Currarino syndrome (CS) (1) is a rare condition with a highly variable clinical presentation, which in some cases is inherited in an autosomal dominant fashion (2, 3). Up until 2008, around 300 cases had been published (4). Orphanet (5) reports a prevalence of 1 – 9 : 100 000. Around two thirds of cases are familial, with an even gender distribution. Among sporadic cases, there is a marked female preponderance (2).
The condition was described in The Lancet as early as 1837 (6), but it was only in 1981 that the radiologist Currarino and his colleagues published the work that led to common usage of the term Currarino syndrome (7). They describe the syndrome as a triad consisting of sacral, anorectal and presacral anomalies. The deformity of the sacrum (os sacrum) is variable, but in its typical form it is pathognomonic for the disorder. In such cases, a partial unilateral agenesis of the sacrum and coccyx gives them an appearance resembling a sickle, crescent or scimitar (a Turkish sword) (Fig. 1).
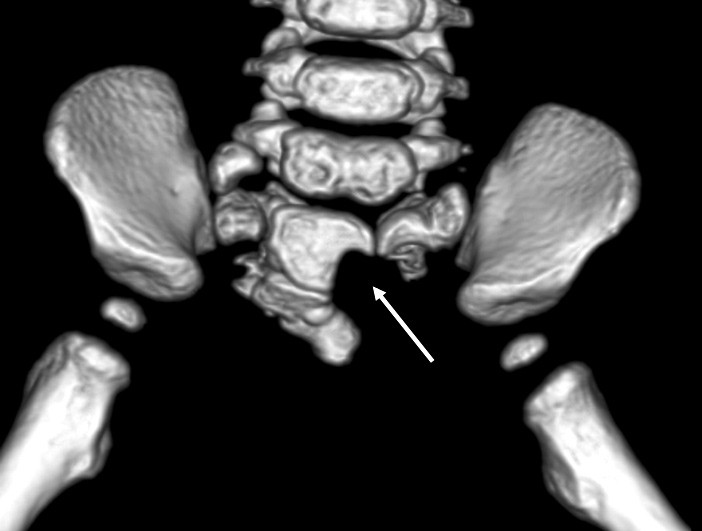
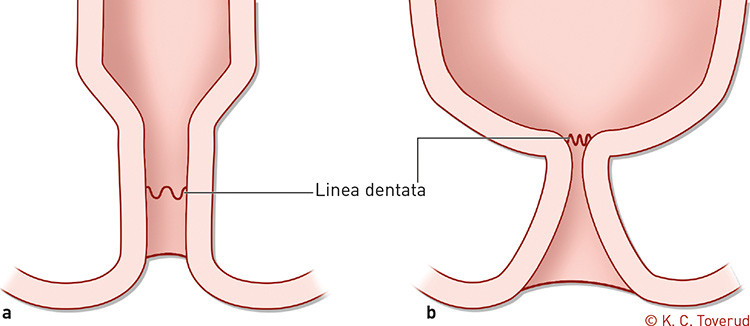
In its classic form, the anorectal anomaly consists of a very characteristic anal stenosis (Fig. 2). The anus is funnel-shaped up to the dentate line, where there may be any degree of stenosis. The presacral anomaly is described as a mass – it may consist of a teratoma, a meningocele, an enteric cyst or any combination of these.
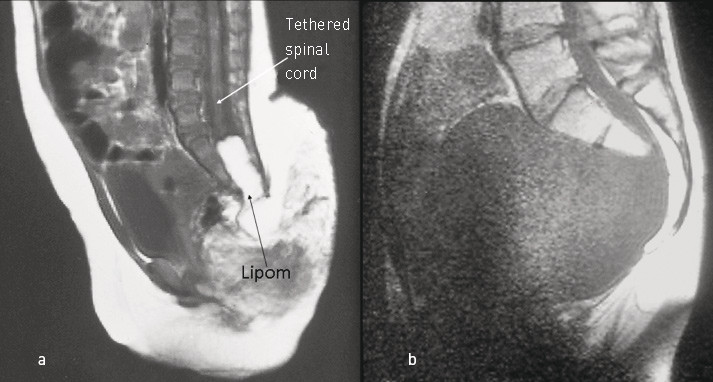
Many patients have intraspinal anomalies, most often a tethered spinal cord (Fig. 3a). In this condition, the lower part of the spinal cord including the medullary cone is distally displaced, often stretched, and may contain foreign tissues such as lipomas and teratomas. When the spinal cord is tethered in a sacral position, neurological signs and symptoms (paresis, sensory loss and pain) often progress with the lengthwise growth of the spine. Many patients also have urogenital anomalies (1, 2).
There is extensive phenotypic variation, which can make it difficult to distinguish Currarino syndrome from other congenital disorders of the sacral region, for example anal atresia. One patient with familial Currarino syndrome may have the entire triad as well as a tethered spinal cord and pronounced symptoms, while another family member with the same mutation may be completely without symptoms. In one series, it was found that a third of relatives with the mutation were asymptomatic (2). The large number of asymptomatic individuals combined with the extensive phenotypic variation leads to uncertainty in estimating the incidence of the condition.
The first disease gene was identified in 1998 through an analysis of three families, one of which was from our department (8). Familial Currarino syndrome is associated with dominantly inherited loss-of-function mutations in the MNX1 gene (motor neuron and pancreas homeobox 1 – previously called HLXB9) (8). The MNX1 gene has been localised to chromosome 7q36 (9). It consists of three exons and encodes a transcription factor. More than 70 disease-associated mutations have been identified so far (10, 11). No genotype-phenotype correlation has been identified (2, 3, 12).
Rikshospitalet began to use the diagnosis Currarino syndrome in 1986, but we have been treating patients with the condition since 1961. We now have, even by international standards, a large series of patients and asymptomatic relatives with mutations in the MNX1 gene. In recent years, access to molecular diagnostic tools has greatly improved our options for investigation. Here we present our experience of the investigation and treatment of this syndrome.
Material and method
The material is based on the medical records of patients with Currarino syndrome admitted to the Paediatric Surgical Service and the Department of Neurosurgery at Rikshospitalet over the period 1961 – 2012. Patients treated before 1986 were identified upon subsequent contact with the hospital and were included in the series if they retrospectively fulfilled the diagnostic criteria for Currarino syndrome. Other than mutation analyses, no new tests have been performed in these patients. Patient data are stored in an internal quality registry in the Paediatric Surgical Service in consultation with the Privacy and Data Protection Supervisor at Oslo University Hospital.
Patients were diagnosed with Currarino syndrome if they had symptoms and either a complete Currarino triad or a pathognomonic sacral bone deformity or a typical anal stenosis and any sacral bone deformity or carried a mutation in the MNX1 gene and belonged to a family with the syndrome. Individuals who were without symptoms, but who belonged to a family with Currarino syndrome and had a mutation in the MNX1 gene or a pathognomonic sacral bone deformity were registered as asymptomatic mutation carriers (Box 1).
Diagnostic criteria used to make a diagnosis of Currarino syndrome or mutation carrier.
Patients received a diagnosis of Currarino syndrome if they had symptoms and fulfilled one of the following criteria:
-
Complete Currarino triad
-
Any sacral bone deformity
-
Anal stenosis
-
Presacral mass
-
-
Pathognomonic sacral bone deformity
-
Typical anal stenosis and any sacral bone deformity
-
Mutation in the MNX1 gene and member of a family with Currarino syndrome
Individuals in families with Currarino syndrome were defined as asymptomatic mutation carriers if they did not have symptoms and fulfilled one of the following criteria:
-
Mutation in the MNX1 gene identified
-
Pathognomonic sacral bone deformity identified
All patients and, with one exception, all relatives carrying an identified mutation underwent X-ray examination of the sacrum. Most of the patients, but none of the asymptomatic mutation carriers, then had CT and/or MRI scans of the pelvic region, the lumbosacral spine and the spinal cord, as well as ultrasound examination of the urinary tract. Other tests varied according to individual needs. Four patients with anal stenosis also had a bowel biopsy in order to exclude Hirschsprung’s disease.
Mutation analysis
From as far back as 1986, patients and their first-degree relatives have, in connection with genetic counselling, been offered the option of having their DNA stored for future genetic analysis. In 2009, when the relevant mutation analysis became available at Rikshospitalet, the individuals in question were contacted and offered the analysis as well as further genetic counselling. The results of the mutation analyses in the four families included in our series have recently been published (11).
Results
Over the period 1961 – 2012, we treated 29 patients with Currarino syndrome. With the aid of X-ray examinations and mutation analyses, we also identified eight asymptomatic mutation carriers. Mutations in the MNX1 gene were detected in 15 patients and in seven asymptomatic mutation carriers (Table 1). In the group with sporadic Currarino syndrome, 12 of the 14 patients were girls, whereas there was an even gender distribution in the group with familial syndrome. There were no obvious differences between boys and girls in the symptom profile (data not shown). All patients and three asymptomatic mutation carriers (seven examined) had deformity of the sacral bone. With one exception, all patients had presacral masses; 19 had anal stenosis and ten had a tethered spinal cord (Table 1).
Table 1
Findings from genetic analysis and imaging diagnostics of patients with Currarino syndrome and relatives, Rikshospitalet 1961 – 2012. Familial cases were identified through the detection of a pathognomonic sacral bone deformity via X-ray and/or through mutation analysis.
| Patients with familial Currarino syndrome (n = 15) |
Asymptomatic mutation carriers in a Currarino syndrome family (n = 8) |
Patients with sporadic Currarino syndrome (n = 14) |
Total (n = 37) |
|
| Deformity of sacral bone |
15 |
3¹ |
14 |
32 |
| Anal stenosis |
11 |
0 |
8 |
19 |
| Presacral mass |
15 |
–² |
13 |
28 |
| Tethered spinal cord |
4 |
–² |
6 |
10 |
| Documented mutation in the MNX1 gene |
14³ |
7¹ |
1⁴ |
22 |
| [i] | ||||
[i] ¹ 7 of 8 examined
² None examined
³ 14 of 15 examined
⁴ 7 of 14 examined
Table 2 summarizes the interventions performed on the 29 patients. The great majority had had symptoms since birth, and two thirds were diagnosed in their first year of life. There were no obvious differences between familial and sporadic cases with regard to clinical presentation and course of treatment. A total of 26 of the 29 patients had constipation, including cases with neonatal bowel obstruction. Symptomatic urogenital anomalies were identified in twelve patients.
Table 2
Treatment of 29 patients with Currarino syndrome at Rikshospitalet over the period 1961 – 2012
| Treatment |
Number of patients |
| Colostomy |
15¹ |
| Dilatation of anal stenosis |
14 |
| Bowel resection/plastic surgery for anal stenosis |
8 |
| Surgery for presacral tumour/meningocele/enteric cyst |
16 |
| Laminectomy |
14² |
| Surgery for urogenital anomalies |
7 |
| [i] | |
[i] ¹ At last contact, 5 and 2 patients still had a colostomy and appendicostomy, respectively
² Ten of whom underwent surgery for a tethered spinal cord and 4 for a meningocele/intraspinal lipoma
When the data were analysed in 2012, none of the patients had died as a consequence of Currarino syndrome. However, there had been significant morbidity. For instance, the four patients with the most severe phenotype had undergone between 13 and 18 operations related to Currarino syndrome.
All 19 patients with anal stenosis, and seven without, had constipation requiring treatment. Four patients had had biopsies taken, none of which revealed any pathology of the enteric nervous system. Colostomies had to be performed on 15 patients, and five of them still had a colostomy and two an appendicostomy for antegrade colonic irrigation when the study was closed. Moreover, the great majority of patients used various types of laxatives and enemas.
Fourteen of the patients with anal stenosis underwent attempted anal dilatation using Hegar dilators of increasing diameter. This was successful in only two cases. Resection or plastic surgery for anal stenosis was performed on eight patients; however, five of them still have a colostomy or appendicostomy.
Sixteen patients had had their presacral masses excised. Teratomas were the most common cause, alone or in combination with a meningocele or an enteric cyst. Twelve patients with masses diagnosed by means of imaging had not undergone surgery. None had developed malignant disease.
Ten of 14 laminectomised patients had had a tethered spinal cord released, in some cases in combination with removal of a meningocele or intraspinal lipomas. Apart from urinary incontinence, which affected four, no patients had appreciable neurological sequelae caused by Currarino syndrome.
Twelve patients had urogenital anomalies, and seven of them had undergone surgery. The most common indication for surgery was vesicoureteral reflux (five patients), but conditions such as ureterocele, septate vagina and rectovaginal fistulas also required surgical treatment.
Genetics
Mutations in the MNX1 gene were detected in all 14 analysed patients from four families (families A-D), and also in seven of the 17 asymptomatic relatives who were tested. The mutations identified are shown in Table 3.
Table 3
Mutations in patients with Currarino syndrome and first-degree relatives. Of the sporadic cases, seven patients and five relatives were examined. Genetic findings from family A have previously been published by Ross et al. (8) and from all families by Holm et al. (11)
| Gene tested patients and relatives (n = 43) |
Patients with documented mutation (n = 15) |
Asymptomatic mutation carriers (n = 7) |
Type of mutation |
|
| Family A |
15 |
7 |
4 |
c.340delC |
| Family B |
8 |
3 |
3 |
Deletion of entire MNX1 |
| Family C |
6 |
2 |
0 |
c.53dupC |
| Family D |
2 |
2 |
– |
c.336dupG |
| Sporadic |
12 |
1 |
0 |
Deletion of entire 7q36 including MNX1 |
Seven of the 14 patients with sporadic Currarino syndrome underwent genetic testing. One patient had a deletion of the entire 7q36 region, which contains a large number of genes, including MNX1. In the remaining six, no MNX1 gene mutations were found.
There was marked phenotypic variation. For instance, three of the six members of family B who had the gene deletion were completely asymptomatic, while another was among the most severely affected in the entire series.
Discussion
In our material, constipation requiring surgical treatment has been the dominant clinical symptom of Currarino syndrome. Half of our patients have also required neurosurgical interventions and one quarter, urogenital operations. No patient has died as a consequence of the condition, but there has been substantial morbidity and a considerable number of interventions – several patients have undergone surgery more than ten times.
Almost all patients suffered from constipation, and this proved difficult to treat. More than half of the patients had a colostomy established in infancy or in early childhood. While anal stenosis is indisputably a physical obstacle, in the great majority of patients the presacral mass is so small that it is hard to imagine that it represents a physical impediment. The frequent spinal cord pathology makes it reasonable to suspect that some patients may also have a neurological basis for their bowel disorder. This interpretation is supported by the fact that seven patients were severely constipated even though they did not have anal stenosis. The disappointing results achieved with dilatation treatment and bowel resection/plastic surgery, resulting in seven patients needing a permanent ostomy, also support the idea that anal stenosis cannot be the sole explanation for the constipation.
The possibility that the enteric nervous system is involved in Currarino syndrome is sparsely covered in the literature. Before we became aware of the diagnosis, bowel biopsies were taken from four of our patients to exclude Hirschsprung’s disease. None of them showed any histological evidence in support of this diagnosis. Moreover, in occasional case reports, the rectum is described as having normal innervation in Currarino syndrome (13) – (15). However, a combination of Currarino syndrome and Hirschsprung’s disease has been described in about ten cases (16). We have therefore updated our routines, and rectal suction biopsy is now included in the workup of all Currarino syndrome patients with constipation and is considered especially important in cases without anal stenosis.
Presacral tumours and the risk of malignancy
None of our patients experienced malignant transformation of their presacral tumour. Prior to 1999, only a single death from malignant disease had been reported in children with Currarino syndrome (17). Accordingly, our view was that excision of the presacral masses was not a matter of any urgency.
However, it has now been reported that at least four girls, aged between one and three years (17) – (20), and six adults (21) – (26) with Currarino syndrome developed presacral malignancy – and at least seven of them have died. Based on current knowledge of the risk of malignancy, we have revised our position and now recommend surgery within the first year of life.
Neurosurgery
Half of our patients have undergone neurosurgery. The fact that so many patients have neurological deficits has contributed to the term «Currarino syndrome» being preferred to «Currarino triad» (2).
In the early days, we were more reluctant to operate to release a tethered spinal cord. The possibility of inadvertent nerve damage during surgery made us fear that loss of function could outweigh the benefits gained by releasing the stretched spinal cord. Patients were observed and operated upon on recognition of incipient neurological deficits. However, positive experiences with improved diagnostics, superior microsurgical technique and peroperative neurophysiological monitoring have now led us to recommend surgery earlier.
Anterior meningoceles through the defect in the sacrum pose special problems. Due to their connection with the cerebrospinal fluid system, these meningoceles may appear as growing, expansive processes in the pelvis and abdominal cavity. Puncturing the meningocele may lead to meningitis (Fig. 3B). Cases of meningitis associated with Currarino syndrome differ somewhat from meningitis in general in that they are often caused by intestinal bacteria and may be polymicrobial (27). Meningoceles complicated by meningitis, along with malignancy, constitute the two main causes of mortality associated with this syndrome (21, 28, 29).
Genetics
Patients should be referred for genetic counselling and their close relatives offered screening (2, 3, 12, 30, 31). Before genetic diagnostics became available, it was recommended that the examination of parents, siblings and children begin with X-ray of the sacrum (2, 3),(12). Today, it is natural to begin the screening of first-degree relatives with mutation analysis.
Knowledge of whether or not a child has inherited the family’s mutation has important consequences with regard to treatment. Children with a mutation should be examined using diagnostic imaging and, if necessary, undergo surgery. Children without a mutation can be considered healthy, and require no further follow-up.
A characteristic feature of the mutation-carrying population is the relatively large number of asymptomatic individuals. In the four families included in our series, 33 % of those shown to have the mutation were asymptomatic (Table 3). The corresponding figure in the study by Lynch and co-workers was also 33 % (2). We do not know how many relatives declined the offer of mutation analysis. If there are undiagnosed mutation carriers in the families, the proportion of asymptomatic individuals could be even higher.
Abnormality of the MNX1 gene was detected in only one of the seven sporadic cases examined. This patient had a terminal deletion of chromosome 7q, which includes the MNX1 gene among others and which is known from the literature (32, 33). A lower prevalence of mutations in sporadic cases of Currarino syndrome is consistent with previous observations, which have shown that mutations are present in the majority of familial cases, but in only 30 % of sporadic cases (10). One possible explanation could be that some of the sporadic cases may be caused by a somatic mutation. It is also conceivable that in the sporadic cases, there may be mutations in unknown sequences that are involved in regulating the expression of the MNX1 gene.
The great majority of articles on Currarino syndrome are case reports. In view of the condition’s varied clinical presentation and the fact that the syndrome as an entity is relatively young, it is not surprising that there are contradictory findings and recommendations in the literature. However, our experiences and opinions correspond well with two previous, larger, works: Lynch and colleagues’ collected material on 205 patients (2) and Crétolle and colleagues’ review of 50 patients treated at a single institution in Paris (4).
With backing in the literature, we now recommend that asymptomatic mutation carriers should be examined with MRI (3, 14, 25, 34), and that presacral tumours (except for small meningoceles) should be removed (3, 25, 34).
The patient depicted in Figure 3b and the patient with deletion of 7q36 have given their consent to the publication of this article.
MAIN POINTS
Currarino syndrome is an autosomal dominant hereditary condition with a highly variable clinical presentation
Over the course of a 50-year period, Rikshospitalet has treated 29 patients with this syndrome
Constipation requiring treatment was the main problem in almost all patients; half underwent a colostomy and the majority had presacral tumours
Patients and their first-degree relatives should be referred for genetic counselling and mutation analysis
- 1.
OMIM. (Online Mendelian inheritance in Man) #176450, 2012. Currarino syndrome. www.omim.org (10.12.2012).
- 3.
Scherer S, Martucciello G, Belloni E et al. HLXB9 (MNX1) and sacral agenesis and the Currarino syndrome. In: Epstein CJ, Erickson RP, Wyndshaw-Boris A, red. Inborn errors of development. The molecular basis of clinical disorders of morphogenesis. Oxford: Oxford University Press, 2008: 740 – 6.
- 5.
Orpha #1552, Currarino syndrome. www.orpha.net (10.12.2012).
- 6.
Bryant T. Case of defiency of the anterior part of the sacrum, with a thecal sac in the pelvis, similar to the tumour of spina bifida. Lancet 1837; I: 358 – 60.
- 25.
Urioste M, Garcia-Andrade MC, Valle L et al. Malignant degeneration of presacral teratoma in the Currarino anomaly. Am J Med Genet A 2004; 128A: 299 – 304. [CrossRef]
- 28.
Amacher AL, Drake CG, McLachlin AD. Anterior sacral meningocele. Surg Gynecol Obstet 1968; 126: 986 – 94. [PubMed]