Red blood cell transfusion at Ullevål University Hospital – indications, consumption and blood group immunisation
Material and method
Study design and consents
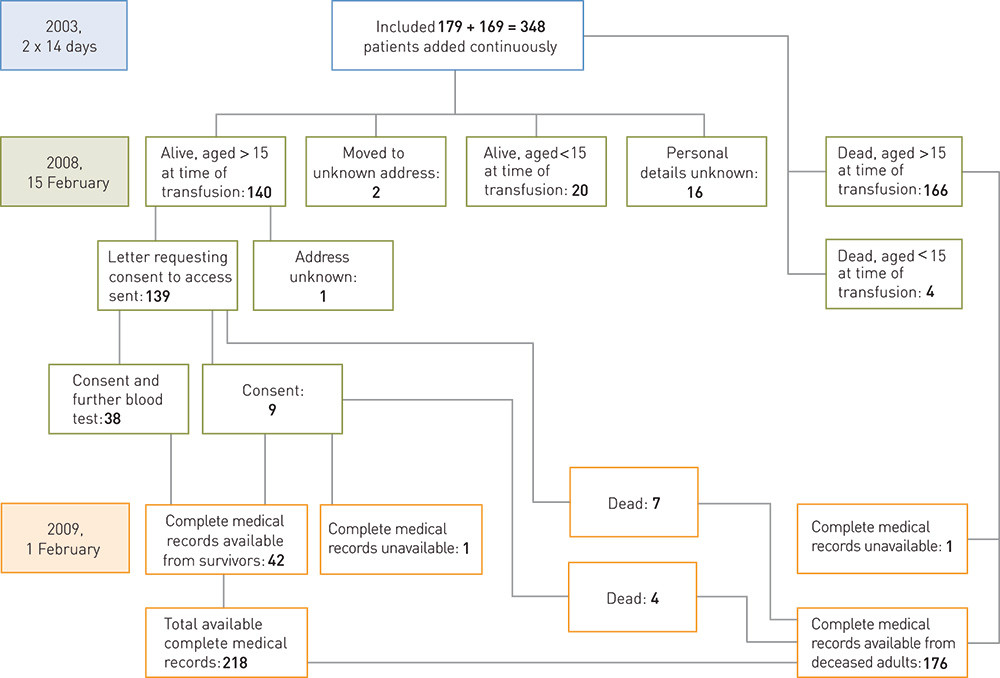
All patients who received transfusions at Ullevål University Hospital in the periods 1 – 14 May and 1 – 14 September 2003 were included. The two recording periods comprised 7.6 % of the year 2003, and were chosen because it was assumed that they were representative of the ordinary running of the hospital. The study design is shown in Figure 1. The study was approved by the Regional Ethics Committee for the Eastern Norway Regional Health Authority in 2003.
The numbers of transfusion episodes and transfused units of red blood cell concentrate in the two periods were recorded for the whole cohort in 2003 and processed in pseudonymised form. Occurrences of irregular blood group antibodies before transfusion were stored in pseudonymised form.
In 2006, the Norwegian Directorate of Health gave its consent to applications to access the records of patients who were registered in the National Population Registry as having died and who had been over 15 years old at the time of the transfusion.
As per 15 February 2008, the Population Registry was consulted regarding patients who were registered as having died. At the end of February 2008, a letter was sent to all assumed survivors who had been aged over 15 at the time of the transfusion. The letter contained a request for consent to access the records, and the recipient was asked to give a new blood sample for examination for irregular blood group antibodies. Reminders were not sent. The Population Registry was also consulted with respect to patients who had died when all the data was compiled as at 1 February 2009.
Testing for occurrences of irregular blood group antibodies in patients who gave a new blood sample in 2008 was carried out as pretransfusion testing, using the same method that was used in 2003. The results were stored in pseudonymised form.
Review of patient records
All records to which access had been consented were reviewed by one of the authors (IN). All data collection was terminated as at March 2009. ICD 10 diagnosis, surgical interventions, transfusions and patient notes about transfusion and the occurrence of irregular blood group antibodies prior to transfusion and according to a new survey in 2008 were registered and stored in pseudonymised form.
Statistics
The Mann-Whitney U test was used to test the difference between the medians of two groups with non-normal distribution. The chi-square test and the chi-square test for trends were used to test proportions.
Results
Patient characteristics at the time of transfusion
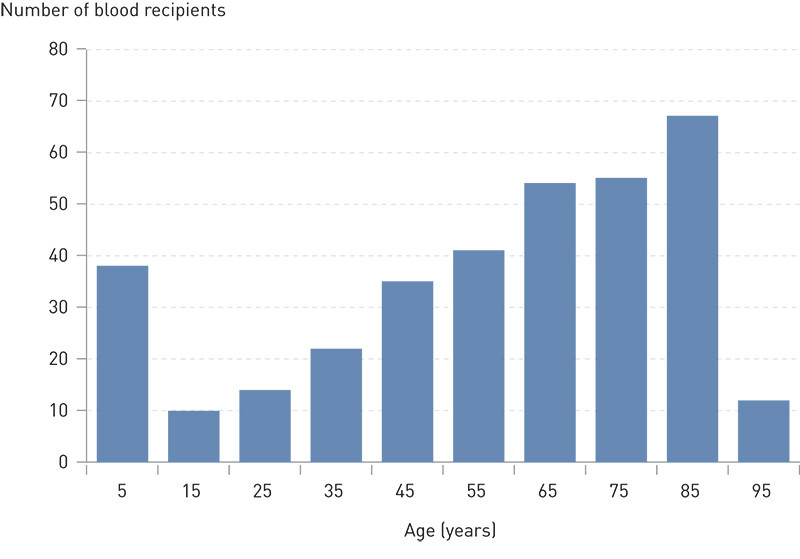
During the two recording periods, red blood cell transfusions were given to 179 and 169 patients, respectively, a total of 348. There were 178 women and 170 men. The age distribution is shown in Fig. 2 – the median age was 62.8, the spread 0 – 99 years. 188 patients (54 %) were aged 60 or over; there were 38 (11 %) in the age group 0 – 10.
Survival and access to patient records
Fig. 1 is an overview of developments in the study and the patient population. As at 15 February 2008, 170 (49 %) were registered in the Population Registry as having died, 97 of them men and four who were less than 15 years old at the time of the transfusion. There were insufficient personal details about 16 patients to find them in the Population Registry, and two had moved abroad. Letters were not sent to 20 survivors who were less than 15 years old at the time of transfusion. One patient aged over 15 was not sent a letter, due to a mistake. Thus letters were sent to 139 assumed survivors. Replies were received from 47, all giving consent to access to their records.
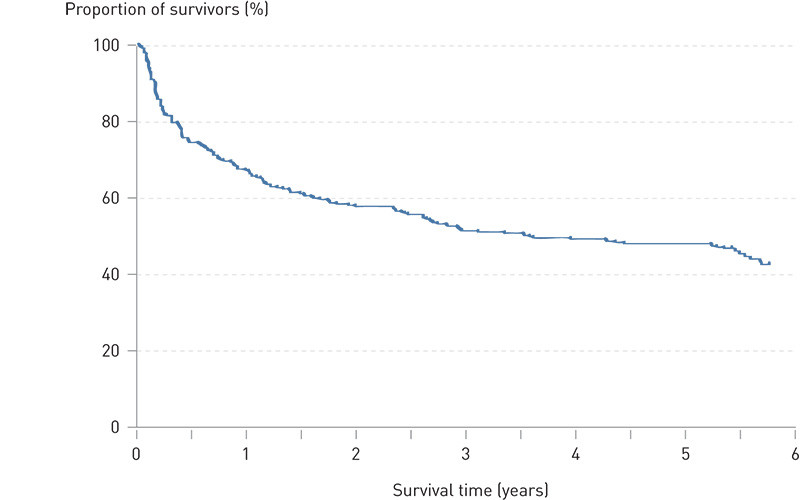
A search in the Population Registry at the beginning of March 2009 found a further 11 patients registered as having died. Four of them had given consent to access to their records. This brought the number of patients registered as having died to 181 (52 %). Complete records were available for 218 of the 220 who had given consent to access. Crude survival for up to 5 3/4 years, based on the number actually registered as dead, is shown in Fig. 3.
Diagnoses and mention of the transfusion in patient records
The primary ICD 10 diagnosis was found for 176 deceased and 42 surviving patients, 218 in all, as at 1 February 2009 (Table 1).
Table 1
ICD 10 diagnoses on discharge of 218 patients where access to records was possible
| Diagnostic group (ICD 10 diagnostic code) |
No. (%) |
Dead |
Surviving |
| Cancer (C00 – 97) |
76 (34, 9) |
73 |
3 |
| Injuries (S00–T98) |
36 (16,5) |
23 |
13 |
| Cardiovascular disease (I20 – 89) |
34 (15,6) |
27 |
7 |
| Diseases of the oesophagus (K22 –93) |
19 (8,7) |
17 |
2 |
| Anaemias (D50 –64) |
12 (5,5) |
11 |
1 |
| Diseases of the musculoskeletal system (M) |
9 (4,1) |
3 |
6 |
| Obstetrics (O) |
3 (1,4) |
0 |
3 |
| External causes |
29 (13,3) |
22 |
7 |
| Total |
218 (100) |
176 |
42 |
There were no notes on transfusion in 59 (27 %) of the 218 records to which we gained access (Table 2). Haemoglobin concentration prior to transfusion was recorded for significantly more patients with non-surgical diagnoses (ICD 10 groups A-C, I-K, N) than for patients with surgical diagnoses (ICD 10 groups M, O, S, T) (chi-squared test p < 0.001). Fifty-two (24 %) patients had a haemoglobin concentration prior to transfusion of ≥ 8 g/100 ml.
Table 2
Medical records and their contents concerning indications in 218 complete patient records
| ICD 10 diagnostic codes M, O, S, T |
Other ICD 10 diagnostic codes |
|
| No record or only mentioned transfusion¹ |
24 |
35 |
| Bleeding, reduced general condition |
12 |
28 |
| Numeric pretransfusion haemoglobin values listed² |
13 |
106 |
| Total |
49 |
169 |
| [i] | ||
[i] ¹ Of which 46 entirely without note
² Of which 52 with Hb ≥ 8 g/100 ml
Consumption of red blood cell concentrates
A total of 1 162 units of red blood cell concentrate were transfused in 471 transfusion episodes. This accounted for 8.3 % of the annual consumption at Ullevål University Hospital (unpublished statistics from the Oslo Blood Bank). No patients received transfusions in both recording periods.
One unit was administered in 83 episodes (18 %) and two units in 290 (62 %). Only one transfusion episode was recorded for 276 (79 %) of the patients. Of these, 219 (79 %) received one or two units. The maximum number of transfusion episodes was ten. Significantly more units of red blood cell concentrate were given at the first transfusion episode to patients who received more than one transfusion than were given to those only registered with one episode (chi-squared test for trend p < 0.001).
The median number of transfused units in the recording period was two (spread 1 – 32). Exchange transfusions were given to three neonates (one unit to each).Massive transfusion, defined as ten or more units in the course of 24 hours, was administered to a total of five patients (1.4 %).
Transfusion in connection with surgery
For 89 (41 %) of the 218 patients whose records were available, transfusion took place in connection with surgery performed during the recording period. These 89 patients (26 % of all those in the study) were given a total of 449 units of red blood cell concentrate (39 % of the total number of units), distributed among 157 transfusion episodes (33 % of the total number). The number of transfusion episodes and the number of transfused units were thus somewhat higher in relation to the number of patients for those who had undergone surgery compared with those who had not, but the differences were not statistically significant (Mann-Whitney U test p = 0.057 and p = 0.099).
Table 3 shows red blood cell consumption in connection with the various groups of interventions. Consumption was significantly higher in relation to the number of patients in connection with cardiovascular surgery than in connection with musculoskeletal surgery (Mann-Whitney U test p < 0.05).
Table 3
No. of units red blood cell concentrate administered with different types of surgical intervention
| Minimum |
Median |
Maximum |
Number of patients |
|
| Neurosurgery |
2 |
3 |
14 |
9 |
| Heart and major thoracic vessels |
1 |
4 |
60 |
16 |
| Other thoracic interventions |
2 |
5 |
7 |
10 |
| Gastrointestinal tract |
1 |
2 |
19 |
14 |
| Muscoloskeletal system |
1 |
2 |
10 |
22 |
| External causes |
1 |
2 |
8 |
18 |
| Total |
1 |
3 |
60 |
89 |
Prevalence of blood group antibodies
In pretransfusion tests in 2003, irregular blood group antibodies were found in 11 of 348 patients (3.2 %).
Of the 47 survivors who gave consent to access to their records, 38 gave blood samples for a further pretransfusion test. Irregular antibodies were detected in blood samples from three of them in 2003, and the same antibodies were detected in 2008. Of the other 35, anti-Rh(D), anti-Rh(C), anti-Rh(E) and anti-K were detected in one patient (immunisation rate = 2.6 %).
Discussion
Age
Elderly patients made up the majority of those who received transfusions. Similar findings have been reported from several Western countries (10) – (12, 14) – (17). It is reasonable to assume that elderly people also predominate among transfusion recipients in Norway as a whole, but this must be confirmed in more wide-ranging studies. The relatively large number of young patients can be partly attributed to the fact that Ullevål University Hospital had the largest paediatric department in Norway, with a separate centre for children with cancer.
Diagnoses
More than 50 % of the patients had the primary diagnosis of cancer or other chronic disorders with an uncertain prognosis. This is close to what is reported from other countries (14, 15, 18, 19). The figure is also consistent with the fact that about 60 % of the patients received transfusions that were not related to surgery, and that more than one third of the patients were dead one year after the transfusion. It can therefore be assumed that many received a transfusion as part of palliative treatment.
A regional study from France reported that almost 50 % of the transfusion recipients had cancer (11). From the Netherlands, 22 % were reported to have a cancer diagnosis (19). Our figures may be higher than the Norwegian average because Ullevål University Hospital had a large cancer centre, but there is reason to believe that cancer patients also constitute a large group of transfusion recipients, perhaps the largest, in Norway. It will be of considerable interest to obtain more definite data about this to enable us to assess the country's blood requirements in coming years.
In Denmark and Sweden (16) and in the Netherlands (20) mortality among transfusion recipients has been found to be significantly higher than mortality among the age-adjusted normal population for as long as 17 years (16) and ten years (20) after transfusion. This is consistent with the fact that transfusion, also to long-term survivors, is often given to those with life-shortening diseases. This supports our interpretation. Our survival data are similar to those presented by Kamper-Jørgensen et al. (16) and Borkent-Raven et al. (20).
Patient notes about transfusion
A substantial number of transfusions were given without any reason being recorded in the patient's notes. Transfusion is only supposed to be used when other treatment will not achieve the desired aim, within a reasonable time period and use of resources; it is not intended as therapy that is used regularly without further deliberation (1). Our findings may indicate that transfusion is not infrequently viewed as a routine measure that does not require a note in the patient's records.
Number of red blood cell concentrates per transfusion episode
The majority of patients received one or two units of red blood cell concentrate, and most of them did not receive a further transfusion during the recording period. In a normovolemic, haemodynamically stable person weighing 70 kg there will be an increase of just under 1 g/100 ml in Hb concentration per red blood cell concentrate (1, 21). The evidence base for red blood cell transfusion is generally inadequate (22), most of all in a palliative context. There is reason to question the usefulness of raising the Hb concentration by 1.5 – 1.8 g, particularly in normovolemic, non-bleeding patients with an Hb concentration of > 8 g/100 ml. Patients with symptomatic coronary disease constitute a possible exception (23, 24). Our findings therefore indicate that a number of transfusion episodes involving 1 – 2 units could have been avoided without major problems for the patient, and that there is accordingly a significant potential for saving on use of red blood cells. In order to obtain more reliable Norwegian figures on this point, similar studies should be conducted at more hospitals.
We found a tendency for patients who received three or more red blood cell units at the first registered transfusion episode to receive further transfusions more frequently than those who only received 1 – 2 units. It is reasonable to suppose that the former more often received transfusions as part of more intensive treatment than the latter.
Blood group immunisation
A prospective English study in 1996 showed that transfusion led to detectable blood group immunisation nine months later in about 8 % of those who received transfusions in connection with elective surgery (25). Our figures are too small for further analysis, and the apparently low immunisation rate may be due to some induced antibodies no longer being detectable. A prospective study is indicated to investigate whether blood group immunisation occurs more rarely with today's Norwegian transfusion practice than has previously been described from abroad.
Limitations of the study
The study does not represent a full description of clinical transfusion practice at Ullevål University Hospital; the recording periods are too short for that, and the number of patients too low. We would particularly like to point out that an unknown number of patients may have received transfusions or undergone surgery in advance of or subsequent to the two recording periods. We also lack access to clinical data and intervention codes for more than a third of the patients, including all those who were under the age of 15 at the time of the transfusion. Finally, there are 16 patients with unknown details (transfusion administered on an emergency person identification number) that it has not been possible to track in the Population Register. The number who have died may therefore be higher than we have been able to register.
On the other hand, the study covers 7.6 % of the year 2003 and 8.3 % of the consumption of red blood cells at Ullevål University Hospital, and all transfusions in the recording period are included. We also had access to clinical data for almost two third of the patients (62.6 %). We therefore believe that, given the aforementioned reservations, the study can be described as a «representative short-term picture» of transfusion activities at Ullevål University Hospital in 2003. Extrapolating the results to Norway as a whole is more uncertain, but our study was conducted at a hospital that covers all somatic specialities and has yielded findings similar to those reported internationally. It is therefore reasonable to suppose that the findings do not represent any major deviation from the situation at Norwegian hospitals generally.
Comparison with other countries
Only 1.4 % of the transfusion episodes involved ten or more units («massive transfusion»). At Copenhagen's Rigshospitalet, which has a trauma centre about the same size as Ullevål University Hospital had, 5.6 % of the transfusion episodes in 2009 were «massive» (26). This may reflect a generally more restrictive transfusion culture in Norway than in Denmark, which consumes 60 % more red blood cell concentrates annually in relation to the population than Norway does (3). The high consumption there is hardly positive for the patients (27). At the same time, red blood cell consumption in France is approximately 10 % lower in relation to the population than consumption in Norway (3). Consumption in the Netherlands is also substantially lower than in Norway (3).
The differences between the countries cannot be explained on the basis of a patient age or diagnosis spectrum, and no published data indicates that the differences result in a measurable effect on survival or quality of life. It is therefore reasonable to suppose that Norway has a not insignificant potential for saving blood, despite the fact that our consumption is lower than in the other Nordic countries (3).
Measures for optimising practice
We recently found that the consumption of red blood cell concentrates for multi-trauma patients at Ullevål University Hospital was reduced by about 25 % from 2002 to 2007 as a consequence of the introduction of stringent transfusion protocols (28). The reduction had no effect on the patients’ haemoglobin concentration. There is reason to assume that the introduction of more protocols of this type may result in reduced consumption of red blood cell concentrates in other parts of the hospital as well. Another means of encouraging saving might be for doctors who order a transfusion to be required to state the diagnosis and haemoglobin concentration when this is possible. At present these data are not communicated to the blood bank.
In our view, patients’ notes specifying the haemoglobin value should be a minimal requirement that is only waived in acute situations. Such a requirement might have a disciplinary effect on the use of indications and contribute to restricting consumption.
From 2000 to 2008, Norway's use of red blood cell concentrates increased by about 14 % (3). In years to come, there will be more elderly people (29), so a continued rise in consumption can be expected. However, Norwegian consumption has stagnated since 2008 (3). In the Netherlands, consumption of red blood cell concentrates has declined by almost 20 % since 1998, despite the fact that it was considerably lower than Norwegian consumption at the outset (3, 30). It seems reasonable to suppose that the changes reflect a tendency towards a more restrictive transfusion practice in both countries (2, 30, 31).
Conclusion
Red blood cell transfusion is given most often to elderly patients with chronic diseases with an uncertain or poor prognosis, particularly cancer. The consumption pattern is the same as that reported from other Western countries. Consumption is generally highest for patients who undergo surgery. The clinical documentation is not infrequently incomplete. In some cases the use of indications appears to be based on routines and to too limited an extent on scientific evidence and pathophysiological understanding.
We assume that Norway as a whole has a similar consumption pattern and that there is potential for saving on the use of red blood cell concentrates. New data imply that consumption countrywide is stagnating and that the situation is thus improving. As a rule, red blood cell transfusion does not lead to blood group immunisation.
Tabell
| Main points |
|
- 1.
Håndbok i transfusjonsmedisin. www.helsedirektoratet.no/publikasjoner/handbok-i-transfusjonsmedisin/Sider/default.aspx (27.12.2011).
- 2.
Engoren M. Transfusion: is merely not bad good enough? Acta Anaesthesiol Scand 2011; 55: 907 – 9. [PubMed]
- 3.
Flesland Ø, Sjøberg JJ. Blodtransfusjonstjenesten i Norge: statistikk for 2010 (Norwegian Transfusion Statistics 2010). www.hemovigilans.no/Dokumenter (27.12.2011).
- 4.
Heier HE. Produksjon og forbruk av blodprodukter i Norge. Tidsskr Nor Lægeforen 1993; 113: 18 – 22.
- 5.
Sirchia G, Giovanetti AM, McClelland DBL et al. Safe and good use of blood in surgery. Use of blood products and artificial colloids in 43 European communities. Luxembourg: Office for Official Publications of the European Communities, 1994.
- 8.
Friedman MT. Blood transfusion practices: a little consistency please. Blood Transfus 2011; 9: 362 – 5. [PubMed]
- 9.
Liumbruno GM, Rafanelli D. Appropriateness of blood transfusion and physicians’ education: a continuous challenge for Hospital Transfusion Committees? Blood Transfus 2012; 10: 1 – 3. [PubMed]
- 13.
Heier HE, Hervig T. Mot permanent blodkrise i Norge? Dagens Medisin 10.10.2010.
- 15.
Cobain TJ, Vamvakas EC, Wells A et al. A survey of the demographics of blood use. Transfus Med 2007; 17: 1 – 15. [PubMed]
- 21.
Klein HG, Anstee DJ. Mollison’s Blood transfusion in clinical medicine. 11. utg. Oxford: Blackwell, 2005: 383.
- 24.
Klein HG, Anstee DJ. Mollison’s Blood transfusion in clinical medicine. 11. utg. Oxford: Blackwell, 2005: 386 – 7.
- 27.
Johansson PI, Titlestad K, Møller BK et al. Overtransfusion – en dansk uvane med alvorlige konsekvenser. Ugeskr Læger 2009; 171: 1072. [PubMed]
- 29.
Brunborg H, Texmon I. Befolkningsframskrivninger 2010 – 2060. Økonomiske Analyser 2010; nr. 4: 28 – 39.
- 31.
Reine PA, Kongsgaard UE, Smith-Erichsen N. Hemoglobingrenser og transfusjonspraksis blant norske anestesiologer. Tidsskr Nor Lægeforen 2004; 124: 2610–2. [PubMed]