Limited malignancy screening of patients with idiopathic venous thromboembolism
Trousseau’s syndrome, which describes a significantly high incidence of venous thromboembolism in cancer patients, has been known since the mid-1800s (2) – (4). Sometimes venous thromboembolism may be the first sign of malignancy. In 27 – 46 % of cases the condition has no readily identifiable risk factor, and is therefore often called idiopathic venous thromboembolism (5).
Studies reveal occult cancer in 4 – 13 % of patients with idiopathic venous thromboembolism (3, 6) – (14). Identification of cancer is of significance not only for the handling of the cancer, but also for how the thrombosis ought to be treated (15). International guidelines recommend that patients with idiopathic venous thromboembolism undergo limited screening for occult malignancy, and more extensive screening if the limited screening reveals abnormal findings (16) – (18).
However, there is no full consensus on what «limited screening» should entail, or which patients should be offered more extensive screening. In the only randomised study involving a comparison of extensive work-up with no screening with occult malignancy in mind, it was suggested that occult maligancy could be detected at an earlier stage, but there was no clear answer as to whether it was of significance for the prognosis (19). Although some studies have shown that some patients still have an early stage of cancer when occult malignancy is detected (7), it is not clear whether early detection as a result of extensive screening is of importance to the prognosis and the patient’s quality of life (6).
As a result of the Norwegian Coordination Reform and use of new oral anticoagulants as an alternative to low-molecular heparin and warfarin, follow-up of patients with venous thromboembolism will be transferred to the primary health service earlier than previously. There is a need for clear guidelines as to what screening should be done, and who should do what (9).
Since 2003, we have been following a systematic, limited strategy with regard to such screening at the Thrombosis Clinic at Bærum Hospital.
The main purpose of this study was to investigate the extent to which limited screening for malignancy in cases of idiopathic venous thromboembolism, as conducted at Bærum Hospital, detects occult cancer. At the same time, we wanted to investigate how large a percentage of patients with idiopathic venous thromboembolism were found to have cancer in the course of the first year after the thrombosis diagnosis, and whether they differed from those who were not found to have cancer with respect to age, gender and location of the thrombosis.
Material and method
The records of all the patients registered with ICD 10 diagnostic codes for deep vein thrombosis and/or pulmonary embolism (all I80 and I26 codes) at Bærum Hospital in the period 1.1. 2003 – 31.12. 2008 were reviewed to check that the diagnostic code was correct. If there was a definite or highly probable radiological diagnosis, the diagnosis was regarded as confirmed. If the radiological diagnosis was uncertain, a discretionary evaluation was made, and considerable weight was attached to the assessment originally made by the doctors providing treatment.
Patients were excluded if the venous thromboembolism in question was associated with known cancer, surgical procedures or injuries during the previous three months, immobilisation because of illness, pregnancy/giving birth, oestrogen therapy, hereditary disposition for thrombosis (except heterozygote factor V Leiden mutation), severe infection, severe dehydration, intravenous drug abuse or a central venous catheter.
Venous thromboembolism without these exclusion criteria was defined as idiopathic venous thromboembolism. All patients who had suffered idiopathic venous thromboembolism were invited to take part in a follow-up project which involved completing a questionnaire. Participants were asked whether they had had other illnesses after the venous thromboembolism. The records of all respondents were also reviewed with malignancy in mind, and all information about cancer was verified in the records. The records of those who had died or had not responded to the survey were reviewed with respect to malignancy following venous thromboembolism. In the latter cases, the cancer diagnosis was based solely on information in the patients’ records.
«Occult cancer» is defined as malignancy that was not known when venous thromboembolism was detected, but was identified in the course of the 12 months following the diagnosis date.
The limited screening at Bærum Hospital involves patient-reported history with emphasis on possible malignancy symptoms, a physical examination with examination of lymph nodes and rectal exploration, palpation of the testicles and prostate in men and palpation of the breasts in women. Standard blood tests are performed, including haematology tests, liver and kidney function, CRP and, in men, a PSA test. Urine and stool are tested for the presence of blood. Women are also referred for a mammogram and gynaecological examination if this has not been done for the past 12 months. If CT thorax has not been carried out in connection with the diagnostic work-up for pulmonary embolism, an X-ray thorax is taken as part of the malignancy screening. Further tests are carried out if indicated by abnormal findings in the limited screening. These tests depend on which abnormal findings are made.
The data are described in terms of a frequency distribution for category variables and a median (minimum/maximum) for continuous variables, as these do not have a normal distribution. The results of the tests conducted in the limited screening were designated positive if there was a certain deviation from the normal in the records, and negative if no abnormal findings had been observed.
The incidence of new cancer cases in patients with idiopathic venous thromboembolism during the first year following the thrombosis were compared with the incidence of cancer in an age- and gender-matched control group from the general population of Asker and Bærum Municipality in the years 2003 – 2008, based on figures from the Cancer Registry of Norway obtained specifically for this project. The chi-squared test was used for the comparison. Negative predictive value was used to determine whether limited malignancy screening of the patient population in question would be capable of excluding occult cancer.
The point of departure for the study was the Bærum Hospital Thrombosis Register, which is consent-based and approved by the Regional Ethics Committee. The Data Protection Officer for Research has recommended that it be expanded. All patients treated for thrombosis at the Bærum Hospital Thrombosis Clinic are invited to take part when they are diagnosed.
Thrombosis data, therapy, risk factors and related conditions are registered for those who consent. All information about subsequent cancer is collected retrospectively, as described above, and incorporated in the Thrombosis Register in the case of those who have consented to this. For those who had died or not given their consent, a journal review was conducted as part of quality assurance pursuant to Section 26 of the Norwegian Health Personnel Act.
All personal data were processed as described in the recommendation by the Data Protection Officer. The recommendation entails anonymisation in the absence of consent.
Results
In the period 2003 – 2008, 974 patients were treated for venous thromboembolism at Bærum Hospital. Idiopathic venous thromboembolism was identified in 499 of them (Fig. 1). Patient data and the location of the thrombosis are presented in Table 1. 129 patients had died when this survey was conducted. Of the 370 who were still alive, 295 (80 %) answered the questionnaire.
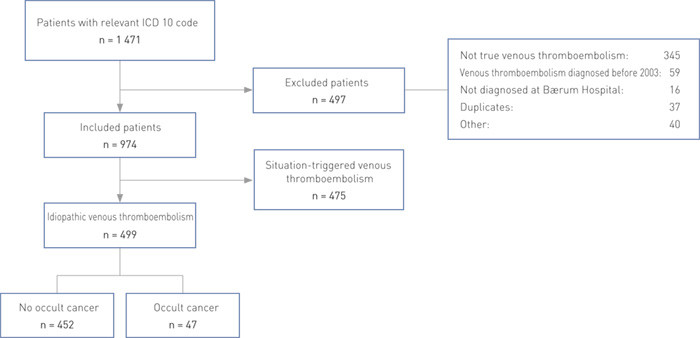
Table 1
Demographic data for patients with idiopathic venous thromboembolism diagnosed at Bærum Hospital in the period 1.1. 2003 – 31.12. 2008 (N = 499). No. (%) unless otherwise specified. Some patients have venous thromboembolism in several locations
| Demographic variables |
Data |
|
| Age (years), median (min–max) |
75 |
(20 – 97) |
| Gender |
||
| Female |
233 |
(47) |
| Deep vein thrombosis, leg |
244 |
(49) |
| Thigh |
175 |
(35) |
| Calf |
69 |
(14) |
| Right |
110 |
(45) |
| Left |
135 |
(55) |
| Pulmonary embolism |
274 |
(55) |
| Venous thromboembolism elsewhere |
17 |
(3.4) |
Of the 499 patients, 47 (9.4 %; 95 % CI 7.1 – 12 – 3) developed cancer during the first 12 months following detection of venous thromboembolism (Fig. 1). This is significantly higher than the annual incidence of cancer in the general population of Asker and Bærum during the same period (1.9 %; 95 % CI 0.8 – 3.5: p < 0.001). As regards the location of the malignancy, most organ systems were represented, with lungs (n = 10), prostate (n = 7), unknown origin (n = 7), pancreas (n = 5), colon/rectum (n = 3) and breast (n = 3) the most common.
A limited screening strategy, with further screening only if indicated by clinical or laboratory anomalies, resulted in negative findings in 72 % of the cases (Fig. 2). The remainder of the patients had positive results in one or more individual tests in the screening strategy.
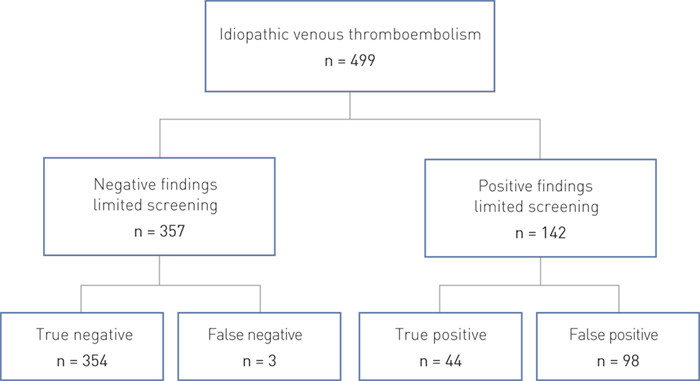
Limited screening yielded no findings in three of the patients in whom occult malignancy was later detected, while further screening found no malignancy in 98 of the patients who had tested positive in the limited screening. The limited screening strategy had a negative predictive value of 99 %. The screening strategy had a sensitivity of 94 % and a specificity of 78 %. There were no evident differences with respect to age, gender or location of the thrombosis between the groups in whom occult malignancy was or was not detected (Table 2).
Table 2
Comparison of patients with and without occult cancer after diagnosed idiopathic venous thromboembolism at Bærum Hospital in the period 1.1. 2003 – 1.12. 2008 (N = 499). No. (%) unless otherwise specified. Some patients have venous thromboembolism in several locations
| Patients with occult cancer (n = 47) |
Patients without occult cancer (n = 452) |
|||||
| Age (years), median (min–max) |
75 |
(23 – 91) |
75 |
(20 – 97) |
||
| Gender |
||||||
| Female |
18 |
(38) |
215 |
(48) |
||
| Male |
29 |
(62) |
237 |
(52) |
||
| Deep vein thrombosis |
24 |
(51) |
220 |
(49) |
||
| Thigh |
17 |
(36) |
158 |
(35) |
||
| Calf |
7 |
(15) |
62 |
(14) |
||
| Right |
11 |
(23) |
99 |
(22) |
||
| Left |
14 |
(30) |
121 |
(27) |
||
| Pulmonary embolism |
27 |
(57) |
247 |
(55) |
||
| Venous thromboembolism elsewhere |
3 |
(6) |
14 |
(3) |
||
Discussion
In this retrospective study, malignancy was detected in 9.4 % of the patients with idiopathic venous thromboembolism in the first year following thrombosis, compared with 1.9 % in the general population, taking account of age, gender distribution and geographical factors. There was no difference in age, gender or thrombosis location among those in whom occult malignancy was detected and those in whom it was not detected. The limited malignancy screening after detection of idiopathic venous thromboembolism which is used at Bærum Hospital had a high negative predictive value (99 %).
The incidence of occult malignancy in patients with idiopathic venous thromboembolism in our study was of the same order of size as that reported in international literature (3, 6) – (14, 20). As far as we are aware, similar surveys from Norway have not been published previously. In contrast to the findings of a number of other studies, we found no difference in age distribution between those who developed cancer during the first year and those who did not. This indicates that malignancy screening should not only be carried out for the over 60s age group, as some have proposed (6, 21, 22). As we did not find any differences between men and women either, both genders should be screened to the same extent.
Earlier studies have indicated that the location of the thrombosis has a bearing on the the probability of underlying malignancy (22, 23) and that certain types of cancer are more strongly associated with venous thromboembolism than others (4, 21, 24). We found no evidence of this in our material. The number of cases of occult malignancy in our study is so limited that we cannot attach great statistical weight to this, but since the cancer cases are so different, we cannot simply disregard any particular organs.
Extended screening strategy normally implies CT thorax and abdomen in addition to endoscopy. Ultrasound scanning of the abdomen, which was widely used earlier, has proved of limited use in malignancy screening (25). CT scanning of the thorax and abdomen is probably the most useful radiological examination. However, the radiation stress may be substantial, and the scan is resource-intensive for both the health service and the patient (25) – (27).
Thorough cost-benefit analyses of extended malignancy scanning are complicated. Few studies, not including ours, have been conducted to this end, and cost estimates appear to vary from one country to the next (25, 28). The largest and most recent study on the topic concluded that financial results cannot really be made the basis for a cost-benefit analysis (29). The costs are affected by the many false positive findings of extended screening that made further work-up necessary.
False positive findings were made in 98 of the patients in our study. The extra burden this implies should be in focus when determining the extent of the screening that should be chosen (30).
There is sound evidence to indicate that extensive screening for malignancy in cases of idiopathic venous thromboembolism would detect more cases of cancer earlier (6, 28). However, it is still unclear from relevant literature whether such screening has an acceptable cost-benefit ratio when the burden on the whole patient group and the health service is weighed up against the number of patients with an improved prognosis and reduced mortality.
Doubt has been expressed as to whether earlier detection of occult cancer actually yields any prognostic gain, because in most cases the cancer has already reached an advanced stage when idiopathic venous thromboembolism is identified (9, 31). In our study, occult cancer was detected in three patients after limited screening had not revealed signs of malignancy. We cannot exclude the possibility that early diagnosis could have been of significance to the prognosis of some of them, which could weigh in favour of more extensive screening of all patients. However, the large number who would have to be examined, and the radiation strain the whole group would be subjected to, weighs against extensive screening of all patients.
The only randomised study comparing extensive screening with no screening for occult malignancy showed that occult malignancy could be detected at an earlier stage, but provided no clear answer as to whether this was of significance for the prognosis (19). The study raised a number of ethical questions with respect to how the control group should be handled as regards information, and many of the patients in the control group were given more extensive screening than they should have been because some doctors believed it would be unethical not to do so. The study was also discontinued after five years, with 201 included patients of the planned total of 1 000. This means that the results are difficult to interpret.
The high incidence of occult cancer underscores the importance of having an effective malignancy screening plan. It is especially important these days, when more responsibilities are being transferred from the specialist health service to the primary health service as a result of the Coordination Reform, and when new oral anticoagulants are being introduced (9). Both these factors will lead to more outpatient treatment of patients with venous thromboembolism. Many will probably only have one outpatient visit to the hospital in the acute phase before being transferred to the primary health service for further follow-up. Tasks such as malignancy screening will therefore to a large degree have to be taken over by primary doctors.
Our study has its limitations. It was not conducted prospectively with a view to detecting occult cancer. However, screening of patients with idiopathic venous thromboembolism has been described in detail in procedures, and records of the time of diagnosis have been systematically kept by a competent group of doctors and nurses at the Thrombosis Clinic, Bærum Hospital.
We cannot exclude the possibility that those who did not answer the questionnaire on their state of health after having venous thromboembolism may have been found to have cancer without our learning of it. But patients in Asker and Bærum all have Bærum Hospital as their primary hospital, so that most cases of cancer would have been referred there. If there have been more cases of cancer than those we have registered, it means that the incidence of occult cancer is even higher than the 9.4 % we have found, and that the limited screening may have missed cases other than the three we have registered.
Conclusion
Almost 10 % of the patients with idiopathic venous thromboembolism had occult malignancy. Limited screening for underlying malignancy identified the great majority of these cases and appears to be an appropriate strategy for patients with idiopathic venous thromboembolism. Limited screening revealed anomalies in about 20 % of the patients without malignancy being detected.
The Coordination Reform and the introduction of new treatments for venous thromboembolism will mean that the main responsibility for following up these patients will be transferred to the primary health service. This makes it particularly important to plan wisely with respect to the extent of malignancy screening. Our study indicates that malignancy screening should be carried out irrespective of the patient's age and gender and the location of the thrombosis.
This article is based on a master’s thesis presented at the Institute of Health and Society, the Faculty of Medicine, University of Oslo (1).
MAIN POINTS
Occult cancer was found in about 10 % of patients with idiopathic venous thromboembolism
Limited screening revealed the great majority of cases of occult cancer
Patients with occult cancer did not differ from the other patients with respect to age, gender or the location of the thrombosis.
- 1.
Semb VB. Idiopatisk venøs trombose og underliggende malign sykdom. I hvilken grad bør det søkes etter okkult kreft? Oslo: Universitetet i Oslo, 2012: 70.
- 2.
Trousseau A. Lecture XCV – Phlegmatia alba dolens. London: The new Sydham Society, 1872.
- 10.
Enguídanos MJ, Todolí JA, Saro E et al. Usefulness of the tumor markers in the diagnosis of idiopathic deep venous thrombosis associated cancer. An Med Interna 2002; 19: 561 – 6. [PubMed]
- 11.
Cailleux N, Marie I, Primard E et al. Thrombophlebitis and cancer: evaluation of the diagnostic value of abdominal ultrasonography in the acute phase of a deep venous thrombosis. Report of 148 consecutive examinations. J Mal Vasc 1997; 22: 322 – 5. [PubMed]
- 13.
Monreal M, Lafoz E, Casals A et al. Occult cancer in patients with deep venous thrombosis. A systematic approach. Cancer 1991; 67: 541 – 5. [PubMed]
- 18.
Rosovsky R, Lee AY. Evidence-Based Mini-Review: Should all patients with idiopathic venous thromboembolic events be screened extensively for occult malignancy? Am Soc Hematol Educ Program 2010; 2010: 150 – 2.
- 22.
Trujillo-Santos J, Prandoni P, Rivron-Guillot K et al. Clinical outcome in patients with venous thromboembolism and hidden cancer: findings from the RIETE Registry. J Thromb Haemost 2008; 6: 251 – 5. [PubMed]
- 29.
Kleinjan A, van Doormaal FF, Prins MH et al. Limitations of screening for occult cancer in patients with idiopathic venous thromboembolism. Neth J Med 2012; 70: 311 – 7. [PubMed]
- 31.
Sørensen HT, Mellemkjær L, Steffensen HF et al. Incidens af cancer efter påvist primær dyb venetrombose eller lungeemboli. Läkartidningen 2000; 97: 1961 – 4.
Lungekreft er den kreftformen i Norge som tar flest liv (1). En vesentlig årsak til dette er at lungekreft oppdages så sent at to av tre pasienter er inkurable på diagnosetidspunktet. Man må anta at en del av disse kunne fått kurativ behandling om kreftsykdommen var blitt oppdaget tidligere.
I Tidsskriftet nr. 4/2014 viser Semb & Tveit at begrenset malignitetsutredning kan avdekke kreft hos ca. 10% i et pasientmateriale med idiopatisk venøs trombose (2). Hyppigst var lungekreft, som ble funnet hos 10 av 499 pasienter (2%). I artikkelen er det beskrevet tre pasienter som utviklet kreft det påfølgende året etter tromboseepisoden, uten at dette ble diagnostisert ved den initiale utredningen. Det oppgis ikke hvilke kreftformer dette var. Ved den begrensede utredningen ble det tatt røntgen thorax hos dem som ikke hadde fått utført CT thorax som en del av den primære utredningen.
Røntgen thorax har vesentlige svakheter som lav sensitivitet for påvisning av små svulster, men også hyppige falske positive funn. CT thorax har vesentlig høyere sensitivitet og spesifisitet, men er hittil brukt bare i begrenset grad i primærutredning fordi tilgjengeligheten har vært begrenset, undersøkelsen er ansett å være ressurskrevende, og det er betenkeligheter når det gjelder stråledosen undersøkelsen påfører pasienten.
Den teknologiske utviklingen har imidlertid medført at man nå kan utføre CT-undersøkelse av lungene med en stråledose som er marginalt høyere enn den for røntgen thorax. Pasientgruppen som artikkelen omhandler, har en median alder på 75 år og vil neppe utvikle strålerelatert kreftsykdom selv ved normaldose CT, som gir en effektiv dose på omkring 10 mSv (3). Risiko for stråleindusert kreft faller raskt med økende alder. Ifølge risikoestimatene til International Commission on Radiological Protection (ICRP) tilsvarer 10 mSv en livstidstilleggsrisiko for å utvikle kreft på 0,065% for pasienter i alderen 40-49 år, 0,042% for dem mellom 60-69 år og 0,01% for dem mellom 80-89 år (4).
Til forskjell fra normaldose CT vil lavdoseundersøkelsene gi dårlig fremstilling av mediastinum, og granskningen er derfor hovedsakelig begrenset til lungeparenkymet.
Utfordringen med CT thorax er at undersøkelsen er så følsom at det hos mange pasienter, særlig eldre, kan påvises små lesjoner som med høy sannsynlighet er benigne. Det er i oppfølgingen av funn som ikke er entydig benigne, merkostnaden ligger. Dette er prisen man må betale hvis man vil påvise lungekreft mens sykdommen fremdeles er kurabel. Samtidig er det grunn til å tro at økende volum av slike CT-undersøkelser vil gi økt kompetanse hos radiologene. Per i dag er det ingen annen metode enn CT som gir pålitelig fremstilling av lungekreft i tidlig stadium. Etter vår mening bør man vurdere å erstatte røntgen med CT ved thoraxundersøkelser av risikopopulasjoner som den som er presentert av Semb & Tveit.
Litteratur
1. Kreftregisteret. http://www.kreftregisteret.no/no/Registrene/Kreftstatistikk/ (17.3.2014).
2. Semb VB, Tveit A. Begrenset malignitetsutreedning ved idiopatisk venøs trombose. Tidsskr Nor Legeforen 2014; 134: 407-11.
3. Widmark A, Friberg EG. Veileder om representative doser for røntgenundersøkelser. Veileder til forskrift om strålevern og bruk av stråling. Veileder nr. 5b. Østerås: Statens strålevern, 2007.
4. Balonov MJ, Shrimpton PC. Effective dose and risks from medical x-ray procedures. Elsevier Ltd, ICRP 2012