Antithrombotic treatment and/or transcatheter closure may prevent recurrent cerebrovascular episodes in patients with patent (open) foramen ovale and transitory ischaemic attacks (TIA)/ embolic stroke. The Norwegian guidelines for transcatheter closure are based on systematic reviews (1), and the practice of prophylactic treatment varies somewhat.
We present here a young patient with embolic stroke and patent foramen ovale. We then discuss work-up and treatment generally on the basis of updated international literature, including recent results from the first randomised studies in the field: the CLOSURE study (Closure of patent foramen ovale versus medical therapy after cryptogenic stroke) (2), the RESPECT study (Randomized evaluation of recurrent stroke comparing PFO closure to established current standard of care treatment) (3) and the PC trial (Clinical trial comparing percutaneous closure of patent foramen ovale using the Amplatzer PFO Occluder with medical treatment in patients with cryptogenic embolism) (4).
Method
The article is based on a review of relevant Norwegian and English-language literature found by searching PubMed. We used thesearch terms «patent foramen ovale» combined with each of the search terms «review», «transitory ischemic attack», «stroke», «closure» and «treatment», limited to the period 1982 – 1 April 2013.
Original articles, review articles and case reports were evaluated. We emphasise the results of three randomised studies: CLOSURE, RESPECT and PC.
Case report
A man in his twenties had recurrent attacks of migraine with aura, and the last attack occurred six weeks previously. He was a non-smoker and used no medicines. On the day of admission he awoke with a migraine-like headache. Three hours later, just after completing press-ups, he developed numbness in the left arm, paresis in the left side of his body, and dysarthria. A vascular event in the right hemisphere was suspected.
Cerebral computer tomography (CT) performed at the local hospital revealed «hyperdense right middle cerebral artery sign» and cerebral CT angiography revealed an embolic occlusion at the bifurcation of the right middle cerebral artery. A cerebral CT the following day documented infarction of the basal ganglia in the right hemisphere, and a CT angiogram showed complete recanalisation of the right middle cerebral artery. Transoesophageal echocardiography (TEE) with saline contrast two days after admission showed patent foramen ovale with a diameter of 4 mm (Fig. 1).
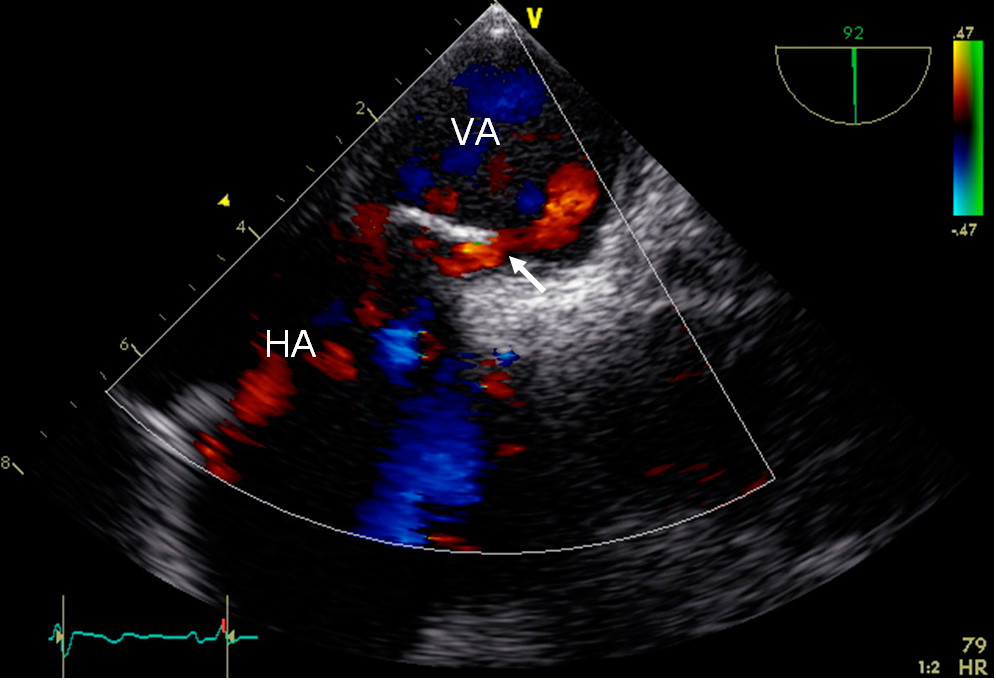
The patient was discharged without sequelae with 75 mg acetylsalicylic acid daily. He was referred to a cardiologist for assessment of transcatheter closure of the patent foramen ovale.
Pathophysiology
Patent foramen ovale is a persistent fetal communication between the right and left atrium due to incomplete closure of the atrial septum. If the right atrial pressure exceeds the pressure in the left atrium, for example in connection with physical exertion or a Valsalva-inducing activity (5), blood will be from the right to the left atrium. Patent foramen ovale is the cause of 95 % of right-to-left shunting in adults (6).
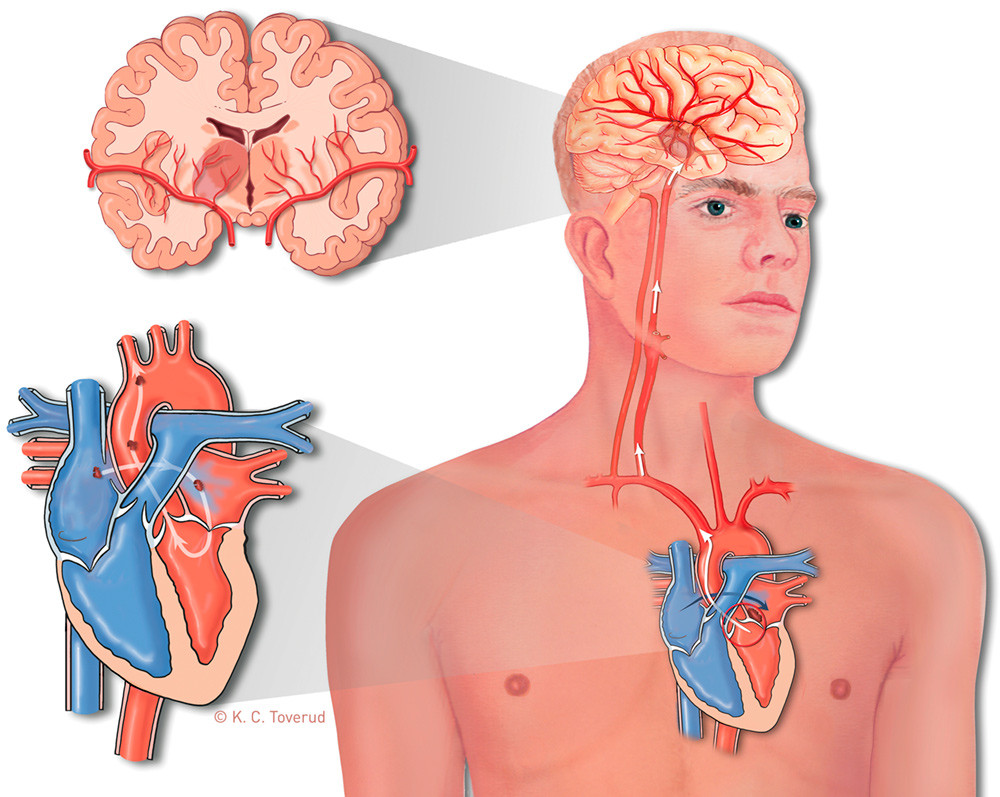
Emboli that pass from the right to the left side as a result of an intracardial shunt are classified as paradoxical (7). Paradoxical embolisation to the brain occurs in cases of patent foramen ovale (Fig. 2) (8).
Prevalence
Patent foramen ovale is common in the general population. Autopsy-based studies show a prevalence of 27.3 % (9). TIA and ischaemic stroke of undetermined cause are classified as cryptogenic. The proportion of cryptogenic strokes is lower among older patients, and estimated at 40 % of the general stroke population (10). In a population-based study from Hordaland, cryptogenic strokes were found in 50.7 % of all patients under the age of 40, despite comprehensive vascular assessment (11).
The results of previous research on the relationship between patent foramen ovale and cryptogenic stroke are conflicting. For example, a meta-analysis of case-control studies confirmed an association between patent foramen ovale and cryptogenic strokes in young patients (age < 50 years) compared with controls (odds ratio (OR) 5.0; 95 % confidence interval (CI) 3.2 – 7.7) (12). This meta-analysis involved studies of different size, and echocardiography was performed on different indications. These factors may have influenced the selection of study participants, and the results must therefore be interpreted with caution.
On the other hand, in one case-control study, patent foramen ovale was found not to be associated with a higher risk of stroke (OR 1.1; 95 % CI 0.6 – 1.9) (13). However, there are several case reports on cerebrovascular accidents where echocardiography has revealed an embolus trapped in the foramen ovale (14) – (16).
Decompression sickness in divers and migraine with aura appear to be associated with patent foramen ovale (17, 18). However, it has now been well documented that migraine with aura is not an indication for closure (19).
Although some studies have shown that a large opening and/or co-existing atrial septal aneurysm increase the risk of stroke (20, 21) other studies have failed to verify this (2, 22) – (24).
Patient evaluation
TEE with saline contrast is the gold standard for diagnosing right-to-left shunt (25, 26). This technique enables visualisation of the patent foramen ovale and diagnosis of the shunt by detection of microbubbles (during a Valsalva manoeuvre) in the left atrium, within three to four cardiac cycles following venous injection of saline (17, 27).
In professional circles, it is usual to assume that there is an etiological correlation between patent foramen ovale and cryptogenic strokes in young patients (age < 50 years) with a positive TEE and an embolic (and not thrombotic) stroke on cerebral MRI and with no other apparent etiology for stroke [19]. Factors supporting such a relationship are the presence of a Valsalva-inducing activity at the symptom onset, detected deep venous thrombosis and/or thrombophilia.
One study reported sensitivity and specificity for patent foramen ovale detection of 100 % and 96 %, respectively, for TEE, and 94 % and 96 % for transthoracic echocardiography (TTE) (28). TEE provides more information about the anatomical details than TTE. However, a Valsalva manoeuvre may be difficult to carry out with TEE because of sedation and the use of a monitor, and TTE may then be a better method.
An alternative method for detecting a right-to-left shunt is transcranial Doppler with intravenous saline contrast and detection of microbubbles in cerebral circulation (7). The method is more sensitive than TEE for detecting small shunts and is useful in the primary assessment. Negative transcranial Doppler rules out a right-to-left shunt.
A D-dimer test on suspicion of deep venous thrombosis has a high sensitivity (90 – 99 %) but low specificity (29). In the PELVIS study (Paradoxical emboli from large veins in ischemic stroke) a higher prevalence of pelvic vein thrombosis was found with MR venography in cases of cryptogenic stroke (20 %) than with ischaemic strokes of known etiology (4 %) (p < 0.025) (30).
In cases of cryptogenic stroke we recommend ultrasound evaluation of the lower limb veins as a matter of routine, with deep venous thrombosis in mind. At the same time, CT or MR venography should be performed to provide an overview of pelvic veins and the inferior vena cava. Tests of thrombocoagulation system function should include activated protein C resistance, antithrombin, protein C and protein S deficiency, lupus anti-coagulant, anti-cardiolipin antibody IgG, anti-cardiolipin antibody IgM and prothrombin gene mutation.
Treatment
According to our national recommendations, secondary prophylaxis for patients with ischaemic strokes assumed to be due to patent foramen ovale should consist of acetylsalicylic acid and dipyridamole/clopidogrel rather than anticoagulation (26).
Transcatheter closure may be considered for recurrent stroke where no other probable cause can be found (26). The American Heart Association and American College of Cardiology have the same recommendation (31). However, the scientific documentation is inadequate (31, 32).
Medicinal treatment
Antithrombotic therapy consists of either platelet inhibition or anticoagulation. The efficacy of acetylsalicylic acid was suggested by a French multi-centre trial, which found that the incidence of recurrent stroke after four years of follow-up of 216 patients with cryptogenic stroke and patent foramen ovale was lower (risk 2.3 %; 94 % CI 0.3 – 4.3) than in stroke patients without an intracardiac shunt (risk 4.2 %; 95 % CI 1.8 – 6.6 %) (21). Both groups were given acetylsalicylic acid (300 mg daily) as secondary prophylaxis. The incidence of recurrent stroke in the same period was four times as high in 51 of the patients with co-existing atrial septal aneurysm (risk 15.2 %; 95 % CI 1.8 – 28.6). The conclusion of the study was that warfarin should be considered for this subgroup.
The PFO in cryptogenic stroke study (PICSS) , on the other hand, evaluated acute stroke patients without and with patent foramen ovale assigned to acetylsalicylic acid (325 mg daily) or warfarin (with INR 1.4 – 2.8) (22). There was no significant difference with respect to two-year risk of recurrent stroke or death between the groups, or for patients with co-existing atrial septal aneurysm.
Surgical intervention
Surgical intervention requires a median sternotomy, treatment with a heart-lung machine, one to two weeks' rehabilitation in hospital and four to eight weeks of sick leave (33). The intervention is not a relevant option since the introduction of transcatheter closure (34).
Transcatheter closure
A systematic literature review of 20 studies to examine the efficacy of transcatheter closure compared to antithrombotic therapy showed that the annual risk of recurrent TIA or stroke was lower in the group with closure (1.3 %; 95 % CI 1.0 – 1.8) than in patients who received antithrombotic therapy (5.2 %; 95 % CI 4.4 – 6.2) (35). This applied in particular to patients with co-existing atrioseptal aneurysm.
Other studies have not shown any risk reduction with closure compared to antithrombotic treatment alone (2) – (4, 36). In a Norwegian study of 15 patients who underwent transcatheter closure, no recurrent cerebrovascular episodes occurred after a median follow-up period of nine months (33).
There are only three randomised studies of secondary prophylaxis for patients with TIA or ischaemic stroke and patent foramen ovale – CLOSURE (2), RESPECT (3) and PC (4).
In CLOSURE, 909 patients aged 18 – 60 were randomised (2). There was no significant difference in the incidence of the primary endpoint, which was a composite of recurrent stroke, TIA or death, after two years. The incidence was 5.5 % in patients with closure (n = 447) and 6.8 % in patients who received antithrombotic therapy (n = 462) (p = 0.37). In both groups, the two-year risk of recurrent stroke was lower than expected (almost 3 %) and in 81 % of the cases due to an etiology other than patent foramen ovale. Conversely, the proportion of TIAs (3.1 % and 4.1 %, respectively) was higher than expected. The results may indicate that the first cerebrovascular episode was not due to patent foramen ovale and that the underlying cause may have been overlooked.
The most common complications of transcatheter closure are dislocation of the closure device, groin haematoma, arteriovenous fistula, thrombus formation in the closure device, pulmonary embolism, haemorrhage, arrhythmia and cardiac tamponade (1). In CLOSURE, catheter treatment increased the absolute risk of serious vascular events by 3.2 % and of induced atrial fibrillation in 5.7 %. One objection to the high complication rates has been that several of the treatment centres involved had too little training and routine in the catheter technique. None of the patients died of neurological causes during the follow-up.
The CLOSURE study provides no evidence of a reduction of recurrent stroke after transcatheter closure of patent foramen ovale than after antithrombotic therapy alone. Subgroup analyses showed corresponding results for large openings with atrial septal aneurysm.
The CLOSURE study had a number of weaknesses, inter alia a low inclusion rate. It took five years to recruit 909 patients, and the average recruitment rate per centre was two patients per year (37). Patients with a number of other risk factors for stroke were also included, including age up to 60, hypertension, diabetes, atherosclerosis and smoking, all of which may represent bias. Not all patients underwent a cerebral MRI.
The medical treatment was heterogeneous: warfarin for 24 months or acetylsalisylic acid or a combination of the two. According to the study protocol, the patients in the group with closure were to use antiplatelet therapy, although 97 of the patients had previously been taking warfarin. The STARFlex closure device (NMT Medical) was used, but is no longer available on the market (38).
In RESPECT, 980 patients aged 18 – 60 were randomised and followed up for 2.5 years (3). The primary endpoint was recurrent stroke or death. There was no significant difference between the groups. The risk of recurrent stroke/death was 0.66 events/100 patient-years in the closure group and 1.38 in the antithrombotic therapy group (p = 0.08). Subgroup analyses showed a significant difference in favour of closure in cases with a large opening (p = 0.01) and co-existing atrial septal aneurism (p = 0.02). The study showed that selected patients with embolic stroke on an MRI scan, without vascular risk factors and with a large opening, may benefit from closure.
In PC, 414 patients were included over four years (4). There was no significant difference in the incidence of the primary end-point, a combination of recurrent stroke, TIA or death, after closure compared with antithrombotic therapy. The primary end-point occurred in 3.4 % of the patients with closure and in 5.2 % of those who received antithrombotic therapy (p = 0.34). Ischaemic stroke occurred in one patient (0.5 %) in the closure group and in five patients (2.4 %) in the group with antithrombotic therapy (p = 0.14). Subgroup analyses revealed a non-significant difference in favour of closure for patients aged < 45 and with co-existing atrial septal aneurism.
The AMPLATZER PFO Occluder device (St. Jude Medical) was used in both RESPECT and PC. The complication percentage was lower than in studies where the STARFlex closure device was used, which indicates a better safety profile. The incidence of atrial fibrillation with transcatheter technique was also lower in RESPECT (3.0 %) and in PC (2.9 %) than in CLOSURE (5.7 %).
Weaknesses of the studies of transcatheter closure
We believe the studies have many limitations. It may be difficult to reveal relevant differences for subgroups. There is also reason to be critical of the inclusion criteria – patients with a tendency to thromboembolic conditions are excluded, while patients with other known risk factors for ischaemic stroke have been included.
Young patients with risk factors for venous thromboembolism, but without other known vascular risk factors, have the best expected closure effect, and should therefore be included first in these studies. For it to be possible to demonstrate a significant reduction in the risk of recurrent stroke with closure, a longer follow-up period (> 2 years) and large study population are also needed.
Furthermore, the closure procedure should be performed in centres with sufficient experience and a low complication rate.
Two randomised studies (REDUCE and CLOSE) of transcatheter closure versus antithrombotic therapy are currently ongoing. The Department of Neurology and the Department of Cardiology at Oslo University Hospital, Rikshospitalet, and Haukeland University Hospital are taking part in the REDUCE study. Hopefully, the results of these studies will yield better documentation, so that we can choose the optimal treatment for the individual patient.
Conclusion
Work-up for patent foramen ovale in patients with cryptogenic stroke should include TEE with saline contrast, ultrasound scans and CT or MR venography of the veins of the lower limbs with deep venous thrombosis in mind, as well as testing of risk factors for venous thromboembolism. The ischaemic stroke must be embolic and confirmed by an MRI examination.
Norwegian guidelines emphasise that the results of ongoing randomised studies will determine whether closure should be performed as secondary prophylaxis. Recent randomised studies indicate that antithrombotic therapy is as effective as transcatheter closure. There is reason to believe that subgroups of patients, such as young patients (age < 50 years) with thrombophilia, major defects or aneurisms, may benefit from closure, but the results of the randomised studies are ambiguous. Closure of patent foramen ovale should therefore be performed mainly as a part of randomised studies such that future recommendations may be based on sounder scientific documentation.
The patient has consented to the publication of the article.
MAIN POINTS
The optimal treatment for patients with cryptogenic stroke and patent foramen ovale is still unsettled
Treatment with antiplatelet drugs remains the first choice for the majority of patients if they cannot participate in a randomised study of transcatheter closure
Transcatheter closure may be appropriate for subgroups of patients with a particularly high risk of recurrent stroke
- 5.
Georgopoulos SE, Chronopoulos A, Dervisis KI et al. Paradoxical embolism. An old but, paradoxically, under-estimated problem. J Cardiovasc Surg (Torino) 2001; 42: 675 – 7. [PubMed]
- 6.
Meier B, Frank B, Wahl A et al. Secondary stroke prevention: patent foramen ovale, aortic plaque, and carotid stenosis. Eur Heart J 2012; 33: 705 – 13, 713a, 713b.
- 7.
Thomassen L, Waje-Andreassen U, Næss H et al. Transkranial dopplersonografi, paradokse embolier og høyre-venstre-shunt. Tidsskr Nor Lægeforen 2003; 123: 789 – 90. [PubMed]
- 11.
Næss H. Hjerneinfarkt hos unge voksne. Tidsskr Nor Lægeforen 2007; 127: 751 – 3. [PubMed]
- 14.
Augusseau MP, Pacouret G, Charbonnier B et al. Embolie paradoxale et thrombus enclavé dans le foramen ovale. Intérêt de l’échographie transoesophagienne. Arch Mal Coeur Vaiss 1997; 90: 1533 – 8. [PubMed]
- 17.
Sivertsen W, Risberg J, Norgård G. Apent foramen ovale og trykkfallssyke hos dykkere. Tidsskr Nor Legeforen 2010; 130: 834 – 8. [PubMed]
- 19.
Dowson A, Mullen MJ, Peatfield R et al. Migraine Intervention With STARFlex Technology (MIST) trial: a prospective, multicenter, double-blind, sham-controlled trial to evaluate the effectiveness of patent foramen ovale closure with STARFlex septal repair implant to resolve refractory migraine headache. Circulation 2008; 117: 1397 – 404. [PubMed] [CrossRef]
- 25.
Friis P, Gjestvang FT, Fausa D et al. Trombotisk eller embolisk hjerneslag? Tidsskr Nor Lægeforen 2001; 121: 2143 – 6. [PubMed]
- 26.
Indredavik B, Salvesen R, Næss H et al. red. Nasjonal retningslinje for behandling og rehabilitering ved hjerneslag. Oslo: Helsedirektoratet, 2010.
- 29.
Freyburger G, Trillaud H, Labrouche S et al. D-dimer strategy in thrombosis exclusion–a gold standard study in 100 patients suspected of deep venous thrombosis or pulmonary embolism: 8 DD methods compared. Thromb Haemost 1998; 79: 32 – 7. [PubMed]
- 33.
Hirth A, Greve G, Rosland GA et al. Kateterbasert lukking av åpentstående foramen ovale hos unge slagpasienter. Tidsskr Nor Lægeforen 2003; 123: 785 – 8. [PubMed]
- 36.
Harrer JU, Wessels T, Franke A et al. Stroke recurrence and its prevention in patients with patent foramen ovale. Can J Neurol Sci 2006; 33: 39 – 47. [PubMed]