MAIN MESSAGE
In 2010 close to 2 % of the Norwegian population received analgesics on reimbursable prescription for chronic non-malignant pain.
This represents a very small proportion of those reporting chronic pain in epidemiological studies.
Fewer than 8 % of these patients were prescribed opiods, corresponding to 0.14 % of the population,
Many were prescribed short-acting opiods or opiods in conjunction with benzodiazepines, which is contrary to Norwegian guidelines.
Several epidemiological studies show that the prevalence of chronic pain in Norway is approximately 24 – 30 % and that only a minority of these cases are reported to be cancer-related (1) – (3). In a Norwegian telephone interview study, 14 other European countries and Israel reported that around half (52 %) of those with chronic pain used prescribed analgesics at the time of the study (1). Approximately one third did not receive pain treatment. This reflects the fact that many people control their chronic pain without pharmacological treatment. However, some have such severe chronic pain that use of analgesics becomes necessary. In Norway up until 2008, only patients with certain specific diagnoses such as arthrosis or rheumatoid arthritis were prescribed non-opioid pain relievers under the drug reimbursement scheme. In the same period it was possible for individual patients to apply for reimbursement of the costs of long-term opioid treatment.
Changes to the drug reimbursement scheme from 2008 meant that patients with chronic pain and significantly reduced quality of life and ability to function were able to obtain pain-relieving medication under the drug reimbursement scheme using reimbursement code-71 (chronic non-malignant pain). This scheme applies to all types of severe, chronic pain and is not limited to particular diagnoses. Reimbursement for opioids additionally requires that the risk of dependency be considered by a specialist or hospital department as subordinate to the need for pain relief (4).
Opioids are indicated for pain relief for a minority of patients with chronic non-malignant pain for which non-opioid analgesics give insufficient relief. National guidelines recommend the use of long-acting formulations when opioids are indicated for this population (5, 6). Further, concomitant use of other potentially addictive drugs and a combination of short and long-acting opioids are advised against (5, 6).
A fairly recent study shows that the percentage of the population regularly using opioids on a continuous basis for non-malignant pain was 1.08 % at the beginning of 2007 (7). It is also known that patients with a high usage of opioids are often prescribed other potentially addictive drugs in addition (8, 9). A study by the National Prescription Database showed that 50.5 % of those taking > 365 defined daily doses (DDD) of codeine in 2005 also used > 100 defined daily doses of benzodiazepines (8). Another study has shown that the majority of those taking high doses of opioids on a continuous basis take short-acting opioids (9), which is not in accordance with the Norwegian guidelines (5).
The research questions for this study were:
-
How many patients are prescribed analgesics with reimbursement code -71?
-
Which pain-relieving drugs are prescribed under reimbursement code -71?
-
Which opioids, opioid dosages and opioid formulations are prescribed with reimbursement code -71?
-
What other pain-relieving and potentially addictive drugs are prescribed to patients who are additionally prescribed opioids under reimbursement code -71?
Material and method
Data source
Since 1 January 2004, all Norwegian pharmacies have been obligated to report data on all dispensed prescriptions to the National Prescription Database. The information registered is date of dispensing, generic name, product name, size of pack, number of packs, ATC (Anatomical Therapeutic Chemical Classification System) code, amount measured as defined daily doses, and price. A defined daily dose is the assumed average maintenance dose per day for a drug used for its main indication in adults (10).
Patients are identified using a unique encrypted number instead of their personal identification number so that they can be followed pseudonymously over several years. The patients’ age, gender, municipality of residence, pharmacy and where relevant the drug’s reimbursement code are stored. The prescribers are pseudonymised in the same way with a unique identification number, and information regarding their specialty, gender and age is stored (11).
Study population
In this study all those for whom at least one drug was dispensed under reimbursement code -71 in 2009 and/or 2010 were included (Table 1). The study is based on data for all prescriptions dispensed to patients in 2009 and 2010.
Table 1
Overview of number of patients receiving drugs with pain-relieving effect under reimbursement code -71 in 2009 and 2010. The sum of the number of prescriptions dispensed and the percentages for each medication exceeds the total number of patients (N) because some received several medications.
| 2009 N = 77 967 | 2010 N = 90 731 | ||||||||
| Number | (%) | Percentage of women | Age (years) Average (SD) | Number | (%) | Percentage of women | Age (years) Average (SD) | ||
| Paracetamol | 25 063 | (32) | 69 | 62 (17) | 33 242 | (37) | 69 | 62 (17) | |
| NSAIDs total¹ | 23 019 | (30) | 66 | 56 (15) | 25 864 | (29) | 66 | 55 (15) | |
| Amitriptyline | 16 955 | (22) | 72 | 54 (15) | 20 654 | (23) | 72 | 54 (15) | |
| Gabapentin | 15 584 | (20) | 61 | 58 (16) | 16 507 | (18) | 61 | 58 (16) | |
| Pregabalin | 9 495 | (12) | 61 | 57 (16) | 8 112 | (8,9) | 62 | 57 (15) | |
| Carbamazepine | 3 486 | (4,5) | 55 | 61 (17) | 3 468 | (3,8) | 56 | 61 (17) | |
| Levomepromazine | 2 620 | (3,4) | 64 | 58 (16) | 2 956 | (3,3) | 63 | 58 (15) | |
| Duloxetine | 944 | (1,2) | 65 | 52 (15) | 1 724 | (1,9) | 67 | 53 (15) | |
| Non-opioids total | 75 643 | (97) | 66 | 59 (17) | 87 863 | (97) | 66 | 59 (17) | |
| Opioids | 5 634 | (7,2) | 66 | 61 (16) | 6 875 | (7,6) | 66 | 61 (16) | |
| [i] | |||||||||
[i] ¹ Non-steroid anti-inflammatory drugs
Analysis strategy
Age-stratified prevalence over a one-year period was defined as the number of patients and the percentage of Norway’s population (12) for whom at least one opioid prescription was dispensed under reimbursement code -71 (Table 2).
Table 2
Patients who have been reimbursed for at least one prescription of an opioid under reimbursement code -71 in 2009 and 2010. For each age group the number of recipients and percentage of the country’s population are given. The population figures for 2009 and 2010 are collected from Statistics Norway and were 4 799 252 and 4 858 199 respectively.
| Age group (years) | 2009 | 2010 | |||
| Number | (%) | Number | (%) | ||
| < 40 | 515 | (0.02) | 573 | (0.02) | |
| 40 – 49 | 1 050 | (0.15) | 1 244 | (0.18) | |
| 50 – 59 | 1 237 | (0.20) | 1 499 | (0.24) | |
| 60 – 69 | 1 113 | (0.23) | 1 391 | (0.28) | |
| 70 – 79 | 844 | (0.29) | 974 | (0.34) | |
| > 80 | 875 | (0.40) | 1 194 | (0.54) | |
| Total | 5 634 | (0.12) | 6 875 | (0.14) | |
We have chosen only to present data for 2010 for the analyses of co-medication for reasons of space and because of stable patterns for this type of medicating (Table 3). Co-medication was defined as other drugs prescribed at least once in the period between the first and last reimbursable opioid prescription in 2010, and was classified according to the extent to which opioid users were given mainly long-acting or mainly short-acting opioids or whether they used both types. «Main use» of long-acting/short-acting formulations was defined as at least 90 % of the use measured in defined daily doses being long-acting or short-acting opioid formulations (Table 3). The total number in Table 3 is lower than the total number of opioid users in 2010 because according to our definitions of co-medication there were 431 patients who did not receive co-medication.
Table 3
Overview of number of patients reimbursed for opioids with reimbursement code -71 during 2010 who in addition were dispensed at least one of the drugs below in the period between the first and last opioid prescription. Stratified according to the extent to which patients have received mainly short-acting opioids, mainly long-acting opioids, or both.
| Mainly long-acting (n = 4 047) | Mainly short-acting (n = 1 051) | Short-actingand long-acting (n = 1 346) | ||||||
| Number | (%) | Number | (%) | Number | (%) | |||
| Other opioids without reimbursement | 2 350 | (58) | 580 | (55) | 949 | (70) | ||
| Paracetamol | 1 965 | (49) | 364 | (35) | 684 | (51) | ||
| Benzodiazepines | 1 579 | (39) | 366 | (35) | 686 | (51) | ||
| Z-hypnotics | 1 376 | (34) | 490 | (47) | 552 | (41) | ||
| NSAIDs | 1 318 | (33) | 317 | (30) | 468 | (35) | ||
| Gabapentin | 537 | (13) | 124 | (12) | 242 | (18) | ||
| Pregabalin | 450 | (11) | 77 | (7.3) | 158 | (12) | ||
| Amitryptiline | 445 | (11) | 96 | (9.1) | 166 | (12) | ||
| Levomepromazine | 75 | (1,9) | 17 | (1.6) | 60 | (4.5) | ||
| Carbamazepine | 36 | (0,9) | 7 | (0.7) | 18 | (1.3) | ||
| Duloxetine | 35 | (0,9) | 12 | (1.1) | 23 | (1.7) | ||
Analyses of the average daily dosage of the most commonly used opioids in 2010 (e-Table 4) are based on oral morphine equivalents (OMEQ) (13). The OMEQs for different opioids are more comparable in terms of their equianalgesic effect than DDD. Daily dosages of different opioids were calculated as the total quantity of opioid divided by the number of days from the first to the last prescription, with an addition of 30 days (the treatment period). Those given one prescription during the treatment period or who were dispensed the first and last prescription on the same day were not included in the analyses of average daily dosage. The data were analysed in SPSS version 18.
Table 4
Percentage distribution of patients’ average daily dosage in 2010, stratified in 10 mg strata from 0 to 50 mg, in 50 mg strata from 50 mg to 200 mg as well as from 200 mg to 300 mg and > 300 mg. Daily dosage calculated as total quantity of opioid during the treatment period.
| 2010 | ||||||
| OMEQ¹/day | Morphine (n = 21) | Oxycodone (n = 1 195) | Buprenorphine (n = 1 385) | Fentanyl (n = 85) | Tramadol (n = 743) | Paracetamol/codeine (n = 834) |
| < 10 mg | 1.2 | 14 | 2.3 | 0.1 | 0.5 | 25 |
| 10 – 20 mg | 5.8 | 15 | 16 | 1.2 | 1.9 | 37 |
| 20 – 30 mg | 5.6 | 13 | 20 | 4.3 | 7.5 | 22 |
| 30 – 40 mg | 5.1 | 7.7 | 17 | 3.3 | 13 | 8.7 |
| 40 – 50 mg | 6.5 | 6.0 | 12 | 2.9 | 12 | 3.8 |
| 50 – 100 mg | 22 | 20 | 28 | 26 | 49 | 3.2 |
| 100 – 150 mg | 18 | 8.2 | 3.4 | 18 | 12 | 0.4 |
| 150 – 200 mg | 8.1 | 4.5 | 0.4 | 12 | 2.6 | 0.0 |
| 200 – 300 mg | 11 | 5.5 | 0.6 | 19 | 1.5 | 0.0 |
| > 300 mg | 18 | 6.3 | 0.1 | 14 | 0.8 | 0.0 |
| [i] | ||||||
[i] ¹ OMEQ = oral morphine equivalents
Ethics
The study is based on pseudonymised data from the National Prescription Database and therefore does not require review by the Regional Committee for Medical and Health Research Ethics or the Data Protection Official for Research.
Results
In total 854 977 prescriptions were reimbursed using reimbursement code -71 in 2009 and 2010. Prescriptions that were excluded because they lacked unique patient identification amounted to 0.3 % while other reasons for excluding prescriptions accounted for a further 0.3 % (Figure 1).
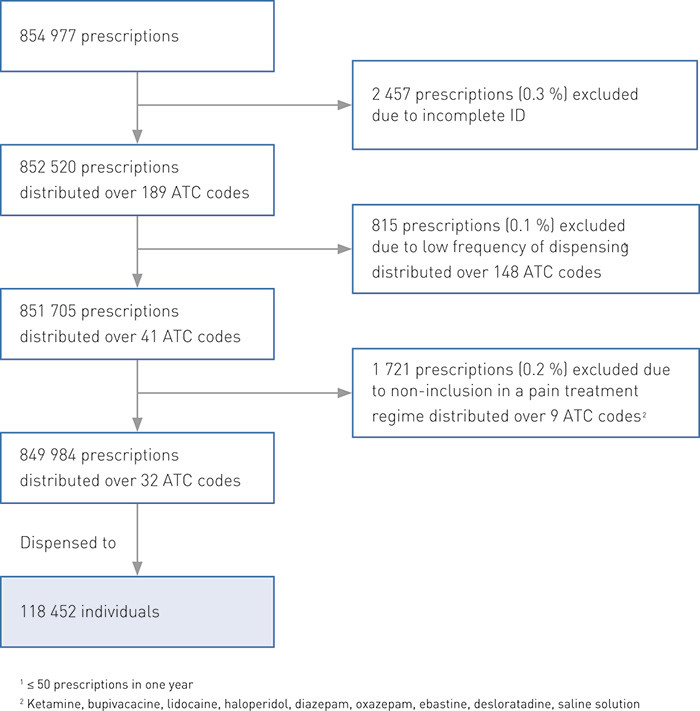
Drugs reimbursed under code -71
77 967 and 90 731 patients respectively were reimbursed for at least one prescription for a pain-relieving drug in 2009 and 2010 (Table 1). In each of these years 7.2 % and 7.6 % respectively were reimbursed for at least one prescription for an opioid. The pain-relieving drugs that were reimbursed for most patients in 2010 were paracetamol (33 242 patients) and non-steroidal anti-inflammatory drugs (NSAIDs) (25 864 patients), followed by amitriptyline, gabapentin, pregabalin and opioids. The proportion of women varied from 55 % to 72 % for the various drugs, with the average age being from 52 to 62 years.
Patients who receive opioids with reimbursement code -71
5 634 and 6 875 patients respectively were given opioids with reimbursement code -71 in 2009 and 2010 (Table 2). 66 % were women and the average patient age was 61 for both years. The prevalence of reimbursed opioid use increased with age.
The different opioids
Oxycodone was the most commonly dispensed opioid with reimbursement code -71 in 2010, followed by buprenorphine and tramadol (e-Table 5). In total 5 792 patients were dispensed at least one prescription for a long-acting opioid and 2 556 received at least one short-acting opioid with reimbursement. This is higher than the total number who had opioids reimbursed (6 875), because there were 1 473 patients who received reimbursement for both short and long-acting formulations.
Table 5
Overview of number of patients who were reimbursed for at least one opioid prescription under reimbursement code -71 in 2009 (N = 5 634) and 2010 (N = 6 875). The sum total for the different drugs exceeds the total number of patients and the total of the percentages exceeds 100 because a portion of the patients were dispensed several formulations or types of drugs in the course of one year. In each of the two years fewer than 15 individuals were reimbursed for the opioids fentanyl (short-acting), hydromorphone (long-acting and short-acting), dihydrocodeine and codeine; these are therefore not included in the table.
| 2009 N = 5 634 | 2010 N = 6 875 | ||||
| Number | (%) | Number | (%) | ||
| Oxycodone (long-acting) | 1 290 | (23) | 1 450 | (21) | |
| Oxycodone (short-acting) | 611 | (11) | 713 | (10) | |
| Oxycodone + naloxone¹ | 13 | (0.2) | 106 | (1.5) | |
| Buprenorphine (transdermal) | 1 500 | (27) | 2 006 | (29) | |
| Buprenorphine (short-acting) | 78 | (1.4) | 86 | (1.3) | |
| Tramadol (long-acting) | 804 | (14) | 1 048 | (15) | |
| Tramadol (short-acting) | 339 | (6.0) | 452 | (6.6) | |
| Paracetamol/codeine | 909 | (16) | 1 029 | (15) | |
| Morphine (long-acting) | 505 | (9.0) | 559 | (8.1) | |
| Morphine (short-acting) | 177 | (3.1) | 196 | (2.9) | |
| Fentanyl (transdermal) | 735 | (13) | 855 | (12) | |
| Ketobemidone | 124 | (2.2) | 145 | (2.1) | |
| Dextropropoxyphene | 123 | (2.2) | 108 | (1.6) | |
| Methadone | 92 | (1.6) | 119 | (1.7) | |
| Pethidine | 31 | (0.6) | 35 | (0.5) | |
| Number who were dispensed (at least one) short-acting opioid | 2 227 | (40) | 2 556 | (37) | |
| Number who were dispensed (at least one) long-acting opioid | 4 673 | (83) | 5 792 | (84) | |
| [i] | |||||
[i] ¹ Targiniq
Opioid formulations and co-medication
Of the patients who received opioids on reimbursable prescriptions, over 30 % received co-medication with benzodiazepines, more than 30 % received benzodiazepine-like sleep medications and over half received other opioids not included in the reimbursement scheme (Table 3). Only 21 % of those who received opioids were not concomitantly prescribed other habit-forming drugs. The prevalence of co-medication with other habit-forming drugs was highest in patients who received reimbursement for both short and long-acting opioid formulations.
Opioid dosage
The percentage distribution of average daily dosage is presented in e-Table 4. Most of those who received paracetamol/codeine combinations were given relatively low daily dosages – 93 % of these received < 40 mg OMEQ daily and 59 % < 20 mg OMEQ daily (corresponding to four paracetamol/codeine tablets). For buprenorphine, too, by far the majority were low and medium dosages: 10 – 100 mg OMEQ daily for 93 % of the patients. The dosages for those receiving tramadol were somewhat higher: 20 – 150 mg OMEQ daily for 93 % of the patients.
For the strong opioids – morphine, oxycodone and fentanyl – the patterns were more ambiguous. While high percentages received > 100 mg OMEQ daily, particularly for morphine and fentanyl, lower dosages were dispensed to a large percentage of the patients who received oxycodone.
Discussion
The main findings of this study are that approximately 2 % of the population receives pain-relieving drugs on reimbursable prescriptions because of chronic non-malignant pain and that 0.14 % of the population is given opioids under this scheme. These are low percentages in view of the fact that 24 – 30 % of the Norwegian population reports to have chronic pain (1) – (3) and that over 1 % of the population uses opioids continuously on a long-term basis for other indications than palliative treatment (7).
A large percentage of those prescribed opioids on reimbursable prescriptions have received both short and long-acting opioid formulations and/or benzodiazepines. This is contrary to national guidelines (5, 6). More patients received pain relievers with reimbursement due to non-malignant pain in 2010 than in 2009, but the patterns appear to be stable. The increase may be due to increased awareness of the scheme and to the fact that there was a limited capacity for specialist assessments regarding opioids when that particular reimbursement right was introduced.
This study shows that in 2009/2010 less than 10 % of all those reporting chronic pain received reimbursement for analgesics under the new regulation. This is a significantly lower percentage than was assumed to use analgesics continuously, based on previous studies. In a telephone interview study conducted in Norway as well as a number of other countries, 52 % of those with chronic pain reported that they «currently used prescribed analgesics» (1). A slightly older Norwegian study showed that 43 % of the portion of the population who reported chronic pain used analgesics (3). In an American telephone interview study, 4.9 % of the general population reported having used opioids on at least five days a week for the previous four weeks (14).
While data on how many people use non-opioid analgesics and co-analgesics on a long-term basis is very uncertain, more reliable data has recently become available on how many use opioids continuously. Data from the National Prescription Database has shown that in excess of 1 % of the population receive opioids on a long-term basis because of other indications than palliative treatment (7).
The number receiving reimbursable opioids for chronic non-malignant pain amounts to 0.14 % of the population in our sample. The interpretation of this difference is either that the overwhelming majority of patients who receive opioids for chronic non-malignant pain are not receiving the refund to which they are entitled, or that a high number of patients receive opioids long-term without fulfilling the conditions for reimbursement of the costs of opioids. The former explanation would imply that many patients have an unreasonable and unnecessary financial burden in addition to their pain disorder; the latter would imply a significant social problem with many patients using opioids on a long-term basis without a good medical indication for this. Since we do not have access to clinical data on patients in the National Prescription Database, it is impossible to decide, based on the data in this study, which explanation is the correct one. There is, however, reason to assume that there are few good indications for long-term treatment with opioids where the treatment does not meet the requirements for reimbursement with reimbursement code -71. Individual doctors should therefore carefully assess whether such patients should either receive opioids under the drug reimbursement scheme, or whether opioid treatment should be discontinued. For patients at the end of life there are other drug reimbursement schemes and prescribing of this type was not included in this study. The number of patients receiving reimbursable opioids for palliative treatment was previously 12 000 – 13 000 per year (9).
Non-opioid analgesics must be the first line of treatment for chronic non-malignant pain. This is reflected in this study, with approximately one third of patients who are reimbursed for analgesics receiving paracetamol and one third being reimbursed for NSAIDs. Paracetamol is relatively safe for long-term use, while long-term use of NSAIDs can give rise to side-effects which in some circumstances may be serious, e.g. gastrointestinal bleeding in the elderly and in patients with heart, lung or kidney disease. In this study we found that many had paracetamol and non-steroid anti-inflammatory drugs reimbursed in addition to opioids. This can give an opioid-saving effect but must be carefully weighed against the risk of side-effects of NSAIDs.
The pattern of prescribing also reflects that amitryptiline is a first-line drug for neuropathic pain. A higher percentage of reimbursements for gabapentin compared to pregabalin is presumably due to the requirement that the cheaper drug, gabapentin, must be tried first, while reimbursement for pregabalin is available only on application from individual patients. The number receiving amitryptiline, gabapentin or pregabalin without being given opioids is considerably higher than the number receiving both opioids and one of these drugs. This indicates that opioids are used as supplementary treatment only in a relatively small percentage of patients with neuropathic pain.
For duloxetine and pregabalin, changes to the reimbursement rules may have influenced the pattern of prescribing between 2009 and 2010. In those cases where reimbursement is linked to specific diagnoses, it is possible to speculate whether there is a tendency to make diagnoses that qualify for reimbursement. Patients who receive medium or higher dosages of weak opioids should, according to the guidelines, be switched to a depot formulation of a strong, long-acting opioid. Worthy of mention in this connection is that persons using high doses of tramadol or combining tramadol with certain other medicines have a risk of developing serotonin syndrome.
The increased incidence of dispensing of opioids with increasing age is commensurate with the increased prevalence of painful conditions in the elderly (1, 2). Opioids can have a good effect in the elderly while side-effects such as a tendency to fall and reduced cognitive function mean that the effect and the side-effects must be evaluated carefully.
Of those who receive opioids on reimbursable prescription, four times as many primarily receive long-acting opioids as the number receiving short-acting ones. This indicates that there is a general attempt to follow the guidelines on this point. Nevertheless it is worrying and clearly contrary to the guidelines that there are > 30 % who co-medicate with benzodiazepines and > 50 % who co-medicate with other opioids outside of the reimbursement scheme.
Approximately 10 % of the general population receive opioids in the course of a year, mainly in small quantities (9) and presumably because of acute pain. One would expect that acute conditions would also occur in those using opioids on a regular basis, with an indication for short-term increase in the opioid dosage. However, the very high percentage of patients receiving opioids both with and without reimbursement in the course of a year indicates that many who are reimbursed for long-acting opioids use a short-acting drug in addition, without this being attributable to an acute injury or similar circumstance. This type of use is warned against in the guidelines, and may indicate established problematic opioid use or a risk of developing such. The prevalence of co-medication with other potentially addictive drugs is especially high in the group being reimbursed for large quantities of both short and long-acting opioids, and it can be assumed that both the incidence and risk of developing problematic opioid use is therefore higher here than among the other patients in the study population.
The strength of studies based on the National Prescription Database is that the data material includes all prescriptions dispensed in Norway in the study period and that data is available at the individual level that is not based on self-reporting. The weakness of these types of studies is that it is impossible to know when or whether the medication is used by the recipient. Another disadvantage is that there is no access either to individual data on the use of medication in hospitals or nursing homes. There is therefore an underestimation of both the use and dosage of medications, especially for the elderly and chronically ill.
A weakness of this study is the lack of knowledge of whether those who have received different opioids and opioid formulations in the course of the study period use the different drugs concomitantly, whether they have had a change in their treatment or whether they switch between treatments. The sequence in which the prescriptions are dispensed does not necessarily reflect the sequence in which the medications are used.
Opioids were defined according to ATC code N02A, ATC code N07BC02 (methadone) and ATC code R05DA04 (codeine). The opioids were classified into long-acting and short-acting according to their route of administration. Long-acting opioids were defined as depot tablets, depot capsules, depot plasters and all routes of administration for methadone (tablets and oral solution). Short-acting opioids were defined as tablets, capsules, sublingual tablets, suppositories, transmucosal fentanyl, injection forms, oral solution and nasal spray.
Benzodiazepines were defined according to ATC codes N05BA (anxiolytics), N05CD (hypnotics and sedatives) and N03AE01 (clonazepam). Benzodiazepine-type sleep medications were defined according to ATC code N05CF (benzodiazepine-related medicines). Other pain-relieving medications with reimbursement code -71 which were investigated in this study were gabapentin, pregabalin, carbamazepine, amitryptiline, duloxetine, levomepromazine, paracetamol and NSAIDs. The latter was defined according to ATC code M01A.
Conclusion
This study shows that patients who receive analgesics with reimbursement for the indication of chronic, non-malignant pain mainly receive non-opioid analgesics. Since the number who are reimbursed under the drug reimbursement scheme is low compared with the large percentage of the population who have chronic non-malignant pain, there is reason to assume that many patients themselves bear the costs associated with the use of pain-relieving medication. Patients with a good indication for continuous use of both non-opioid analgesics and opioids should, in line with the regulations, receive these drugs under the drug reimbursement scheme.
For those who do not fulfil the requirements for reimbursement it will in many cases be correct to discontinue long-term treatment with opioids. The combined use of different opioids/formulations and/or concomitant use of benzodiazepines has a high incidence and is contrary to the guidelines. This presumably implies an increased risk of problematic opioid use.
- 4.
Folketrygdloven § 5 – 14. Legemidler, næringsmidler og medisinsk forbruksmateriell. 2011. www.lovdata.no/for/sf/ho/xo-20070628-0814.html (29.8.2012).
- 5.
Terapianbefaling: Bruk av opioider ved behandling av langvarige, non-maligne smertetilstander – en oppdatering. Oslo: Statens legemiddelverk, 2008.
- 6.
Bell RF, Bjørner T, Forseth KØ et al. Retningslinjer for smertelindring. Oslo: Den norske legeforening, 2009.
- 7.
Svendsen K, Skurtveit S, Romundstad P et al. Differential patterns of opioid use: Defining persistent opioid use in a prescription database. Eur J Pain 2011. E-publisert 19.12. [PubMed]
- 10.
International language for drug utilization research. www.whocc.no/ (12.10.2011).
- 12.
Statistisk sentralbyrå. www.ssb.no (12.10.2011).