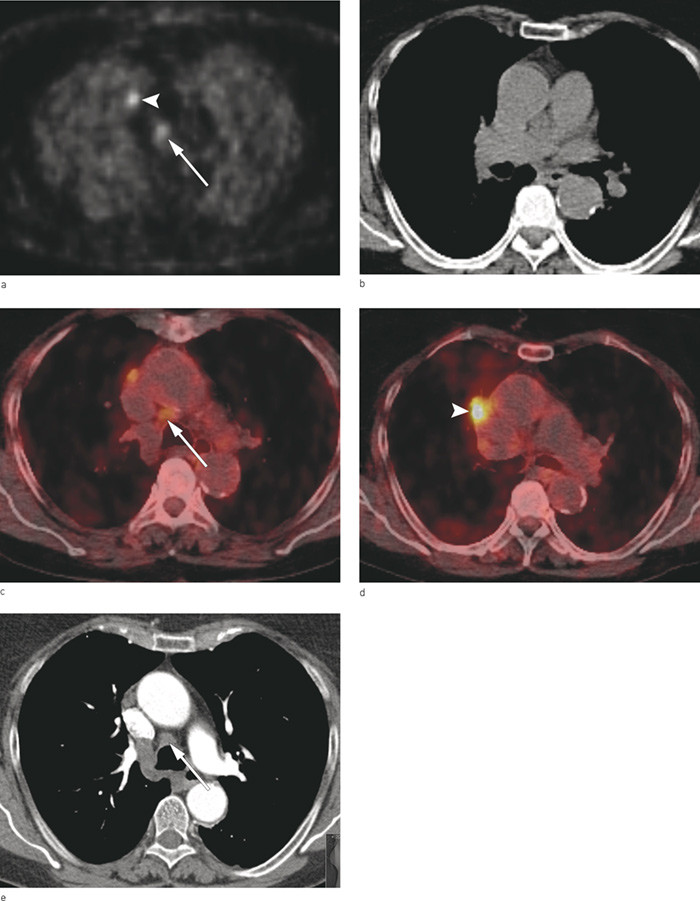
FDG-PET combined with low-dose CT in the same session was carried out using a PET-CT scanner with a 64-channel multidetector CT (Biograph 64, TruePoint PET-CT, Siemens Medical Solutions, Forchheim, Germany). The patient fasted for six hours before the examination. About an hour before scanning, approximately 370 MBq FDG was administered intravenously. PET scans were taken with normal respiration and at the same time a low-dose CT scan was conducted from the top of the head to the thighs. For patients who had not undergone a high quality multidetector CT in the course of the last 4 – 5 weeks at the referring hospital, this was repeated, either in combination with the PET examination or in the Department of Radiology (GE Lightspeed VCT, 64-channel CT scanner) following intravenous injection of 100 ml Visipaque 320 mg I/ml (GE Healthcare, Oslo). The PET and low-dose CT were examined merged and individually. Information from diagnostic CT performed at the referring hospital or at Rikshospitalet was also used in the evaluation. PET and low-dose CT images were examined on a Leonardo workstation (Siemens Medical Solutions, Forchheim, Germany). The CT images were examined in PACS (picture archiving and communication system from Sectra Medical Systems, Linköping, Sweden).
Extrathoracic metastases
If the PET-CT aroused suspicion of a metastatic disease (n = 47), i.e. the PET examination revealed an FDG uptake where malignancy was either very possible or could not be excluded with certainty, a biopsy was conducted of the focus that was most readily accessible for such a procedure. On suspicion of skeletal metastases (n = 9), a positive MRI finding of the area in question was accepted as diagnostic. Other findings were always verified by means of cytology or histology tests.
Metastases to local lymph nodes
Because the most common path by which lung cancer metastasises is via the lymphatic system, either endobronchial or endoscopic ultrasound-guided fine-needle aspiration of PET-positive lymph nodes was carried out. The procedure was also applied to lymph nodes with a minimum diameter of > 1 cm, irrespective of the PET result. The histological or cytological diagnosis was used as the gold standard, and patients found to have cancer infiltration in mediastinal lymph nodes (N2 and N3 disease) were generally regarded as inoperable. The others were referred for surgery. Mediastinoscopy was not carried out.
Statistics
Descriptive analyses were performed with the aid of the register support tool Medinsight, developed at the Institute for Cancer Genetics and Informatics (ICGI), Oslo University Hospital (14). Descriptive data are specified in terms of average value and spread. Sensitivity, specificity, positive and negative predictive value, as well as diagnostic accuracy and kappa coefficient for agreement between PET-CT and actual findings of malignant or benign lymph nodes, are calculated using standard formulae (15).
Results
In the period 2007 – 2011, PET-CT was conducted on 533 patients with lung cancer. Their mean age was 65 (SD 9) for women (n = 250; 47 %) and 66 (SD 9) for men (n = 283; 53 %) (Table 1). Of the 533 patients, 403 (76 %) were assessed as being operable and underwent surgery with curative intent (Fig. 1). PET-CT revealed remote metastases in 47 (9 %) of patients: 12 in the contralateral lung, nine in the skeleton, three in the suprarenal glands and 18 in combinations of these. Metastases to the liver were found in three patients, and to the brain in two. Table 4a-b shows that PET-CT findings were true positive with respect to spreading to mediastinal lymph nodes in 87/533 cases, i.e. PET-CT helped to detect spreading in 16 % of the examinations.
Table 4a
Result of PET-CT with malignant and benign lymph nodes in the mediastinum. Potentially operable patients examined with PET-CT at Rikshospitalet 2007 – 2011
| Malignant lymph nodes (n = 111) |
Benign lymph nodes (n = 422) |
Total |
|
| Positive PET-CT |
87 |
50 |
137 |
| Negative PET-CT |
24 |
372 |
396 |
| Total |
111 |
422 |
533 |
Table 4b
Sensitivitity, specificity and predictive value of PET-CT for spreading to mediastinal lymph nodes among examined patients with potentially operable tumour, Rikshospitalet 2007 – 11
| Measurement |
Estimate (95 % CI) |
||
| Sensitivity (%) |
78 |
(70 – 86) |
|
| Specificity (%) |
88 |
(85 – 91) |
|
| Diagnostic accuracy (%) |
86 |
(83 – 89) |
|
| Positive predictive value (%) |
64 |
(55 – 72) |
|
| Negative predictive value (%) |
94 |
(91 – 96) |
|
| Kappa agreement |
0.61 |
(0.53 – 0.69) |
|
Histological examination of the post-operative tissue specimen or endobronchial or endoscopic ultrasound-guided fine-needle aspiration confirmed 372/533 (70 %) of negative PET-CT findings. 24/533 (5 %) false negatives were found, and 50/533 (9 %) false positives. Sensitivity and specificity were 78 % (95 % CI: 70 – 86) and 88 % (95 % CI: 85 – 91), respectively. The calculated positive predictive value was 64 % (95 % CI: 55 – 72) and the negative predictive value 94 % (95 % CI: 91 – 96). Diagnostic accuracy was 86 % (95 % CI: 83 – 89). Kappa agreement between PET-CT and the actual findings of malignant or benign mediastinal lymph nodes (Table 4b) was calculated to be 0.61 (95 % CI 0.53 – 0.69), which is graded as good (15).
Discussion
This study showed that PET-CT was a useful tool for selecting potentially operable lung cancer patients at Rikshospitalet in the period 2007 – 2011. Pathological uptake of FDG in mediastinal lymph nodes led to targeted biopsies of N2 and N3 lymph nodes, thereby helping to ensure correct staging of the patients’ disease. This is consistent with previous studies (16). PET-CT also led to the detection of occult remote metastases in 9 % of our patients, who had this verified by means of biopsy or MRI of the lesion in question. Without PET-CT, these metastases would very likely not have been detected before they became symptomatic. Although some of these remote metastases can be detected by means of CT, whole body CT scans are not conducted as a matter of routine on asymptomatic patients (17). CT also has limitations when it comes to skeletal metastases, and a recently published meta-analysis (18) shows that PET-CT is superior to MRI or scintigraphy as an imaging method for detecting skeletal metastases. The number of patients with spreading to the contralateral lung (12 (25 %)) may appear high, but it proves to be consistent with a previous study by MacManus et al. (19). Surprisingly, they found metastases in 32 of 167 patients, where the proportion with metastases to the contralateral lung that were not seen on an ordinary CT was 10/32 (30 %). In our view, this strengthens the indication for using PET-CT for assessing lung cancer.
Because the population in our study is not a representative selection of all lung cancer patients in a population, this limits the possibility of generalising our results to a random selection of the general population. We wanted to carry out a PET-CT on all potentially operable patients, but for reasons of capacity had to prioritise. About half of the group who underwent surgery without a prior PET-CT consisted of patients with stage 1A lung cancer. It is therefore reasonable to believe that the prevalence of operable patients in this group was higher than in the one examined with PET-CT. Since calculations of positive and negative predictive value depend on prevalence, we cannot generalise the results to the entire group of potentially operable patients.
Earlier studies have shown that PET-CT may be false positive for spreading to mediastinal lymph nodes in 3 – 16 % of scans (12). In our survey, 9 % of findings were false positives. The reasons for the false positive findings may be inflammation (infectious and non-infectious), granulomas, or they may be iatrogenic, in connection with biopsy-taking. It is therefore important to specify that PET-CT cannot replace invasive examination where the latter is indicated (20). However, the examination can increase the accuracy of further assessment. In practice, we have seen few iatrogenic causes of false positive scans.
PET-CT in combination with diagnostic CT with intravenous contrast yields an accurate assessment of primary tumour, mediastinal lymph nodes and remote metastases. If these scans do not arouse suspicion of spreading to either mediastinal lymph nodes or other organs, the patient can with few exceptions be referred directly for surgery without further scans such as endobronchial ultrasound, endoscopic ultrasound, MRI or scintigraphy (6). This can help to shorten the assessment time. Using PET-CT to detect remote metastases in patients with unknown tumour histology will also enable a biopsy to be taken from the most superficial and readily accessible lesion. Taking a biopsy sample from such a lesion may often be a less risky procedure than taking a sample from a primary tumour in a lung or from lymph nodes in the mediastinum. Mediastinoscopy was not needed in our examination in order to reach a definite diagnosis. In modern lung cancer assessment, the use of PET-CT and endoscopic ultrasound-guided fine-needle aspiration has now largely replaced mediastinoscopy. We found that PET-CT had a 94 % negative predictive value in our population. This indicates that it may be justified to refrain from further assessment if the PET-CT is negative in the mediastinum or other organs; this is also recommended in the guidelines (6). Our results are supported by other, similar studies (21). A Danish study has shown that systematic use of PET-CT is more cost-effective than conventional assessment alone for avoiding futile thoracotomies (22).
However, some lung cancer patients still have such extensive comorbidity that any kind of invasive assessment is contraindicated and a histological diagnosis cannot be made (23). Stereotactic radiotherapy may offer an alternative type of treatment for some of these patients, and they, too, should be assessed with PET-CT to enable optimal planning of the treatment (6). Individual stage IIIA and IIIB patients with certain positive prognostic factors may also be candidates for curative radiotherapy even if from a technical point of view they are not operable, as also indicated in the Norwegian guidelines (6).
We found a kappa agreement of 0.61. Kappa agreement is a measure of the reliability of the examination and reflects the extent to which there will be consistency between two examinations if they are repeated under identical conditions. Kappa gradation tables have been developed, in which agreement of over 0.6 is graded as good (i.e. > 60 % better agreement in the event of examination than if the result of the examination had been entirely random). We also found a diagnostic accuracy of 86 % (95 % CI: 83 – 89). This represents the sum of all positive and negative examinations that were true positive or true negative, divided by the total number of scans carried out.
Conclusion
PET-CT was a useful tool for selecting potentially operable lung cancer patients at Rikshospitalet in the period 2007 – 2011. We found a high negative predictive value for spread to mediastinal lymph nodes. Provided that the population we scan with PET-CT does not change over time, this indicates that patients with a negative PET-CT can with few exceptions be referred directly for surgery without further invasive assessment. This is also consistent with national guidelines (6).
In cases where there was pathological FDG uptake in lymph nodes and remote metastases, PET-CT helped in the targeting of further invasive diagnostics. Its positive predictive value in our dataset was low, and positive findings – irrespective of whether these are in local lymph nodes or in other foci – must be assessed further in all patients where findings of malignancy are of decisive importance for further treatment (4, 21).
We should like to thank Nurse Cathrine R. Berg of the Department of Respiratory Medicine for her work in updating and maintaining the database, and Dr Liv Ingunn B. Sikkeland for reviewing and assisting with the didactic presentation.
- 1.
Cancer Registry of Norway. Cancer in Norway 2011 – Cancer incidence, mortality, survival and prevalence in Norway. Oslo: Kreftregisteret, 2013. http://kreftregisteret.no/no/Generelt/Nyheter/Cancer-in-Norway-2011/ (5.2.2014).
- 4.
Naidoo R, Windsor MN, Goldstraw P. Surgery in 2013 and beyond. J Thorac Dis 2013; 5 (suppl 5): S593 – 606. [PubMed]
- 5.
Silvestri GA, Gould MK, Margolis ML et al. Noninvasive staging of non-small cell lung cancer: ACCP evidenced-based clinical practice guidelines (2nd edition). Chest 2007; 132 Suppl 3): 178S – 201S.
- 6.
Nasjonalt handlingsprogram med retningslinjer for diagnostikk, behandling og oppfølging av lungekreft. Oslo: Helsedirektoratet, 2014. www.nlcg.no/sites/default/files/files/140203%20Lungekrefthandlingsprogram(2).pdf (5.2.2014).
- 7.
Kunnskapssenteret. Metodevarsel. Klinisk nytte av PET integrert med CT for ikke-småcellet lungekreft. Mednytt nr. 3/2010. www.kunnskapssenteret.no/Publikasjoner/_attachment/9539 (5.2.2014).
- 8.
National Comprehensive Cancer Network. Clinical practice guidelines in oncology. www.nccn.org/professionals/physician_gls/f_guidelines.asp (6.2.2014).
- 11.
Berstad AE, Kolbenstvedt A, Aaløkken TM et al. Computertomografi ved lungekreft – teknikk og kvalitet. Tidsskr Nor Lægeforen 2003; 123: 3384 – 6. [PubMed]
- 13.
Berstad AE, Solheim H, Bugge A. Molekylær avbildning. Tidsskr Nor Legeforen 2010; 130: 1622. [PubMed]
- 14.
Medinsight. Om Medinsight. http://medinsight.no/om-medinsight/ (6.2. 2014).
- 15.
Altman DG. Practical statistics for medical research. 1.utg. New York, NY: Chapman and Hall, 1991.