Should genetic findings from genome research be reported back to the participants?

Genome research, i.e. the use of exome or whole-genome sequencing technologies, is now entering medical research and diagnostics at full speed. This has given rise to an intense ethical debate in international and national expert forums as to how this technology should be handled. For example, the issue of whole-genome sequencing has been discussed in most of the meetings of the Norwegian Biotechnology Advisory Board from 2010 to 2012 (B. Solberg, personal communication).
Genome research generates larger and more comprehensive data sets than the genetic analyses that have hitherto been used. In theory, the technology can reveal all large and small variations in the genome as a whole. Even though this represents a scientific advance, it remains an ethical challenge: A researcher may risk – intentionally or otherwise – obtaining genetic information that would be of importance to the individual participant. The question of whether returning research-generated information to individual research participants – we can call it feedback – is to be seen as a moral imperative, has attracted a large amount of academic attention in recent years. If this is implemented in practice, it may give rise to direct consequences for individuals, the research community and society at large. In this article we will present the main arguments for and against provision of such feedback.
Knowledge basis
The search engines PubMed and Google Scholar were used to obtain an overview of the international academic literature related to genetic and genome analyses. The different search criteria are shown in Table 1. Different combinations of words and expressions, one from each of the groups 1, 2 and 3 were used in a single search. In other searches, the words in groups 4 and 5 were added to the different combinations, separately or jointly. This approach was chosen because of the lack of a standardised terminology.
Table 1 Search criteria for literature searches
To identify articles on ethical challenges in genetic and genome analyses in general and provision of feedback in particular, various combinations of words and expressions were used in literature searches in PubMed and Google Scholar. A single search always included words or expressions from Groups 1, 2 and 3, in several cases also in combination with words from Groups 4 and 5.
| Group 1 |
Gene, genetic, genome, genomic, DNA |
| Group 2 |
Incidental, incidental findings, research results, results, information, unexpected findings |
| Group 3 |
Feedback, return, disclose, report |
| Group 4 |
Ethics, ethical |
| Group 5 |
Research |
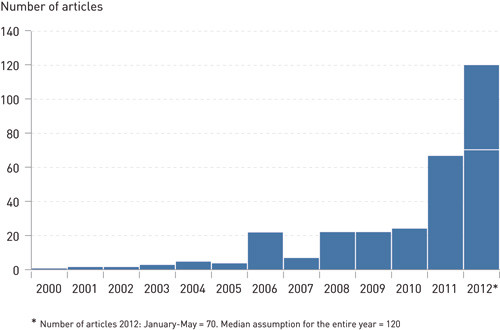
The number of articles describing ethical challenges in genetic and genome analyses in general and provision of feedback in particular has increased significantly in later years (Figure 1). Altogether 266 articles were deemed to be relevant and were registered in a separate data base. Summaries and articles were systematically reviewed. The main arguments «in favour» or «against» were identified and presented.
Arguments in favour of providing feedback
A prominent moral intuition in all those who argue in favour of providing feedback to research participants appears to rest on our obligation to save lives or prevent serious illness if we are in a position to do so. Scanlon (1) and Miller and collaborators (2) refer to this as «the rescue principle». If we interpret this as «an obligation to come to the rescue», the question concerns the number of genetic variations that can be claimed to be of such a nature that «life is at stake» for an individual research participant. On the other hand, if we interpret it as «an obligation to help», we open the field to a wider range of genetic variations that can be deemed to be helpful for the individual to be aware of (3, 4).
However, the point is that international experts who argue on the basis of an obligation to rescue or help tend to conclude that objective criteria associated with clinical benefit may determine the debate on provision of feedback. It is claimed that high clinical benefit ought to indicate provision of information on individual findings, whereas low clinical benefit would indicate no feedback. Another expression that embodies the same idea is the term «actionable». Findings that can give rise to action – that are «actionable» – should be reported back, because in this case one can be reasonably certain that the information may be of help to the research participant (5).
On the other hand, it may be difficult to determine what types of genetic information could be perceived as useful and «actionable» by each individual. Even though preventive treatment for a certain condition may not be widely available, it is conceivable that individuals would welcome this information about risks. This has led some theoreticians onto the «self-determination track», according to which a respect for the research participant indicates that each individual should be able to decide what kind of genetic information he or she wishes to have access to or have reported back (6, 7). Here, the autonomy of the research participant is promoted. Feedback will be triggered by the availability of results and the preferences of the individual participants, as described by Budin-Ljøsne (8). Angrist represents the unconditional advocates of self-determination (9). He claims that only the individual will be able to assess what he/she may deem to be of benefit for his/her life – anything else should be perceived as illegitimate paternalism. Participants in genome research have the right to receive their entire genome on a memory stick if they so wish, he writes.
The self-determination track is also reinforced by the ideal of mutuality and reciprocity in research. McGuire & Lupski (10) and Fernandez (11) have pointed out that participants contribute to research, and it is not unreasonable to give them something in return. It is easy to maintain that the participants are to some extent «taken advantage of» if they are only to contribute, but remain barred from receiving in return something that they may perceive as beneficial. This is one of Greely’s main grievances; he exhorts us to «take to the streets» to fight for a general obligation to provide feedback of research-generated genetic and genome information (12). In addition, various studies show that when laypeople are asked, the vast majority would want feedback of genetic information (13) – (15).
Ravitsky & Wilfond pursue a third road (16). Clinical benefit is their key issue, while self-determination, autonomy and mutuality are not given as much weight. Instead, they include assessments of research contexts. For example, they claim that a possible proximity between the researchers and the participants, in combination with the assessment of clinical benefit, may decide whether feedback is necessary or not. The authors refer to ongoing, clinically based studies of families with hereditary diseases. The context, characterised by an overlap of examination, research and treatment, as well as close involvement of researchers with the participants, may be a strong argument in favour of providing feedback. This contrasts sharply with, for example, epidemiological studies, in which the researchers are not involved in a therapeutic relationship with the participants.
Miller and collaborators also emphasise the relationship between the researchers and the participants as an argument in favour of provision of feedback (2). For these theoreticians, however, the existence of proximity is not the decisive factor for provision of feedback, it is rather seen as the researcher’s professional obligation, given his or her superior expert knowledge. When a researcher has been given access to our genome, a professional stance would indicate reporting of any «errors» found in it, much the same as we would expect a craftsman to tell us about any faults that he accidentally discovered in our home. To Miller and collaborators, this converts into an argument in favour of provision of feedback from research directly involving patients, as well as from population-based studies. The obligation to report can be claimed to be part of the researcher’s professional duties.
Many of the proponents of feedback from genome research also claim that in addition to catering to the autonomy and welfare of the participants, feedback will serve to increase openness about research and increase the motivation for participating (17), as well as reducing the opportunities for indicting researchers for having withheld information «that could have been of material benefit» to the individual. In the same way as measurement of cholesterol levels and blood pressure has for many years served as an «enticement» for people to participate in large population studies, provision of feedback on genetic variations may help ensure future recruitment to such studies.
Arguments against providing feedback
The arguments used against provision of genome information are of a deontological, consequentialist and pragmatic nature. Some theoreticians are concerned that the nature of the obligations in a doctor-patient relationship is completely different from that in a researcher-participant relationship (18, 19). Research is not intended to be of benefit to the participant. Those who harbour such an idea will soon fall prey to what has been referred to as «the therapeutic misconception» (20) – (22). Several sceptics, for example Forsberg and collaborators, argue that the obligations of the researcher, apart from not causing harm to or inflicting strain on the participants, primarily consist in maximising the generation of knowledge (23). This argumentation is frequently used in the context of population-based studies, in which many presumably healthy people participate (and furthermore, people are never invited to participate because they may possibly be ill). Furthermore, the insights to be gained from such basic research are intended to be of benefit to future groups of patients (24, 25). As part of a society with universal, evidence-based health services, we can say that we constantly receive something in return for our participation in medical research (23). Here, the ideal of mutuality is interpreted at the aggregate, collective level. We have no claim to individual benefits from participating in research projects, since we enjoy their advantages in the form of high-quality, evidence-based health services.
In a consequentialist perspective, it is further argued that the commitment to knowledge will be rendered more difficult if requirements to provide feedback are introduced (26, 27). The focus will change, since human and financial resources are diverted away from research towards individual facilitation, for example in the form of an increased need for quality assurance, more thorough interpretation of results, direct counselling and further follow-up of individuals and their families (14, 28).
The argument pertaining to limited obligations is also reinforced by the situation established by modern population studies and bio-bank research. Here, there is little similarity to a doctor-patient relationship. Instead, research is undertaken on a de-identified material that may have been donated many years earlier. The researchers involved never meet the research participants, many of the researchers have no medical training and the relationship to the participant is of a completely abstract nature (29).
Another argument based on consequentialist ethics focuses on how feedback of genetic risk information from research may harm individuals (30, 31), as well as increase the «medicalisation» of the population and thus give rise to unnecessary use of healthcare resources on presumably healthy people (32, 33). Refraining from providing feedback prevents any such harmful effects. Nobody will be worse off than they were prior to the research project, says Parker (34), thus claiming that provision of feedback from research is absolutely no obligation. In many cases, no provision of feedback may therefore be the safest option. According to Ossorio (27), one cannot be criticised for having treated the research participant merely as a means to an end in the Kantian sense, as long as the participant was informed in advance of this practice of non-provision of feedback.
The pragmatic argument focuses on the unsuitability of research results for provision of feedback: The data as such may be fraught with sequencing errors (35). For practical, technical, financial and scientific reasons, the requirements for quality may be somewhat lower than for corresponding diagnostic analyses. It can also be claimed that most of the genome information that has been generated, especially from population-based studies, is fraught with great uncertainty in terms of its validity and benefit at the individual level (36, 37).
In addition to these arguments, some claim that genome research has no special position or exception from research as such, and that findings that are important to individuals may also result from research of a «non-genetic» character. Insisting on provision of feedback from genome research may thus be seen as an expression of a genetic «exceptionalism», which is unfortunate at the individual as well as the social level (33). In a cultural sense, genetic variants may easily be ascribed with an exaggerated importance for the health and behaviour of individuals.
By themselves, these arguments do not preclude a recognition of the fact that researchers, like everybody else, have an obligation to save lives if they are in a position to do so. The question remains, however, whether this obligation to save lives is particularly relevant with regard to genetic predispositions for illness, according to those who oppose provision of feedback.
Discussion
In the bio-ethical literature on provision of feedback of genetic risk factors that have been identified by genome research, the dominating assessment appears to be that it is right to provide opportunities to let the participants receive such feedback. The number of articles with arguments in favour of providing feedback seems to be much higher than the number of articles with counterarguments. Furthermore, weighty consensus articles have argued in favour of providing feedback (5, 38), while none have defended «non-feedback». The articles we have reviewed have been published in a variety of journals in the fields of bio-ethics, medicine and natural science. They are thus being read by a wide audience, and must be assumed to be potentially able to exert a considerable influence on biomedical research practices.
Provision of feedback is further promoted by the fact that the arguments «in favour» appear to be more in line with the «gut feeling» of people in general than the arguments «against». Several empirical articles show that the majority would want or expect individual genetic information if they were to volunteer to participate in research. Corresponding attitudes can also be seen in a Norwegian report from 2010, based on a population study commissioned by the Directorate of Health and conducted by Perduco in cooperation with the authors of this article, among others. Here, altogether 41 % responded that it would be unacceptable if genetic researchers did not provide individual feedback on elevated risk of illness (39). Facilitating provision of individual feedback from research is justified with reference to easily understandable and powerful arguments, such as respect for the participants and their autonomy, concern for the participants’ health and welfare, the obligation to help and the ideal of mutuality. This approach focuses on the individuals and ensures their interest and rights by letting the wishes of the individual decide. This also appears to be in line with classical research ethics, by giving precedence to the welfare and interests of the individual, above the interests of society.
The justifications used by the opponents of feedback are less easily accessible. Arguing that researchers in a moral sense do not owe the participants any feedback may easily seem egotistic and a defence of the interests of the powerful. Nor is arguing against «the therapeutic misconception» especially easy if the individual research participant envisions that he or she may stand to benefit if the researchers prepare the ground for this therapeutic misconception. Although the arguments may be less easily accessible and are in a minority in the literature, this does not mean that they are weak. The possibility that presumably healthy people may be worried and medicalised as a result of receiving feedback from genetic research is very real. This was a major topic in Norway as recently as in 2007, with the so-called MIDIA affair (40).
There may thus be weighty reasons to attempt to maintain a distinction between healthcare and research, wherever this is possible and meaningful. It is far from obvious that basic research institutions should produce individualised genetic risk information and provide (or feed) such information to re-identified individuals. This may even be claimed to harm research as well as participants. In genome sequencing in large population studies, such as the Nord-Trøndelag Health Study (HUNT), there is a considerable potential for turning healthy people in the population into people who are genetically at risk. Research ethics should as far as possible balance individual perspectives against perspectives of social medicine and the internal obligations of research institutions.
We are standing at a crossroads with regard to assessing whether returning research-generated genetic risk information at the individual level constitutes a moral imperative. Here, individually based research ethics runs up against social-medical concerns and research-based obligations. An equilibrium between these has probably not yet been reached, neither in Norway, nor internationally. Irrespective of the conclusion arrived at, the conclusion and the justifications must be communicated clearly and understandably to the participants. This is the unquestionable obligation of the researchers and the research institutions to the participants.
Tabell
| Main message |
|
- 1.
Scanlon TM. What we owe to each other. Cambridge, MA: Belknap Press of Harvard University Press, 1999.
- 5.
Wolf SM, Crock BN, Ness BV et al. Managing incidental findings and research results in genomic research involving biobanks and archived data sets. Genet Med 2012; 14: 361 – 84.
- 8.
Budin-Ljøsne I. A review of ethical frameworks for the disclosure of individual research results in population-based genetic and genomic research. Research Ethics 2012; 8: 25 – 42. [CrossRef]
- 29.
Solberg B, Steinsbekk KS. Managing incidental findings in population based biobank research. Nor J Epidemiol 2012; 21: 195 – 202.
- 39.
Perduco. Bioteknologiloven. Undersøkelse om holdninger til etiske problemstillinger, 2010. http://helsedirektoratet.no/kvalitet-planlegging/bio-genteknologi/Documents/bioteknologi-etikk.pdf (18.6.2012).
- 40.
Frydenberg K. Forskning til hvilken pris? Hvor stor skade kan vi tåle hvis forventet resultat er veldig viktig? Utposten 2007; 3: 6 – 9.