Cancer in children is rare, and accounts for only 1 % of all cancer incidences in Norway (1). Care for children with cancer is special because the patient is growing and developing, and most have the prospect of a long life after curative cancer treatment. The prognosis for children with cancer is normally good (5-year survival > 80 %) (2, 3) and the number of children who survive cancer is rising steadily (4). We now assume that one in 640 adults in the age group 20–39 is a long-term survivor of childhood cancer (4).
There has been a significant increase in recent years in the attention surrounding children who survive cancer treatment, and in 2006 Oeffinger et al. presented a major, ground-breaking American study in which they found that 62.3 % of 10 337 survivors of childhood cancer had at least one chronic sequela an average of 17.5 years after treatment (5). The condition of 27.5 % of the whole study population was defined as serious or life-threatening. A recent Swedish study has shown that impaired work capability and a greater need for public support also apply to Nordic survivors of childhood cancer (6). Long-term survivors of brain tumour are the most severely affected. The most serious late effects of childhood cancer treatment are neurological sequelae, cardiological disease, secondary malignancy, hormonal imbalance and cognitive impairment (7). One study has shown that secondary malignancy after treatment for childhood cancer occurs in 9.7 % of cases an average of 17.8 years after the primary cancer diagnosis (spread 5 – 35.2 years) (8). It is maintained that long-term survivors of childhood cancer have 11 times as high a mortality rate as the general population (9). Higher mortality due to other types of cancer and cardiovascular disease persists more than 25 years after the start of treatment. This underscores the need for evidence-based follow-up after treatment of children and adolescents with cancer (10).
Many survivors of childhood cancer do not receive targeted long-term follow-up, and few are aware of the risk of late effects of the disease (11, 12). A recent Norwegian study of long-term survivors of malignant lymphoma in childhood and/or adolescence confirmed that the patients lacked knowledge of the possible late effects of their treatment (13).
Our study concentrates on long-term survivors of acute lymphoblastic leukaemia (ALL), who comprise a third of the cases of childhood cancer (14, 15). The clinical view has long been that this group only suffers limited late effects, particularly since prophylactic radiation of the central nervous system (CNS) has been dropped from first-line treatment (16). However, recent studies show that there is also an increased incidence of morbidity and mortality among long-term survivors of ALL. Subjects who have received radiotherapy are the biggest risk group, but treatment of ALL without radiation also increases the risk of treatment-related late effects (17, 18). The hypothesis for our study was that adults who had been treated for ALL as children or adolescents knew little about the possible late effects of the disease.
Material and method
Study population
From June 2009 to July 2011, adults who had been treated as children or adolescents for ALL were invited to take part in the study ALLBARN at Oslo University Hospital.. The study, including subsidiary studies, was approved by the Regional Committee for Medical and Health Research Ethics (REK) and by the Data Protection Officer at Oslo University Hospital.
The Cancer Registry identified relevant participants on the basis of the following criteria:
-
Acute lymphoblastic leukaemia diagnosed in the period 1970 – 2002
-
Age < 16 years at the time of diagnosis
-
Age at the time of the survey > 18 years
-
At least five years since termination of cancer treatment
-
Treated at Oslo University Hospital, Rikshospitalet or Ullevål
A total of 210 persons were alive and satisfied the selection criteria. The study included 160 participants after informed consent had been obtained, and 140 came for a clinical examination (Fig. 1).
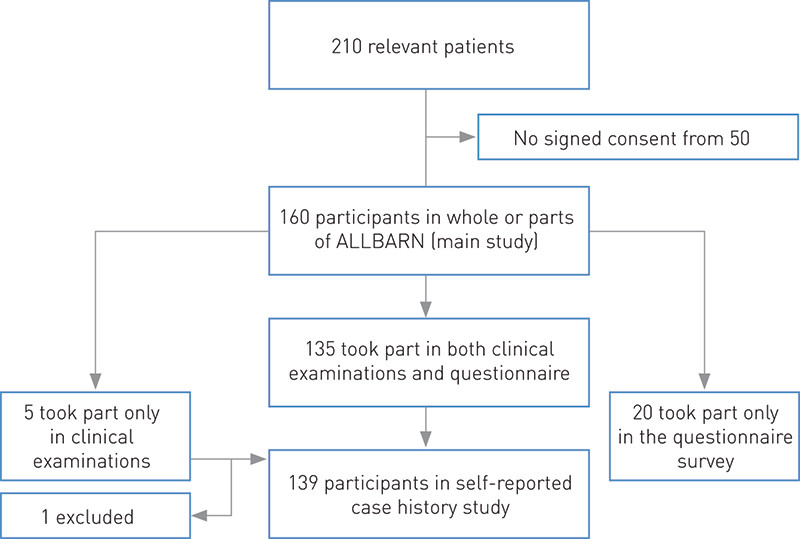
The self-reported case history study
In connection with a clinical examination, all participants in the ALLBARN study were interviewed by a doctor who kept records about their diagnosis, treatment and the risk of late effects after treatment of ALL. This part of the study was titled the Self-reported case history study (Egenanamnesestudien). The interview guide is available as an appendix to this article, see here. It is based on similar studies carried out previously (11) – (13). Patient data on cancer diagnosis, treatment details, identified late effects of treatment and further follow-up were obtained from the patient records at Oslo University Hospital, Ullevål or Rikshospitalet, and used as a basis for comparison with the participants’ responses in the interview.
Data were processed in the statistical dataprocessing and analysis tool PASW Statistics 18.0, and the results are presented descriptively.
Results
Demography
139 adults were included in this substudy. Table 1 shows demographic data on age and follow-up. Most participants were treated at Oslo University Hospital, Rikshospitalet (n = 110). The remaining 29 had had treatment at the Department of Paediatric Medicine at Oslo University Hospital, Ullevål.
Table 1
Demographic/clinical data for 139 adults treated for acute lymphoblastic leukaemia as children. Number (%) unless otherwise specified
| Selection | |
| Median age on diagnosis (range) | 5 (0 – 15) |
| Median age on examination (range) | 28 (18 – 46) |
| Median observation time (range) | 23 (7 – 40) |
| Gender | |
| women | 68 (49) |
| men | 71 (51) |
| Education | |
| ≥ Tertiary | 62 (45) |
| < Tertiary | 77 (55) |
| Treatment | |
| prophylactic radiation of CNS | 20 (14) |
| treatment for recurrence | 20 (14) |
| anthracyclines | 106 (76) |
| Received summary of treatment | 12 (9) |
| Continuous follow-up for long-term effects | 26 (19) |
| Long-term effects detected before inclusion in the study | 32 (23) |
Previous treatment
All those included had had chemotherapy, either as their only treatment (n = 120) or in combination with radiotherapy, surgery (testicular ablation) or stem cell transplantation. Fourteen patients had a high cumulative dose of anthracyclines (> 300 mg/m2), 20 had had prophylactic radiation of the central nervous system, three had undergone allogenic haematopoietic stem cell transplantation (bone marrow transplantation) and 20 had experienced ALL recurrence.
Knowledge of previous treatment
All patients knew that they had had leukaemia and how old (+/– 1 year) they had been at the time of diagnosis. Two participants did not remember that they had had chemotherapy, and some of them mistakenly believed that they had had radiotherapy (n = 5) or stem cell transplantation (n = 5). All those who had received stem cell transplants were aware of it, while three patients did not know they had had radiotherapy.
Late effects
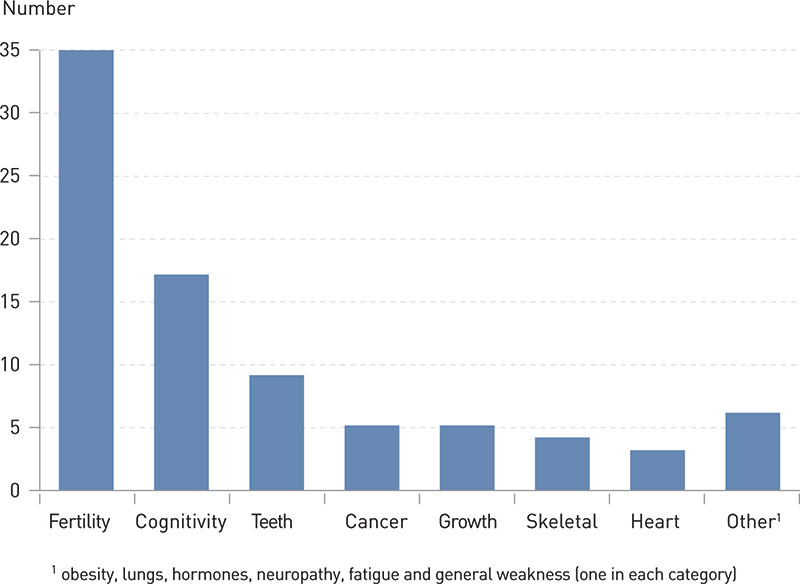
According to the patients’ own information in the interview and/or patient records from prior to inclusion in the ALLBARN study, 107 of the participants (77 %) had no recognised sequelae due to the treatment. None were diagnosed as having secondary cancers. The most common problems according to the records or the patient’s own reporting were reduced fertility (n = 10), impaired cognitive function (n = 6), reduced growth (n = 6) and skeletal necrosis (n = 5).
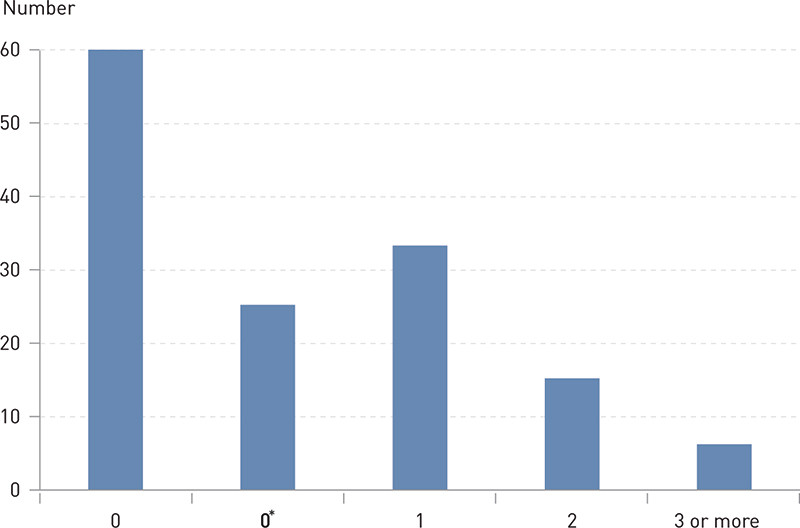
The distribution of level of knowledge of late effects is illustrated in Fig. 2. Knowledge of the risk of late effects varied considerably within the group. 60 participants (43 %) did not know that treatment of ALL in children could result in treatment-related sequelae. Twenty five participants (18 %) knew that treatment of childhood cancer could have effects in the longer term, but did not know of any examples of such effects. The remainder were able to give one or more examples of potential late effects. Reduced fertility was the best known sequela, and Fig. 3 illustrates the number of study participants who reported different types of late effects.
Follow-up
Twelve patients (9 %) remembered that they had received a written summary of their own treatment (Table 1). Twenty-six patients (19 %) reported that they had been followed up on account of a health problem after the termination of scheduled follow-up (10 years post end of treatment) by the department responsible for their treatment, while 113 (81 %) found that they were neither in nor had been in a structured follow-up system after the completion of standard outpatient follow-up at the regional hospitals (median 12 years of standard follow-up).
Discussion
In this cross-sectional study of adults who had received treatment for ALL during their childhood or adolescence, we found that knowledge of the risk of late effects after cancer treatment was limited. Whereas over half of the participants (61 %) were unable to cite examples of late effects of the treatment, they were well updated on their diagnosis and treatment.
Strengths and weaknesses
Oslo University Hospital, Rikshospitalet and Ullevål, has been and is responsible for the treatment of 50 % of all Norwegian children with cancer. We therefore assume that about half of all Norwegian survivors of ALL in their childhood or adolescent years who were treated in the period 1970 – 2002 were invited to take part in the ALLBARN study. The self-reported case history study included 66 % of these, i.e. about one third of all relevant subjects in Norway. Although this is a limited selection, we believe that the results can provide an indication of the general level of knowledge of late effects, but of course the group that did not take part in the study could have affected the outcome. However, no detailed non-response analyses were carried out on the patient material.
Another limitation is the quality of the medical records obtained from the period prior to inclusion in the ALLBARN study. These records were often deficient and difficult to find. A prospective system for reporting late effects, for example a public register of late effects, would have picked up more valid background information, but no such register is established in Norway. This may be one of the reasons why there were fewer patients with recognised late effects in the group we studied than have been reported by others (5). The fact that the participants were not systematically examined for late effects at the time of the self-reported case history study, and that ALL does not result in late effects that are as visible as in many other childhood cancers, are other possible reasons for the low prevalence of late effects in the material.
Knowledge about late effects
It has long been known that prophylactic radiation of the central nervous system in cases of ALL can result in long-term cognitive and endocrinological effects (17, 19), and in most international treatment protocols the use of prophylactic radiation for ALL is avoided (20). Other long-term effects following treatment of ALL with chemotherapy alone are regarded as modest, however, and the general view of many paediatric oncologists has been that the sequelae are negligible. The health service has not been sufficiently aware that the treatment of ALL in children without the use of radiotherapy may also be associated with late cardiological effects (21), secondary malignancy (17, 22), increased risk of novel neoplasms (8, 23, 24), cognitive functional impairment (25), reduced fertility (26), fatigue (27) and obesity (28). The result is lifelong higher mortality for the patient group compared with the general population (29).
This is relatively recently acquired knowledge, and there are still many late effects that have not been sufficiently explored. Many maintain that patients with ALL are less affected by their earlier cancer therapy than patients with many other types of malignancies (e.g. brain tumour), and this claim may be correct. However, it does not alter the fact that children and adolescents who have been treated for ALL are also subject to late effects.
Comparison with a similar Norwegian study
Our findings are consistent with the results of Hess et al., who carried out a similar Norwegian study among adults treated for lymphoma during childhood and adolescence (13). The authors found a significant difference in the knowledge of late effects, depending on the decade in which the patient had been treated, with growing knowledge among the youngest subjects who had been treated in the last decade. This difference was not as distinct in our study (data not shown).
Fertility, cardiovascular disease and cancer
Reduced fertility was the most common late effect the study participants knew of. This is surprising, because no studies have demonstrated a clear connection between treatment of ALL based on chemotherapy alone and seriously reduced fertility (26, 30). However, more intensive treatment with stem cell transplants, surgical removal of the testis (testicular ablation) or radiotherapy of the testis/prophylactic radiation of the central nervous system increase the risk of infertility. But this supplementary treatment would only have been relevant for a minority of the patients with ALL (n = 21). Very few knew of the increased risk of developing subsequent neoplasms (n = 5) and cardiovascular disease (n = 3), and we think these are alarming findings. Knowledge of these serious late effects is important in order for long-term survivors to follow appropriate advice for maintaining a healthy lifestyle and medical awareness.
Portioning out information of this kind so that it benefits the patients is a challenging medical balancing act. Information about possible late effects has to be balanced so that it does not turn patients into invalids, but represents useful information that increases the ability of long-term survivors to nurture their own health. There should be greater focus in research on developing satisfactory methods of communication, because late effects of cancer therapy will be a growing problem as the numbers of long-term survivors of cancer increase.
Follow-up
It is important to determine which system is most suitable for following up Norwegian children with cancer. There are various models, based on centralised follow-up at special clinics, decentralised follow-up by the primary doctor, or a combined solution of primary doctor and specialised clinics (31, 32). Many maintain that there should be a multi-level system that covers late effects of different degrees of severity, with the general practitioner as coordinator. Earmarked funding is required under any circumstances to build up a smoothly functioning system. The implementation of national follow-up plans and recommendations is a prerequisite for success in this important work (33, 34).
We must look at what other countries have established – and learn from it. A number of countries have developed national follow-up plans based on an evaluation by a panel of experts of available literature on risk factors. There is ongoing international harmonisation of guidelines which Norwegian survivors of childhood cancer can benefit from.
Conclusion
Many adults treated for ALL in childhood and adolescence know little of the risk of late effects. There are various models for long-term follow-up, and we believe that steps should be taken to develop appropriate strategies in Norway.
Tabell
Main points |
|
- 1.
Kreftregisteret. Kreft i Norge 2009 – Kreft insidens, mortalitet, overlevelse og prevalens i Norge. Oslo: Kreftregisteret, 2011.
- 2.
Howlader N, Noone AM, Krapcho M et al. SEER Cancer Statistics Review 1957 – 2009 (Vintage 2009 Populations). http://seer.cancer.gov/csr/1975_2008/ (15.9.11).
- 4.
Hewitt M, Weiner SL, Simone JV, red. Childhood cancer survivorship: improving care and quality of life. Washington, DC: National Academies Press, 2003.
- 14.
Kolmannskog S, Flaegstad T, Helgestad J et al. Akutt lymfatisk leukemi hos barn i Norge 1992-2000. Tidsskr Nor Lægeforen 2007; 127: 1493-5. [PubMed]
- 25.
Moleski M. Neuropsychological, neuroanatomical, and neurophysiological consequences of CNS chemotherapy for acute lymphoblastic leukemia. Arch Clin Neuropsychol 2000; 15: 603 – 30. [PubMed]
- 34.
Landier W, Bhatia S, Eshelman DA et al. Development of risk-based guidelines for pediatric cancer survivors: the children's oncology group long-term follow-up guidelines from the children's oncology group late effects committee and nursing discipline. J Clin Oncol 2004; 22: 4979 – 90. [PubMed] [CrossRef]