Warfarin therapy for atrial fibrillation in general practice – is bleeding risk underestimated?
Atrial fibrillation occurs in approximately 10 % of the population aged 75 and above, and the incidence increases with age. The condition gives rise to a fivefold increase in risk for thromboembolic stroke (1). There appears to be the same risk from paroxysmal atrial fibrillation as from permanent atrial fibrillation (2, 3).
There are large individual variations in thromboembolism risk and it is recommended to conduct a risk assessment for each patient, using the CHA₂DS₂-VASc score (4, 5) (Table 1). Olesen et al. published a Danish registry-based cohort study in 2011 which used the CHA₂DS₂-VASc score (6). This study demonstrated that the annual thromboembolism risk in patients with untreated atrial fibrillation varied between 0.8 % and 23.6 %, depending on the risk score (Table 2).
Table 1
CHA₂DS₂-VASc score (Congestive Heart Failure History, Hypertension History, Age > 75, Diabetes Mellitus, Stroke/TIA History, Vascular Disease History, Age 65 – 74, Sex category) for calculating risk for thromboembolic stroke in patients with atrial fibrillation. The different conditions included in the score count for 1 or 2 points and give a total score of 0 – 9 (5)
| Risk factor |
Points |
| Heart failure |
1 |
| Hypertension |
1 |
| Age > 75 years |
2 |
| Diabetes mellitus |
1 |
| Prior stroke |
2 |
| Other vascular disease |
1 |
| Age 65 – 74 |
1 |
| Female |
1 |
Table 2
Annual absolute risk for thromboembolic stroke in patients with untreated atrial fibrillation at different CHA₂DS₂-VASc scores based on figures from the Danish registry (6) and calculated annual absolute risk and absolute risk reduction with warfarin therapy based on a relative risk reduction of 64 % (7)
| CHA₂DS₂-VASc score |
Annual absolute risk untreated (%) |
Annual absolute risk with warfarin therapy (%) |
Absolute risk reduction with warfarin therapy (%) |
| 0 |
0.8 |
0.3 |
0.5 |
| 1 |
2.0 |
0.7 |
1.3 |
| 2 |
3.7 |
1.3 |
2.4 |
| 3 |
5.9 |
2.1 |
3.8 |
| 4 |
9.3 |
3.4 |
5.9 |
| 5 |
15.3 |
5.5 |
9.8 |
| 6 |
19.7 |
7.1 |
12.6 |
| 7 |
21.5 |
7.7 |
13.8 |
| 8 |
22.4 |
8.1 |
14.3 |
| 9 |
23.6 |
8.5 |
15.1 |
In a meta-analysis, anticoagulant therapy with warfarin was calculated to give a relative decrease in risk of 64 % (95 % CI 49 – 74 %) for stroke (7). Novel oral anticoagulants (NOACs), direct thrombin inhibitors and factor Xa inhibitors have been compared with warfarin in a number of large- scale studies (8) – (10) and have become useful alternatives. The most recent guidelines issued by the European Society of Cardiology (ESC) (3) recommend the NOACs as first-line drugs, give little place for acetylsalicylic acid, and have increased the recommendation for anticoagulants for patients with lower thromboembolism risk (CHA₂DS₂-VASc score ≥ 1).
Anticoagulant therapy carries an increased risk of bleeding. Major bleeding is defined as a fall in haemoglobin levels to > 2 g/dl, the need for transfusion, or intracranial haemorrhage. The Swedish national quality registry for anticoagulant therapy, AURICULA, found that the annual risk for major bleeding was 2.6 % (11), while in a Norwegian general practice it was 2.4 % (12). These figures apply to warfarin therapy for all indications (atrial fibrillation, pulmonary embolism, venous thrombosis and heart valve repair surgery). In the recent atrial fibrillation studies (8) – (10) the risk for major bleeding with warfarin therapy was higher (3.1 – 3.4 %).
A method has also been developed for calculating individual risk for major bleeding, the HAS-BLED score (Table 3), which is recommended by the European Society of Cardiology (3) and by Norwegian specialists (4). The method has been validated in two studies, one with low and one with high bleeding risk (13, 14) (Table 4). This allows the advantages of anticoagulants to be compared with the disadvantages of an increase in bleeding risk for the individual patient.
Table 3
HAS-BLED score (Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol Concomitantly) for calculating risk for major bleeding in patients with atrial fibrillation receiving warfarin therapy. The different conditions included in the score count for 1 point and give a total score of 0 – 9 (13)
| Risk factor |
Points |
| Systolic blood pressure >160 mm Hg |
1 |
| Renal failure (creatinine > 200 μmol/l) |
1 |
| Liver disease (ALT > 3 × limit value) |
1 |
| Prior stroke |
1 |
| Prior major bleeding |
1 |
| Labile INR (< 60 % of the values in therapeutic range) |
1 |
| Age > 65 years |
1 |
| Use of NSAIDs or blood platelet inhibitors |
1 |
| High alcohol consumption |
1 |
Table 4
Annual absolute risk for major bleeding during warfarin therapy in patients with atrial fibrillation with different HAS-BLED scores calculated in the Euro Heart Survey (13) and SPORTIF studies (14)
| Annual absolute risk for major bleeding ( %) |
||
| HAS-BLED score |
Euro Heart Survey |
SPORTIF |
| 0 |
1.1 |
0.9 |
| 1 |
1.0 |
3.4 |
| 2 |
1.9 |
4.1 |
| 3 |
3.7 |
5.8 |
| 4 |
8.7 |
8.9 |
| 5¹ |
12.5 |
9.1 |
| [i] | ||
[i] ¹ None of the studies had patients with HAS-BLED score higher than 5
Warfarin therapy in Norway has largely been carried out by general practitioners who have amassed considerable experience (12) and with a good standard of quality assurance (15). The purpose of the present study is to study warfarin therapy in patients with atrial fibrillation in a general practice by looking at target range attainment for INR values, mapping the therapy at different levels of thromboembolism risk, and calculating the benefit of warfarin therapy by also calculating the bleeding risk.
Material and method
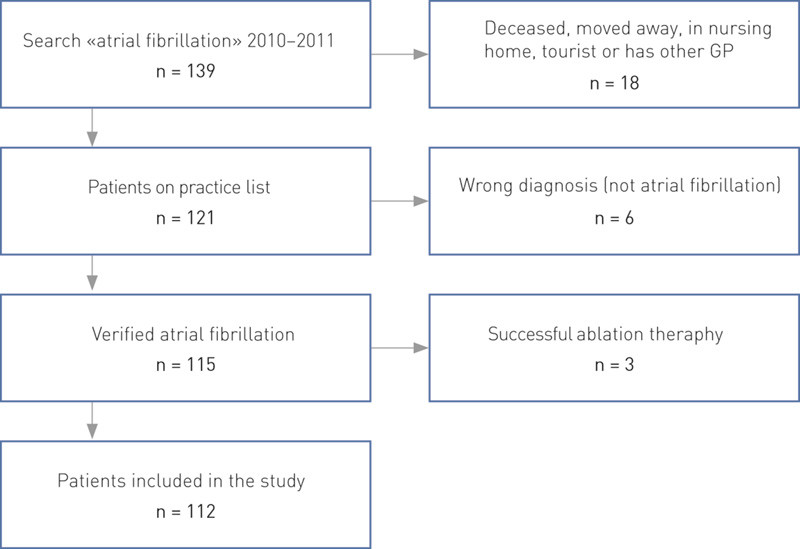
The general practice group Legegruppen Grandgården in Arendal, Norway has six general practitioners. We searched in the data records on the diagnosis «atrial fibrillation» for the years 2010 and 2011, and included in the study all the patients on the practice list who had the diagnosis atrial fibrillation in November 2011 (Fig. 1). We included patients with paroxysmal atrial fibrillation even if they had sinus rhythm at that point in time. Patients who had undergone successful surgery with ablation therapy were not included in the study. We reviewed the records and entered the following variables: Age, gender, type and duration of atrial fibrillation, any ablation therapy, anticoagulant therapy, its duration and the reason for any seponation, all INR measurements for one year (November 2010–November 2011) and all data necessary to calculate CHA₂DS₂-VASc scores and HAS-BLED scores (Tables 1, 3).
Most of the data were found in the patients’ summary care records, laboratory test records and regular medication records, but we also searched in the entire text of the patient records and in some discharge notes. The diagnosis hypertension was recorded when the patient used antihypertensives beyond betablockers, which most had been prescribed for frequency regulation, but not if the medication was part of heart failure treatment. Poor blood pressure control was noted if at least one of the last three measurements taken showed systolic blood pressure of ≥ 160 mm Hg. Diabetes was recorded, irrespective of treatment.
We calculated the CHA₂DS₂-VASc score and absolute risk for thromboembolism for each patient individually according to the most recent data from the study by Olesen et al. (6). To calculate the bleeding risk utilising the HAS-BLED method, we used two alternatives. Alternative 1 is based on the Euro Heart Survey study (13), in which an annual major bleeding rate was found in 1.5 % of the participants. Alternative 2 is based on the SPORTIF studies (14), in which the annual major bleeding rate was 3.7 %. With an expected 64 % relative risk reduction with warfarin therapy (7) we were able to calculate the benefit of therapy thus: Net benefit of warfarin therapy = Reduction of thromboembolism risk (risk for thromboembolism × 0.64) – Risk for major bleeding. For example, a male atrial fibrillation patient aged 70 with hypertension will have a CHA₂DS₂-VASc score of 2 (Table 1) and an annual risk for thromboembolism of 3.7 % (Table 2). If he has a HAS-BLED score of 1 (Table 3), he has an annual risk for major bleeding of 1.0 % (alternative 1) or 3.4 % (alternative 2). The calculated net benefit of warfarin therapy using alternative 1 will thus be (3.7 × 0.64) – 1.0 = 1.4, that is, a positive net benefit. Using alternative 2 the calculation is (3.7 × 0.64) – 3.4 = –1.0, that is, a negative net benefit.
All the data were entered in the statistics analysis program NSDstat. The median value was used to calculate central values in the data set.
The study has been submitted to the Regional Committee for Medical and Health Research Ethics, which has recommended that it be carried out, although the Committee is evaluating it as a quality assurance project. The study has been accepted by the Norwegian Social Science Data Services (NSD).
Results
We identified a total of 139 patients with a recorded diagnosis of atrial fibrillation in the period 2010 – 2011. 112 patients with verified atrial fibrillation in November 2011 were included in the study (Fig. 1). On 1 December 2011 we had 5,962 patients on the practice list, giving a prevalence of diagnosed atrial fibrillation of 1.9 %. The median age was 79.0 years with an age range of 17 – 94 years, and 60 % were men. 76 patients had permanent atrial fibrillation, 25 had paroxysmal atrial fibrillation and 11 had persistent atrial fibrillation.
Anticoagulant therapy
82 patients (73 %) used warfarin, four of them in combination with platelet inhibitors. 16 patients (14 %) used only acetylsalicylic acid or another platelet inhibitor, two used new oral anticoagulants and 12 patients (10 %) used no anticoagulant therapy. 81 of the warfarin users attended check-ups at the surgery with a total of 751 INR measurements taken over the course of one year, with a median value of 9.3 (range 5 – 16) measurements per patient per year. That gives an interval of 5.6 weeks between each test. Of the INR measurements 69 % were within the target range, 14 % below and 17 % above. The target range was INR 2.0 – 3.0, apart from in the case of five patients who had undergone heart valve repair surgery, who had an INR target range of 2.5 – 3.5. The period for ongoing anticoagulant therapy was 0 – 24 years, the median value 5.0 years. The duration of atrial fibrillation was 0 – 25 years, the median value 5.7 years.
30 patients (27 %) did not use warfarin, and of these 11 (10 %) had never used it. 19 patients (17 %) had used warfarin previously, seven of whom had discontinued because of bleeding risk and 12 because of low thromboembolism risk.
Thromboembolism risk and bleeding risk
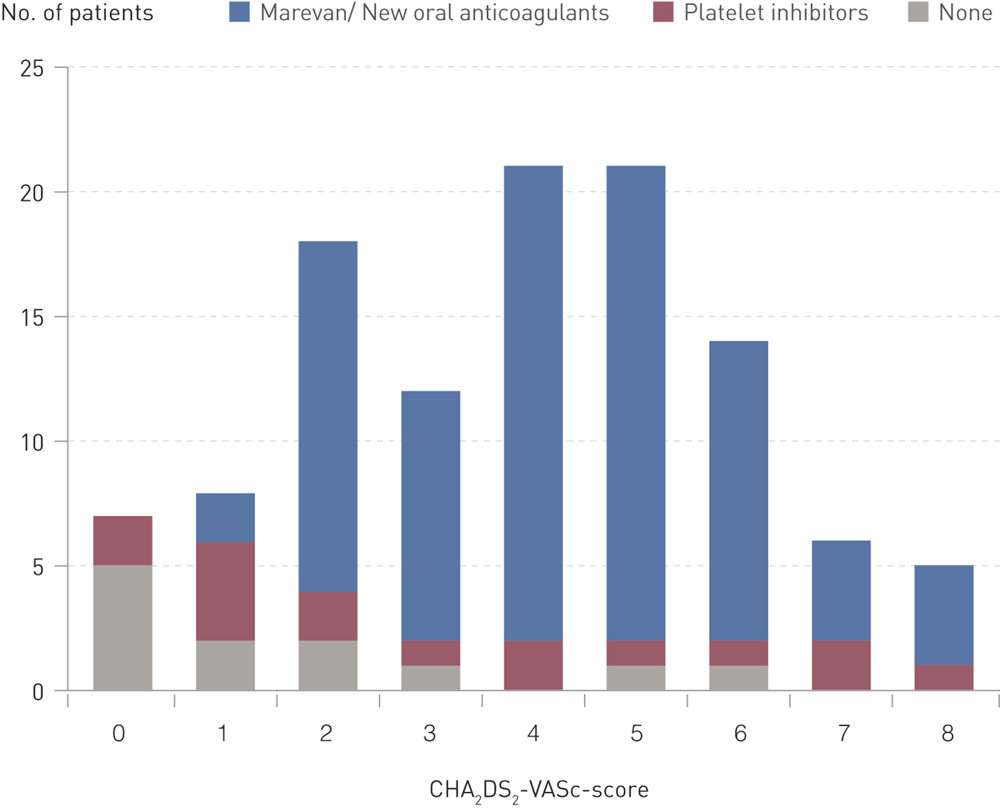
The median value for the patient group’s CHA₂DS₂-VASc score was 4.0 (range 0 – 8). The patients were divided up into risk groups according to the CHA₂DS₂-VASc score, with an overview of anticoagulant therapy in each group (Fig. 2). 15 patients scored ≤ 1. Of these, two (13 %) used warfarin, six (40 %) used acetylsalicylic acid and seven (47 %) had no treatment. Of 97 patients with a CHA₂DS₂-VASc score of ≥ 2, 82 (85 %) used warfarin or new oral anticoagulants, ten (10 %) used blood platelet inhibitors and five (5 %) had no treatment. 88 patients (79 %) had a HAS-BLED score of 0 – 2, while 24 (21 %) had a HAS-BLED score of ≥ 3.
Benefits of treatment
Benefits of treatment were calculated according to the two HAS-BLED studies, using both the study population from EHS (alternative 1) and from SPORTIF (alternative 2). With alternative 1, 102 patients (91 %) were calculated to have benefit of anticoagulant therapy, while with alternative 2, 74 patients (66 %) would have benefit. We found no treatment benefit for patients with a CHA₂DS₂-VASc score of 0 (Table 5). With scores of 1 and 2 there were large differences between the two calculation methods. Using the EHS study population we calculated benefit for 75 % and 94 % of our patients respectively, but using the SPORTIF study population the percentage of patients with benefit were 12 % and 6 % respectively. With a CHA₂DS₂-VASc score of 3, there was better conformity between the calculation methods, and at a score of ≥ 4, both calculation methods showed that all our patients would have considerable benefit of warfarin therapy.
Table 5
Number of 112 patients with atrial fibrillation at Legegruppen Grandgården in the period 2010 – 11 who are calculated to have net benefit of warfarin therapy based on two different studies
| CHA₂DS₂-VASc score |
Number of patients (n = 112) |
Patients with calculated net benefit of warfarin therapy¹ |
||||||
| Based on EHS study |
Based on SPORTIF study |
|||||||
| Number |
Percentage (95 % CI) |
Number |
Percentage (95 % CI) |
|||||
| 0 |
7 |
0 |
0 |
(0 – 41) |
0 |
0 |
(0 – 41) |
|
| 1 |
8 |
6 |
75 |
(35 – 97) |
1 |
12 |
(3 – 53) |
|
| 2 |
18 |
17 |
94 |
(72 – 100) |
1 |
6 |
(0 – 27) |
|
| 3 |
12 |
12 |
100 |
(74 – 100) |
5 |
42 |
(15 – 72) |
|
| 4 – 8 |
67 |
67 |
100 |
(95 – 100) |
67 |
100 |
(95 – 100) |
|
| [i] | ||||||||
[i] ¹ Thromboembolic absolute risk reduction is greater than absolute risk for major bleeding. Risk for thromboembolic stroke is calculated from the Danish registry (6). Relative risk reduction with warfarin therapy is set at 64 % (7). Risk for major bleeding is calculated using HAS-BLED score with two alternative studies, Euro Heart Survey (EHS) (13) and SPORTIF (14)
Discussion
85 % of our patients with a CHA₂DS₂-VASc score of ≥ 2 and 75 % of all of our patients with atrial fibrillation used warfarin or new oral anticoagulants. 69 % of the INR measurements were within the therapeutic range.
The patients with high thromboembolism risk had a high calculated benefit of warfarin therapy, also if there was high bleeding risk. In the patients with low thromboembolism risk the calculated absolute risk reduction was so small that the risk for bleeding counterbalanced the benefits.
The percentage of warfarin users among patients with atrial fibrillation in the European Society of Cardiology (ESC) member countries in 2003 was 65 % (16), the same figure as in 2013 (17). In our general practice in 2006, 57 % of INR values were within the target range (12). In the three large-scale atrial fibrillation studies that formed the basis for the introduction of new oral anticoagulants, 64 % (8), 55 % (9) and 66 % (10) of the measurements were in the therapeutic range, and in the Swedish registry AURICULA this figure was 76 % (11). The high percentage of warfarin users and improvement of the INR values in Norwegian general practice may be explained by the long experience with warfarin therapy in Norway (12) and the high standard of Norwegian quality assurance (15).
The calculated benefit of the treatment depended very much on the calculation method used. Using the population in the EHS study with a low bleeding risk demonstrated that almost all the patients with a CHA₂DS₂-VASc score of > 0 had benefit of warfarin therapy, as the European Society of Cardiology notes (3). Using the population in the SPORTIF study with a high bleeding risk demonstrated that there was little benefit at a CHA₂DS₂-VASc score of < 3. The choice of study population is therefore an important premise for the result.
The Euro Heart Survey on Atrial Fibrillation described «real-life» treatment of atrial fibrillation in 5,333 patients from different types of cardiology practices in 35 European Society of Cardiology (ESC) member countries (16). The survey formed the starting point for constructing the risk scores CHA₂DS₂-VASc score (5) and HAS-BLED score (13). The EHS study employed only a one-year observational period, the patients had a low average age (66 years) and only 65 % used warfarin. The entire patient material in the study was used to develop the HAS-BLED score (including the 35 % of patients who did not receive anticoagulant therapy). The incidence rate for major bleeding was therefore only 1.5 %. For that reason, the absolute risk values for major bleeding are low (Table 4). Not until a HAS-BLED score of 3 is reached does the absolute risk for major bleeding exceed 3 %. The European Society of Cardiology bases its guidelines on this study when they recommend the use of the HAS-BLED method and say that a HAS-BLED score of 3 is the limit that indicates bleeding risk (3, 4). The authors of this first HAS-BLED study believed there could be an underestimation of bleeding risk and that more studies were needed (13). This was done when the HAS-BLED score was applied to the studies SPORTIF₃ and SPORTIF₅, randomised atrial fibrillation studies in which all the participants received anticoagulant therapy (14). Here, the average risk for major bleeding was 3 %, about the same as in the other more recent atrial fibrillation studies (8) – (10), and already at a HAS-BLED score of 1 the 3 % absolute risk for major bleeding is exceeded (Table 4).
In our own 2006 study (12) of warfarin therapy in general practice, the annual risk for major bleeding was 2.4 %, but for the sub-group with atrial fibrillation the risk was 3.7 %. We believe that the calculations carried out on the basis of the SPORTIF studies with an approximately 3 % annual risk for major bleeding give a result closer to the actual risk for our atrial fibrillation patients than the EHS study does.
We have presumed that warfarin gives a 64 % reduction of thromboembolism risk, based on a meta-study (7) which builds on studies carried out many years ago. This may mean that the effect has been overestimated, since there is now better treatment available for hypertension and other risk factors (18).
Our method entailing retrospective examination of patient records has its weaknesses. Greatest reliability is achieved from recording «hard» data such as gender, age, diagnosis, medication and laboratory test results, while case histories may be incomplete. There may be under-reporting of data, particularly with a view to factors for bleeding risk (alcohol consumption, over-the-counter purchases of non-steroidal anti-inflammatories (NSAIDs) and so on). On the other hand, it is a strength that this is a report coming from an ordinary Norwegian general practice.
The «accounts» we set up to calculate the net benefit of anticoagulant therapy required us to assess a major bleeding episode as equal in seriousness to a thromboembolic stroke. This is debatable. The outcome of both conditions can vary from death to moderate illness without sequelae. Our analysis of «profit» and «loss» is nevertheless a reminder that we must not do more harm than good to the patient with our treatment.
In 2011, only two of our patients used a new oral anticoagulant (dabigatran), although the use of these drugs is expected to increase substantially (19). The new oral anticoagulants require no INR control, but nevertheless careful monitoring of the risk for bleeding (20). The Norwegian Knowledge Centre for the Health Services has stated that the knowledge base for these drugs is somewhat weak and that their place in the treatment is still unclarified (21).
We find little benefit of warfarin therapy for patients with low thromboembolism risk. A similar risk-benefit assessment was carried out in a study of the American ATRIA population in which more than 13,000 patients with atrial fibrillation were followed for seven years (18). The study was retrospective, the calculation method different, and intracranial haemorrhage was used instead of the HAS-BLED score, but the conclusion is the same, that there is little benefit of warfarin therapy for patients with low thromboembolism risk. The European Society of Cardiology’s guidelines recommend anticoagulant therapy for patients with a CHA₂DS₂-VASc score of ≥ 1 (with the exception of women < 65 years without other risk factors). Could this recommendation be based on too low an estimate of bleeding risk? Using the SPORTIF study, we do not get into credit in our «accounts» until a CHA₂DS₂-VASc score of 3. Our material is small and our method has weaknesses, but the results indicate that prospective, controlled studies should be conducted to determine whether anticoagulant therapy for atrial fibrillation provides an overall benefit for patients with low thromboembolism risk.
We should like to thank Christian Borchgrevink, Harald Reiso and the other members of the research group of general practitioners from Agder for their inspirational assistance. This study was funded by a grant from the Norwegian Committee on Research in General Practice.
MAIN POINTS
In a Norwegian general practice, 85 % of the patients with atrial fibrillation and with thromboembolism risk corresponding to a CHA₂DS₂-VASc score of ≥ 2 received effective anticoagulant therapy
For patients with high thromboembolism risk there was a high calculated benefit of warfarin therapy
For patients with low/moderate thromboembolism risk the calculated benefit of therapy was dependent on the calculation method
Applying European guidelines for anticoagulant therapy to our group of patients demonstrated that bleeding risk appears to be underestimated
- 2.
Risøe C, Gjesdal K. Antikoagulasjonsbehandling ved forbigående atrieflimmer. Tidsskr Nor Lægeforen 2004; 124: 950 – 2. [PubMed]
- 3.
Camm AJ, Lip GY, Caterina R et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation. Eur Heart J 2012; 33: 2719 – 47. [PubMed]
- 4.
Aamodt AH, Sandset PM, Atar D et al. Atrieflimmer og hjerneslag. Tidsskr Nor Legeforen 2013; 133: 1453 – 7. [PubMed]
- 6.
Olesen JB, Lip GY, Hansen ML et al. Validation of risk stratification schemes for predicting stroke and thromboembolism in patients with atrial fibrillation: nationwide cohort study. BMJ 2011; 342: 1 – 9. [CrossRef]
- 12.
Bratland B. Warfarinbehandling i allmennpraksis. Tidsskr Nor Lægeforen 2006; 126: 162 – 5. [PubMed]
- 14.
Lip GY, Frison L, Halperin JL et al. Comparative validation of a novel risk score for predicting bleeding risk in anticoagulated patients with atrial fibrillation: the HAS-BLED (Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol Concomitantly) score. J Am Coll Cardiol 2011; 57: 173 – 80. [PubMed] [CrossRef]
- 15.
Reikvam Å, Sandset PM, red. Warfarinbehandling i praksis – tryggere antikoagulasjon. 2. utg. Skriftserie for leger: Utdanning og kvalitetsutvikling. Oslo: Den norske legeforening, 2010. http://legeforeningen.no/PageFiles/25973/Warfarinbehandling%20i%20praksis.pdf (6.12.2013).
- 17.
Ingen bedring i slagforebygging. Pressemelding fra ESC 2013. Dagens Medisin 4.9.2013.
- 19.
Nasjonal rådgivende spesialistgruppe innen antikoagulasjon. Informasjon om de nye perorale antikoagulasjonsmidlene dabigatran, rivaroksaban og apiksaban. Oslo: Helsedirektoratet, Statens legemiddelverk, 2013.
- 20.
Ghanima W, Atar D, Sandset PM. Nye perorale antikoagulasjonsmidler – en oversikt. Tidsskr Nor Legeforen 2013; 133: 1940 – 5. [PubMed]
- 21.
Efficacy and cost-effectiveness of new oral anticoagulants compared to warfarin for the prevention of stroke in patients with atrial fibrillation. Oslo: Nasjonalt kunnskapssenter for helsetjenesten, 2013. www.kunnskapssenteret.no/publikasjoner/efficacy-and-cost-effectiveness-of-new-oral-anticoagulants-compared-to-warfarin-for-the-prevention-of-stroke-in-patients-with-atrial-fibrillation?language=english (6.12.2013).