The climacteric is the period immediately before and after the menopause, which marks the end of a woman’s fertile period. The menopause is a natural life phase, but in the 1960s and 70s it assumed the status of a deficiency disease that had to be treated with hormones. The publication of books such as Feminine forever in the US (1) and Alltid kvinne (Always a woman) (2) in Norway combined with proactive marketing by pharmaceutical companies contributed to this. The menopause was claimed to represent a «castration» of women that could be prevented by the use of hormones. As well as helping to retain youthfulness and sex appeal, hormone therapy was said to prevent depression, various kinds of afflictions, coronary heart disease and dementia in women. Scientific studies in the 1980s and 90s also supported the likelihood of this (3). Hormone therapy was known to prevent osteoporosis in an effective manner (4). The correlation between the use of oestrogen and increased risk of breast cancer was reported as early as 1976 (5), but it was the results of the Women’s Health Initiative study in the US (6) and the ensuing publicity that led to a clear decline in sales of oestrogen products. The randomised clinical study found a significantly greater risk of breast cancer in those receiving oestrogen and progestogen compared with the placebo group. The risk normalised when the therapy was discontinued. The British Million Women cohort study also confirmed the correlation (7).
A study from the «Women and cancer» cohort with data from 31 451 Norwegian postmenopausal women aged 45 – 64 showed that the relative risk (RR) of breast cancer was 2.1 (95 % CI 1.7 – 2.7) at the current level of oestrogen use. Follow-up of the cohort was undertaken from 1996 up to and including 2002 when the decline in sales commenced. The attributable risk, i.e. the percentage of breast cancer cases that can be attributed to oestrogen use was calculated at 27 %. Long-term use (> 5 years) of combination therapy resulted in the highest risk (RR 2.8; 95 % CI 2.0 – 4.0) (8). Norwegian figures show that the sale of hormone products for systemic use fell by over 45 % from 2001 – 05 and is now at the 1990 level (9). Figure 1 shows the sales curve for hormone products as well as the annual incidence of breast cancer among Norwegian women aged 45 – 64.
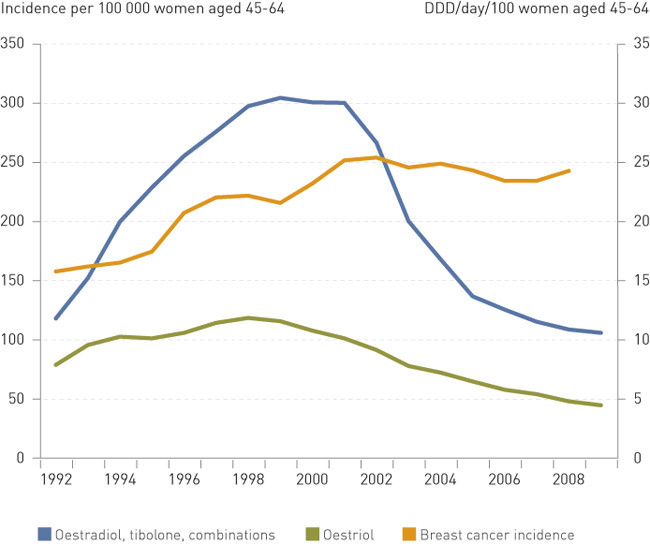
A study of the pattern of use among women in the «Women and cancer» study after 2002 shows a tendency to increased treatment length among existing users, while the recruitment of new users has decreased (10).
We conducted a nested case-control study based on the «Women and cancer» study with follow-up of breast cancer or cancer in situ from 2004 – 08. The purpose of the study was to calculate the risk of breast cancer and attributable risk for use of systemic hormone therapy. We also wanted to examine whether risk estimates have changed since 2003.
Material and method
«Women and cancer» is a prospective study of 172 000 randomly selected Norwegian women (11). They have responded to from one to three questionnaires about the use of hormones, their menstrual status, reproductive history, illness and cancer in the family as well as lifestyle habits such as diet, smoking, physical activity, alcohol consumption and sun exposure. The women are monitored continuously for breast cancer through the Cancer Register of Norway.
In connection with the study a biobank was established with blood samples of 50 000 women aged 46 – 63, collected in the period from 2003 – 06. The blood samples were obtained at the women’s local medical office, and sent by post together with a two-page questionnaire about menstrual factors, food intake, smoking, weight and height, use of medication, hormones and dietary supplements and alcohol intake (blood sample collection form) The biobank constitutes the post-genome part of the study and is described in more detail elsewhere (12).
From 2004 to the end of 2008 a total of 473 cases of invasive breast cancer and 116 cases of cancer in situ were registered in the post-genome cohort. For each case we selected a control who matched in age and the time of blood sampling. The background for this pair matching is a gene expression study with micro-matrix analysis of blood. Only postmenopausal women were included in the study (358 with invasive breast cancer, 83 with cancer in situ, 451 controls). Information from the blood sample collection form and previous questionnaires were linked together in a data set.
The «Women and cancer» study has been approved by the Northern Norway Regional Committee for Medical and Health Research Ethics. All samples and data have been stored and processed in accordance with the licence granted by the Data Inspectorate.
Menstrual status
The women’s menstrual status was determined on the basis of the information in the blood sample collection form and previous questionnaires. They were categorised as premenopausal if they answered «Yes» to the question of whether they menstruated; postmenopausal if they answered «No» or if they gave an unclear answer and were over the age of 53; and perimenopausal if they ticked «Irregular». Women who had had a hysterectomy, used hormones or gave an unclear answer and were under the age of 53 were categorised as «Uncertain».
Use of hormone therapy
Women who stated in the blood sample collection form that they had used a product containing oestrogen in the course of the previous week were considered to be current users. No distinction was made between sequential and continuous oestrogen. We checked the previous questionnaire to see whether the women had also used oestrogen products earlier. If the woman had replied «Yes» to the use of hormones during the menopause in any of the questionnaires, she was classified as using hormones «at some time». If the woman was classified as using hormones «at some time», but was not a «current» user, she was categorised as a «former» user. Information on age at the commencement of the therapy was used to calculate the total duration of use. We assumed that the women continued the therapy in the period between the two questionnaires. The length of time from the last questionnaire to the blood sample collection form was one year on average. Women who answered «No» in both questionnaires to the question on use were categorised as «never-users».
Data processing
Controls with the same year of birth and time of blood sampling were selected at random among the women in the cohort. Transformation of variables and logical regression were conducted with SPSS version 17.0. We adjusted for age, body mass index, age at menarche, breast cancer in mother, parity, age at first birth and earlier use of hormone therapy. All variables are expected to have an effect on the risk of breast cancer.
Results
A total of 892 women were included. Eight women who stated that they had used hormone therapy were excluded from the analyses because the name of the product was not mentioned. Table 1 provides the women’s characteristics.
Table 1
Characteristics patients and controls
| Patients (N = 441) |
Controls (N = 451) |
||||
|
|
N |
% |
N |
% |
|
| Average age in years |
56,8 |
56.6 |
|||
| BMI |
|||||
| < 25 kg/m |
219 |
49.7 |
196 |
43.5 |
|
| 25 – 29 kg/m |
164 |
37.2 |
179 |
39.7 |
|
| ≥ 30 kg/m |
51 |
11.6 |
67 |
14.9 |
|
| Unknown |
7 |
1.6 |
9 |
2.0 |
|
| Parity |
|||||
| 0 children |
41 |
9.3 |
35 |
7.8 |
|
| 1 – 2 children |
249 |
56.5 |
255 |
56.5 |
|
| ≥ 3 children |
150 |
34.0 |
161 |
35.7 |
|
| Unknown |
1 |
0.2 |
0 |
0.0 |
|
| Age at first birth |
|||||
| < 25 years |
234 |
53.1 |
259 |
57.4 |
|
| ≥ 25 years |
161 |
36.5 |
156 |
34.6 |
|
| Unknown |
5 |
1.1 |
1 |
0.2 |
|
| Age at menarche |
|||||
| < 12 years |
44 |
10.0 |
46 |
10.2 |
|
| ≥ 12 years |
387 |
87.8 |
401 |
88.9 |
|
| Unknown |
10 |
2.3 |
4 |
0.9 |
|
| Mother with breast cancer |
46 |
10.4 |
37 |
8.2 |
|
| Hormone therapy |
|||||
| Never |
164 |
37.2 |
202 |
44.8 |
|
| Earlier |
132 |
29.9 |
168 |
37.3 |
|
| Sometime |
277 |
62.8 |
249 |
55.2 |
|
| Now¹ |
145 |
32.9 |
81 |
18.0 |
|
| Oestradiol |
22 |
5.0 |
13 |
2.9 |
|
| Combination product |
99 |
22.4 |
45 |
10.0 |
|
| Tibolone |
21 |
4.8 |
17 |
3.8 |
|
| Oestriol |
2 |
0.5 |
5 |
1.1 |
|
| Progestogen |
1 |
0.2 |
1 |
0.2 |
|
| [i] | |||||
[i] ¹ Only systemic therapy is included
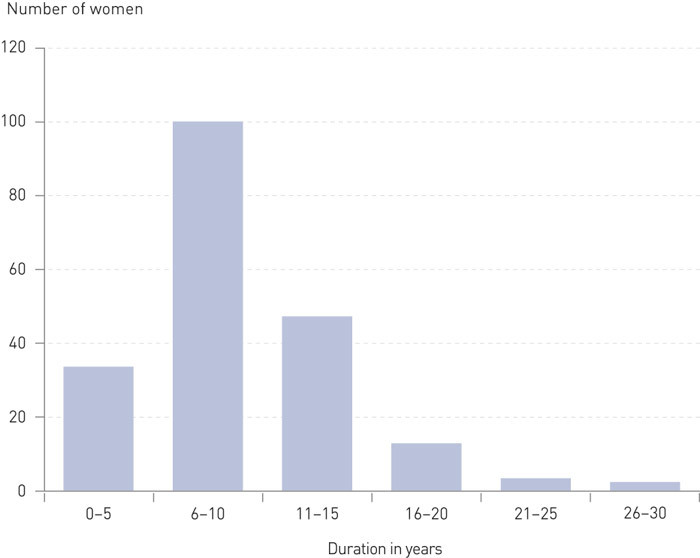
One-fourth of the women were current users. Of these 64 % used combination products. While 15 % used oestradiol alone, 17 % used tibolone. Of these users, 219 (97 %) were registered as users in the previous questionnaire. Of these, 192 had stated their age at the start of hormone therapy and the average duration of use was calculated at 9.6 (1) – (28) years. Figure 2 shows the distribution in duration among current users.
We found that the adjusted risk of breast cancer was significantly higher for current users than for never-users (odds ratio (OR) 2.1; 95 % CI 1.5 – 3.0). The risk was greatest for long-term (> 5 years) therapy (OR 2.4; 95 % CI 1.6 – 3.6) (test for trend, p-value 0.006, data not shown). Compared with never-users, those who used combination therapy had an odds ratio of 2.6 (95 % CI 1.7 – 4.0), and where the duration of use was over five years the ratio was OR 3.0 (95 % CI 1.9 – 4.7). Use of oestradiol or tibolone gave lower, non-statistically significant risk, OR 1.8 (95 % CI 0.9 – 3.8) and OR 1.5 (95 % CI 0.8 – 3.0), but this was based on a small number of users. Sometime-users generally had a somewhat increased risk (OR 1.3; 95 % CI 1.0 – 1.7), while earlier use did not give an elevated risk (Table 2). The results showed only an insignificant change when we looked at invasive breast cancer and carcinoma in situ combined and individually. The average follow-up time (the difference between the date of diagnosis and the date of responding to the questionnaire) was 1.9 (0 – 5) years. Risk estimates remained stable during the follow-up period (data not shown).
Table 2
Odds ratio (OR) with 95 % confidence interval (CI) for breast cancer¹
| Use of hormones |
OR (95 % CI) |
OR (95 % CI) adjusted² |
| Never |
1.0 (ref) |
1.0 (ref) |
| Sometime |
1.4 (1.1 – 1.8) |
1.3 (1.0 – 1.7) |
| Earlier |
1.0 (0.7 – 1.3) |
0.9 (0.6 – 1.2) |
| Now |
2.2 (1.6 – 3.1) |
2.1 (1.5 – 3.0) |
| ≤ 5 years |
1.5 (0.9 – 2.6) |
1.5 (0.9 – 2.7) |
| > 5 years |
2.6 (1.8 – 3.7) |
2.4 (1.6 – 3.6) |
| Combination products |
2.8 (1.9 – 4.1) |
2.6 (1.7 – 4.0) |
| ≤ 5 years |
2.1 (1.0 – 4.2) |
2.1 (1.0 – 4.4) |
| > 5 years |
3.1 (2.0 – 4.6) |
3.0 (1.9 – 4.7) |
| Oestradiol |
2.1 (1.1 – 4.3) |
1.8 (0.9 – 3.8) |
| Tibolone |
1.5 (0.8 – 3.0) |
1.5 (0.8 – 3.0) |
| [i] | ||
[i] ¹ 417 patients and 434 controls with completed data are included
² Adjusted for age, body mass index, age at menarche, breast cancer in mother, parity and age at first birth
Attributable risk in the population
We used the prevalence of current use of hormone therapy among the controls (18.1 %) and the odds ratio 2.1 as a risk estimate to calculate the attributable risk of breast cancer among women aged 47 – 63 in the period 2003 – 08. We found that 16.6 % of cancer cases could be attributed to treatment with products containing oestrogen. Based on the incidence rates from the Cancer Registry in the period 2003 – 08 this constitutes 232 cases of breast cancer annually in women aged 45 – 64.
Discussion
In our study, current users of hormone therapy had a 2 – 4 times higher risk of breast cancer than never-users. This corresponded with the earlier «Women and cancer» study. The risk was greatest with use of combination therapy for more than five years. The prevalence of use was halved from 2002 to 2008 and attributable risk declined correspondingly from 27 % to 16.6 %. Nonetheless, this means that 232 breast cancer cases annually in this age group could be attributed to hormone use. By way of comparison, the European Prospective Investigation into Cancer and Nutrition (EPIC) recruited a cohort of postmenopausal women from various European countries who were all monitored for breast cancer. It was found that current use of oestrogen and combination products gave increased relative risk: 1.42 and 1.77 respectively (13).
In our study the average time from responding to the questionnaire to diagnosis was less than two years. It is a weakness that the control group is not monitored over a longer period, because some may develop cancer later. Hormone therapy may stimulate the growth of cancer cells (14). This may be the reason why users receive an earlier diagnosis. Therefore our study does not in itself provide a basis for stating that the use of hormone products gives increased lifetime risk of breast cancer. However, hormone replacement therapy after the menopause is considered to be a known risk factor (15), and the combination oestrogen-progestogen is classified by the International Agency for Research on Cancer (IARC) as a carcinogen for breast cancer (16).
The study is a nested case-control study in which data on exposition have been collected prior to the cancer diagnosis. As a result, memory bias is of little relevance. We have not taken into account the fact that women may have changed to a different kind of product during the therapy. External validation of the «Women and cancer» cohort has shown that there are few sources of selection bias that prevent the generalisation of results to the female segment of the Norwegian population or preclude the calculation of attributable risk (17).
We chose to include patients who were diagnosed with carcinoma in situ. Invasive breast cancer and ductal carcinoma in situ appear to have the same risk factors and development mechanism (18) – (20). The analysis results only changed insignificantly when we excluded women with cancer in situ.
Studies from several countries show that the decline in the use of hormone therapy is followed by a decline in breast cancer incidence (21). Data from the US Surveillance, Epidemiology and End Results Program showed a decline in incidence from 1999 to 2003 for those over 45, corresponding to the saturation of mammography screening. A sharp drop was observed in the period 2002 – 03 for oestrogen receptor positive tumours in women aged 50 – 69, which may be due to the decline in the use of hormone therapy (22).
In Norway the incidence of breast cancer rose in the 1990s and then levelled off before falling slightly after 2002 (fig 1). The introduction of the Norwegian mammography programme led to the detection of a larger number of Stage 1 tumours, particularly in women aged 50 – 69 (23). Nevertheless, the increase in incidence cannot be explained merely as an effect of the screening itself (24). A recent Norwegian study has shown that the changes in incidence can be attributed almost equally to mammography screening and hormone therapy (25). Hormone therapy correlates with more mammographically dense breast tissue (26). Women who used hormone therapy or had used it recently had a significantly higher average density. Use of combination products and long-term use of these gave the highest density. Increased mammography density is an independent risk factor for breast cancer (27), and can also give false negative mammography findings (28). Mammography is therefore a poorer test for hormone users.
The average duration of use by the women in our study was almost ten years. It is a matter of concern that older women use hormone therapy and that they continue as users when we know that age is a key risk factor and that the risk of breast cancer increases for every year of hormone therapy.
According to a study with data from 2004 on Norwegian GPs’ attitudes to oestrogen therapy for women during and after the menopause, the majority of GPs were familiar with the effects and side-effects. Hot flushes were the most important indication and previous breast cancer and breast cancer in first-degree relatives were contraindications for therapy. The duration of therapy was longer than what is recommended. Moreover, the study indicated that Norwegian women doctors were more updated professionally, more often had a general rule for duration of treatment and administered treatment for a shorter period of time (29). The study also showed that Norwegian doctors have become more restrictive about prescribing hormone therapy but recommendations on use varied. A number of doctors would recommend hormone therapy in the case of minor, less troublesome hot flushes if the woman either had irregular bleeding or felt depressed, had skin ageing changes and was dissatisfied with intimate relationships. Most doctors would neither recommend nor advise against hormone therapy on the basis of the case reports presented. The older the doctor, the greater the tendency to recommend use (30).
Current guidelines state that hormone therapy is indicated when the woman’s afflictions are pronounced and lead to reduced life quality, alternative treatment has had no effect and additionally that the advantages of therapy are greater than the disadvantages (31, 32). Before hormone therapy is commenced, an individual assessment must be conducted with a thorough case history that focuses on the woman’s predisposition to hormone sensitive cancer forms and cardiovascular disease. The Norwegian Society for Gynaecology and Obstetrics has listed the contraindications for hormone therapy (33). In our view, other risk factors, such as high age, late menopause, overweight, late birth or nulliparity and high alcohol consumption should be included in the assessment. According to the Norwegian Medicines Agency the treatment should be reviewed annually. After 2 – 3 years the women must be fully informed of unwanted side-effects and together with the doctor in charge of the case balance the need for therapy against the risk. Hormone therapy must not be used in cases of menstrual irregularities (31).
Tibolone is a tissue-specific regulator with oestrogenic and androgenic properties, and is the latest of the hormone products that improve menopausal symptoms and prevent osteoporosis. Tibolone reduces the level of active oestrogen in breast tissue and breast cancer cells and has proved to have little effect on cell proliferation and breast tissue density (34). Hence use of tibolone might be expected to be more favourable. Some small studies have shown that tibolone reduces the risk of breast cancer (35), but large observational studies have shown the opposite. In both the Million Women and the EPIC studies an increased risk was found with the current use of tibolone, RR 1.45 (1.25 – 1.67) and RR 1.95 (1.43 – 2.65) (7). Until we have more extensive studies and more knowledge about long-term effects we cannot therefore recommend a switch to tibolone.
Conclusion
We found an average of ten years duration of use in established users of systemic hormone treatment. Long-term use of oestrogen and progestogen in combination tripled the risk of breast cancer. It is cause for concern that postmenopausal women continue the therapy.
The study shows that 232 cases annually of breast cancer in Norwegian women can be attributed to hormone therapy.
It is crucial that both doctors and women are well informed of the risk associated with treatment with systemic hormone products containing oestrogen. The commencement of therapy should be based on a thorough individual risk assessment and current guidelines should be followed. For some women who are badly affected by hot flushes, hormone therapy remains the most effective remedy we have.
Tabell
| Main points |
|
- 1.
Wilson R. Feminine forever. New York, NY: M. Evans & company, 1966.
- 2.
Myhre E. Alltid kvinne. Er overgangsalderen en mangelsykdom? Oslo: Cappelen, 1976.
- 9.
Sakshaug S, red. Legemiddelforbruket i Norge 2006 – 2010. Oslo: Folkehelseinstituttet, 2011.
- 11.
Universitetet i Tromsø. Velkommen til Kvinner og kreft. http://uit.no/ansatte/organisasjon/artikkel?p_document_id=150476&p_dimension_id=88111&p_menu=42374 (21.2.2012).
- 16.
The International Agency for Research on Cancer (IARC). Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. Genève: IARC, 2007.
- 17.
Lund E, Kumle M, Braaten T et al. External validity in a population-based national prospective study–the Norwegian Women and Cancer Study (NOWAC). Cancer Causes Control 2003; 14: 1001 – 8. [PubMed]
- 19.
Kerlikowske K. Epidemiology of ductal carcinoma in situ. J Natl Cancer Inst Monogr 2010; 2010: 139 – 41.
- 23.
Hofvind S, Sørum R, Haldorsen T et al. Brystkreftforekomst før og etter innføring av mammografiscreening. Tidsskr Nor Lægeforen 2006; 126: 2935-8. [PubMed]
- 27.
Garrido-Estepa M, Ruiz-Perales F, Miranda J et al. Evaluation of mammographic density patterns: reproducibility and concordance among scales. BMC Cancer 2010; 10: 485. [PubMed]
- 29.
Gjelsvik B, Swensen E, Hjortdahl P. Allmennlegenes syn på hormonbehandling i og etter overgangsalderen. Tidsskr Nor Lægeforen 2007; 127: 1500 – 3. [PubMed]
- 30.
Gjelsvik B. Allmennlegers råd om hormonbehandling i overgangsalderen. Tidsskr Nor Lægeforen 2008; 128: 1660-3. [PubMed]
- 31.
Statens legemiddelverk. Hormontilskudd til kvinner etter menopausen gir økt risiko for brystkreft. www.slv.no/templates/InterPage____16066.aspx (14.2.2012).
- 32.
Norsk legemiddelhåndbok. T14.3 Klimakteriske besvær. http://legemiddelhandboka.no/Terapi/søker/hormonbehandling/19076#i19126 (16.2.2012).
- 33.
Norsk gynekologisk forening. Klimakteriet 2010. http://legeforeningen.no/fagmed/norsk-gynekologisk-forening/veiledere/veileder-i-generell-gynekologi-2009/klimakteriet-2010/ (21.2.2012).
- 34.
Notelovitz M. Postmenopausal tibolone therapy: biologic principles and applied clinical practice. MedGenMed 2007; 9: 2. [PubMed]
I artikkelen "Risiko for brystkreft ved bruk av hormonbehandling i klimakteriet" har Sara Sæther, Kjersti Bakken og Eiliv Lund sett på sammenhengen mellom bruk av hormoner (østrogener og progesteron) i overgangsalder og risiko for brystkreft. Dette har de studert med en nøstet fall-kontroll studie med data fra Kvinner og Kreft (Norwegian Women and Cancer; NOWAC) studien (1). Artikkelen bekrefter tidligere funn for en sammenheng mellom bruk av hormoner og brystkreft.
I slike analyser er det viktig at man kjenner variabler som i tillegg til den man studerer, er assosiert med utfallet (såkalte konfunderende variabler). I dette tilfelle er utfallet risiko for brystkreft. Mammografiscreening er en kjent risikofaktor for brystkreft. Mammografiscreening øker forekomst av brystkreft ved at diagnosetidspunktet fremskyndes i tid og alder (såkalt lead time), samt ved overdiagnostikk. Andelen overdiagnostikk ved mammografiscreening i Norge ligger mellom 15 og 50% (2, 3, 4). Det er en åpenbar svakhet ved artikkelen til Sæther et al. at man ikke har tatt med informasjon om kvinnene har vært til mammografiscreening.
I Norge tilbys kvinner i alderen 50 til 69 år mammografiscreening hver annet år i regi av Mammografiprogrammet. Videre er det hevdet at det er stor grad av opportunistisk screening i Norge basert blant annet på data fra NOWAC-studien (5). Forfatterne har altså data som kan belyse sammenhengen mellom bruk av hormoner og brystkreft ved å ta hensyn til mammografiscreening (både i regi av Mammografiprogrammet og ved opportunistisk screening), som er en kjent konfunderende faktor.
Vi vil gjerne oppfordre forfatterne å belyse hvordan mammografiscreening påvirker deres analyser (både lead-time effekten og overdiagnostikk) og dermed sammenhengen mellom bruk av hormoner og brystkreft. Videre lurer vi på om det det er en assosiasjon mellom bruk av hormoner og mammografi. Mange som bruker hormoner i overgangsalder vil ta rutinemessig mammografi nettopp fordi det er en kjent sammenheng mellom hormoner og brystkreft. Hvordan har dette påvirket resultatene?
Litteratur
1.Sæther S, Bakken K, Lund E. Risiko for brystkreft ved bruk av hormonbehandling i klimakteriet. Tidsskr Nor Legeforen 2012; 132:1330-4
2.Kalager M, Adami HO, Bretthauer M, Tamimi RM. Overdiagnosisi of invasive breast cancer due to mammography screening: results from the Norwegian screening program. Ann Intern Med 2012; 156: 491-9
3.Zahl P, Mæhlen J. Overdiagnostikk av brystkreft etter 14 år med mammograifscreening. Tidsskr Nor Legeforen 2012; 132: 414-7.
4.Zahl PH, Strand BH, Mæhlen J. Incidence of breast cancer in Norway and Sweden during introduction of nationwide screening: prospective cohort study. BMJ 2004; 328: 921-4.
5.Lynge E, Braaten T, Njor SH, Olsen AH, Kumle M, Waaseth M, Lund E. Mammography activity in Norway 1983 to 2008. Acta Oncol 2011; 50: 1062-7