Therapeutic drug monitoring (TDM) can be useful in a number of clinical situations (1). Measuring drug levels in blood, serum or plasma (a definition of these test materials is provided in Box 1) can reveal whether the patient has an aberrant drug metabolism or is non-compliant, or provide an indication of the degree of seriousness of intoxications. The measured levels may be of help in optimising the dosage. Despite a long tradition in Norway of using TDM as a clinical tool (2), no nationwide overview of the TDM repertoire at the various laboratories is available. The purpose of this article was to establish such an overview.
Serum, plasma or whole blood?
The straw-coloured, transparent fluid which is left in a test tube once the blood cells have been removed by centrifugation can be either plasma or serum. The difference between plasma and serum consists in whether only the blood cells have been removed (in which case plasma remains) or whether coagulating factors such as fibrinogen have also been removed along with the sediment (in which case serum remains). In practice, this means that if the sample has been collected in a tube without additives, serum is left after centrifugation, but if an anticoagulant (such as heparin or EDTA) has been added, plasma will remain after centrifugation. Most analyses will return equal results from measurements made in plasma and serum. In Norway, serum is traditionally used for therapeutic drug monitoring, but many other countries use plasma.
Whole blood refers to blood with an evenly mixed content, such as it appears in vivo. In Norway, drug analyses of whole blood are primarily performed in a judicial context, for example in cases of traffic violations or forensic autopsies. However, some drugs, for example immunosuppressives, are routinely analysed in whole blood also for the purpose of therapeutic drug monitoring.
Drug concentrations in whole blood may deviate considerably from those measured in plasma/serum, and a comparison of results across the tested material requires knowledge about the distribution of the individual substance in the various blood segments.
Material and method
The Government’s website provides a list of all public and private hospitals in Norway (3). During the period from October through December 2011, we used this list to call all hospitals. The hospitals’ switchboard operator provided information on the location of laboratories in subordinate units, and the telephone number to these laboratories. All clinical biochemistry and pharmacological laboratories were contacted. The laboratories were requested to report the types of drug analyses (including drugs of abuse and toxic alcohols, but excluding metabolites) they performed in blood/serum at the time of the call. In August 2012, when this manuscript had been accepted for publication, the laboratories were again contacted by e-mail. The heads of the laboratories were thus given the opportunity to undertake quality assurance and, if necessary, update the TDM analysis repertoire as of August 2012.
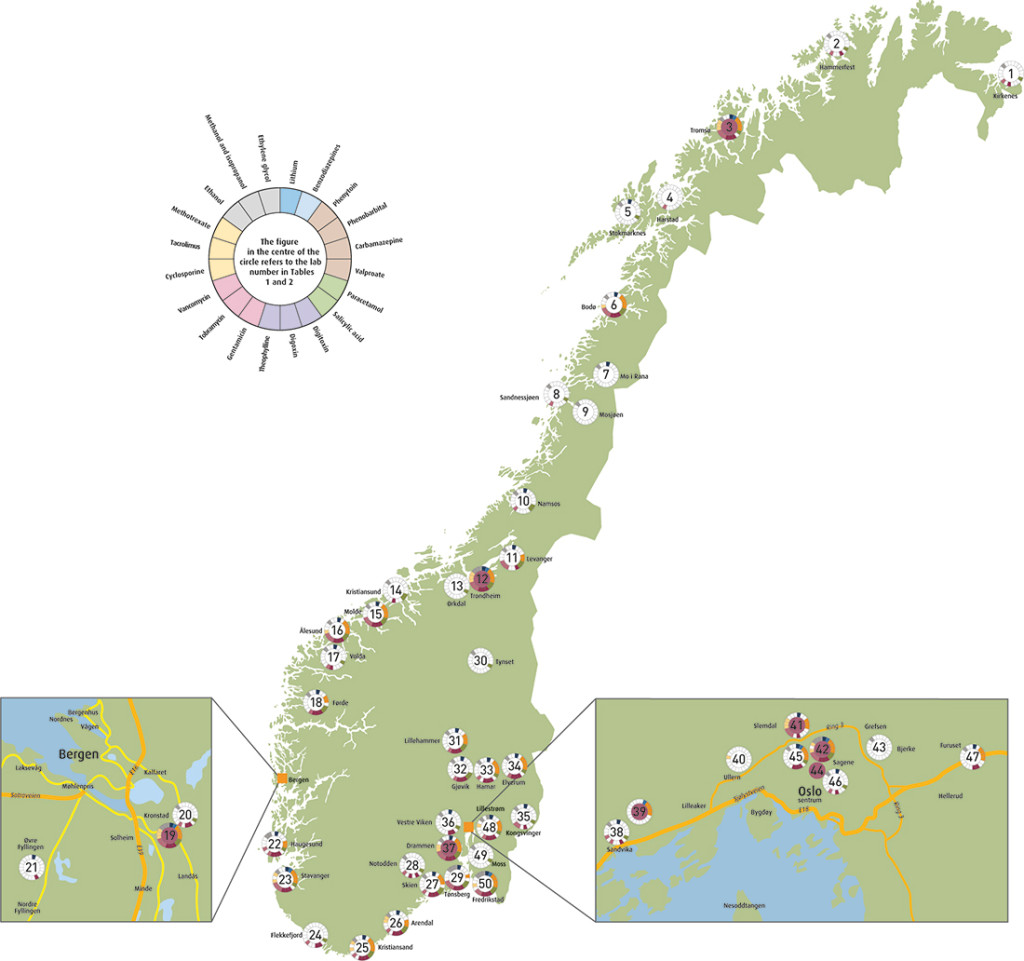
Only the routine repertoire, i.e. the analyses that were listed on the laboratory’s requisition form or similar overviews, was registered. Google Maps was used to locate the laboratories geographically (e-Figure 1). The collection included analyses of blood/serum only; pharmacological analyses in urine – which mainly pertain to testing for drugs of abuse – were excluded.
Results
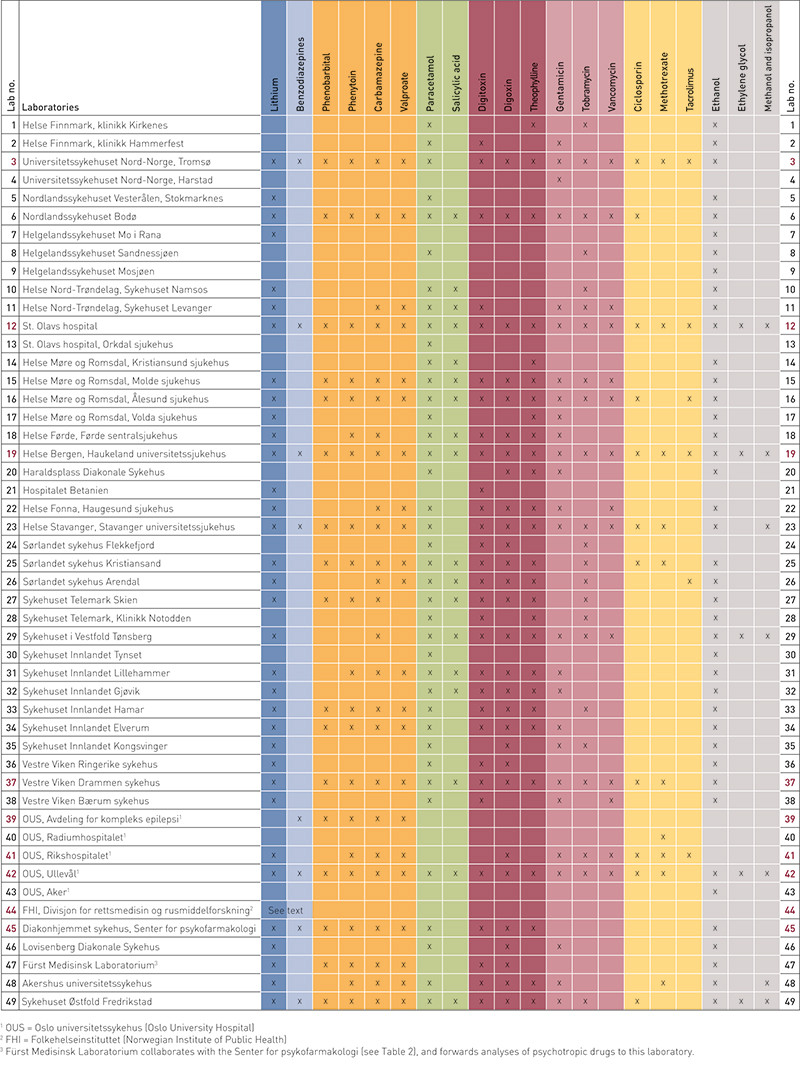
In total, approximately 80 laboratories were contacted, and altogether 49 laboratories that performed TDM analyses in blood/serum were identified. All of these responded to our e-mail in August 2012, and confirmed and/or updated their analysis repertoire. In total, these laboratories offered analyses of 151 different substances. An overview of all the laboratories – with their respective repertoires of drug analyses – is provided in Table 1 and e-Figure 1. Nine specialised laboratories (circles in purple shading in e-Figure 1 and purple numbers in Table 1) had analysis repertoires that went beyond the most common analyses (Table 1). These laboratories are presented separately in Table 2. Table 1 and e-Figure 1 thus provide a complete overview of Norwegian laboratories that perform TDM analyses in blood/serum, while Table 2 provides a complete overview of all TDM analyses that can be performed on a routine basis.
The «typical» TDM repertoire
The laboratories listed in Table 1 had an average analytical repertoire of 7.4 substances in blood/serum (range 1 – 17) when the specialised laboratories were excluded. The substances for which analyses were most commonly available included ethanol and paracetamol, performed by 41 and 39 laboratories respectively. These were followed by lithium, digitoxin, digoxin and theophylline, all performed by approximately 30 laboratories. Four antiepileptics (phenobarbital, phenytoin, carbamazepine and valproate), three antibiotics (gentamicin, tobramycin and vancomycin), as well as salicylic acid, were each analysed by approximately 20 laboratories. The remaining analyses shown in Table 1 were performed in 11 hospitals or fewer, including the specialised laboratories.
The specialised laboratories
As shown in e-Figure 1, there is at least one general pharmacological laboratory within each regional health authority: Northern Norway, Central Norway and Western Norway have their pharmacological departments at the University Hospital of North Norway, St. Olavs Hospital and Haukeland University Hospital, respectively. In South-Eastern Norway there are two general pharmacological laboratories, one in Oslo University Hospital and one at Vestre Viken Drammen. All these laboratories perform a wide range of TDM analyses across various therapeutic fields. However, the laboratories’ ranges of analyses vary in character and scope, as shown in Table 2. For example, that the widest TDM repertoires for benzodiazepines, antipsychotics and antiviral drugs are found in Tromsø, Trondheim and Oslo (Ullevål), respectively.
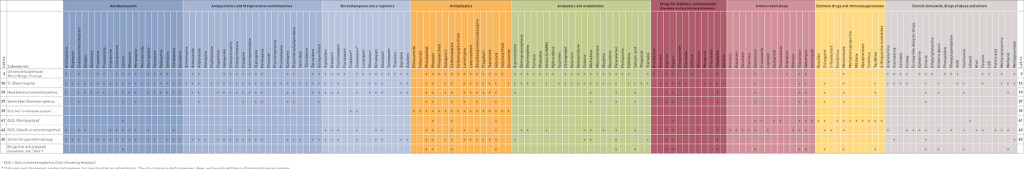
In addition to the five general pharmacological laboratories referred to above, there are four specialised laboratories with TDM repertoires linked to specialised therapeutic areas or functions. All of these are located in the Oslo region. The Centre for Psychopharmacology at Diakonhjemmet Hospital has a broad TDM repertoire of drugs used in psychiatry. The laboratory at the Department of Refractory Epilepsy (formerly Statens senter for epilepsi, SSE) offers the country’s broadest range of TDM analyses for antiepileptic drugs, while the laboratory at Rikshospitalet has the broadest range for antimycotic, immunosuppressive and cytotoxic drugs. The fourth specialised laboratory is the Division of Forensic Medicine and Drug Abuse Research at the Norwegian Institute of Public Health. This laboratory performs a wide range of pharmacological analyses (4), although its activities are primarily related to samples collected in a judicial context, such as traffic violations, incarcerations and forensic autopsies. Its analytical activities are also different from those of the other laboratories included in this overview, since they perform analyses in whole blood samples. As a consequence, the measured levels may differ from those detected in serum or plasma (Box 1). This labaratory does not routinely perform analyses for the purpose of TDM.
Discussion
No requirements exist as to which TDM analyses Norwegian laboratories should perform, neither in large nor in small hospitals. Local variations are therefore considerable. The purpose of this article was to establish an overview of the combined national TDM repertoire for blood/serum. The overview can be used as a tool by clinicians who need to know which analysis can be performed, and where. For the staff at clinical biochemistry and pharmacological laboratories, it may also be beneficial to know where to forward samples for analyses that they do not perform themselves.
The data collection process for this article turned out to be more complicated than we assumed at the outset. The organisational structure of the hospitals was rarely intuitive, and without local knowledge it was difficult to predict which institutions would have their own laboratories. We therefore attempted to use each laboratory as a source to identify other laboratories in the same region, and we believe that we have succeeded in this.
Large hospitals represented an extra challenge, since the same institution could be host to several laboratories. In these cases – where the laboratories were located close to each other – we merged the analyses into a single repertoire. In cases where the laboratories were more distinctly separated in terms of geography and function, such as in Oslo University Hospital, we chose to present the laboratories individually.
This overview must be considered to have a restricted shelf life. The laboratories’ TDM repertoire is constantly changing, which can be illustrated by the digitalis glycosides digitoxin and digoxin. Digitoxin was withdrawn from the market while this study was underway (5), and many patients who used to take digitoxin now take digoxin. This has implied a change in the need for analyses. At the time of the first request (in late 2011), digitoxin and digoxin were analysed by 33 and eight laboratories respectively. At the time of the update in August 2012 numbers had changed to 30 and 29.
Despite the fact that this overview represents a «snapshot» of the situation prevailing in August 2012, we nevertheless consider that it may serve as a useful reference. It is important, however, to be aware that pharmacological laboratories will often be capable of performing a wider repertoire of analyses than those listed in this overview. If the laboratory has access to reference material of the substance to be analysed, a rough chromatographic analysis can usually be developed within a relatively short period of time. To achieve a precise quantification, however, the method of analysis must be validated, preferably through internal and external quality control programmes. This is very resource-intensive. Therefore, pharmacological laboratories tend to remove rarely performed analyses from their routine TDM repertoire, and rather keep these in a separate «research and development repertoire». Such analyses – not shown in this overview – may be requested in special cases, in agreement with the performing laboratory.
Conclusion
We present a snapshot of the TDM repertoire in Norway as of August 2012. The presentation is provided in the form of graphs and tables, to make it easily usable as a tool in daily practice. A web-based, dynamic version of this overview is being planned.
We wish to thank Kari Mette Kalland and PK-Trykk for their help with the graphic design.
MAIN MESSAGE
Altogether 49 laboratories in Norway offer therapeutic drug monitoring (TDM) analyses in blood/serum.
Analyses for a total of 151 different substances are available.
The most comprehensive range of analyses is related to psychotropic and antiepileptic drugs.
TDM is also available within several other therapeutic fields.
- 1.
Hiemke C, Baumann P, Bergemann N et al. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: update 2011. Pharmacopsychiatry 2011; 44: 195 – 235. [CrossRef]
- 2.
Brørs O. Plasmakonsentrasjonsanalyse av legemidler med immunologiske metoder. Tidsskr Nor Lægeforen 2000; 120: 2427 – 9. [PubMed]
- 3.
Helse- og omsorgsdepartementet. Oversikt over landets helseforetak. www.regjeringen.no/nb/dep/hod/tema/sykehus/oversikt-over-landetshelseforetak.html?id=485362 (10.3.2012).
- 4.
Nasjonalt folkehelseinstitutt, Divisjon for rettsmedisin og rusmiddelforskning. Komponentliste, 2011. www.fhi.no/dokumenter/76d62838a9.pdf (10.3.2012).
- 5.
Statens legemiddelverk. Avregistrering av Digitoxin tabletter og overgang til digoksin, 2011. www.legemiddelverket.no/templates/InterPage____83024.aspx. (10.3.2012).