Spasticity is a relatively common phenomenon in patients with a disease or injury of the central nervous system, and is a sign of damage to upper motor neurons. Up to 70 % of patients with spinal cord injuries develop spasticity that may cause considerable disability (1) – (4). How the spasticity develops and the degree of spasticity depend on the location and extent of the injury (whether the injury is complete or incomplete). Spasticity may develop months or years after the acute injury and lead to increased loss of function and hospitalisation (3).
W. Lance defined spasticity as «a motor disorder characterised by a velocity-dependent increase in tonic stretch reflexes (muscle tone) with exaggerated tendon jerks, resulting from hyperexcitability of the stretch reflex» (5). The European working group EUSPASM has defined spasticity as «disordered sensorimotor control resulting from an upper motor neurone lesion, presenting as intermittent or sustained involuntary activation of muscles» (6). Upper motor neuron injury additionally causes paresis, impaired fine motor skills and control of movements and increased fatigue due to movements (6, 7).
The main aim of the article is to provide an overview of the pathophysiology, clinical picture and treatment of spasticity, primarily in patients with spinal cord injuries. The article may help doctors who are involved in the treatment of spasticity in patients with spinal cord injuries.
Method.
The paper is based on literature searches in PubMed using the keyphrases «spasticity» and «spasticity AND spinal cord injury», with the emphasis on clinical studies. Our assessments also rest on our own clinical experience and research at the Spasticity Management Clinic, Haukeland University Hospital.
Pathophysiology
The pathogenesis of spasticity in patients with SCI is multifactorial. A change in the excitability of various supraspinal inhibitory nerve paths used to be regarded as the main explanation. More recent research has shown a change in excitability in the actual motor neurons and interneurons as well (8). Spasticity may be general, regional or localised and associated with tetraparesis, hemiparesis, paraparesis or monoparesis. Spasticity may be caused by cerebral or spinal injuries (9). Spasticity is not a static phenomenon, and when untreated it can lead to secondary reorganisation of both the nervous system and the musculature (7). With serious spinal cord injury, paresis occurs which leads to adaptive shortening of muscles that change afferent input to the spinal cord. This exacerbates the spasticity and causes the development of contractures, abnormal positioning and further loss of function. This development can be influenced by early physical activity and medication (10, 11).
Light to moderate spasticity may have a positive effect on function. Among other things, spasticity may make it possible for patients with lower limb paresis to attain a standing function and more ease of movement, for example transfer from bed to chair (12). Light to moderate spasticity contributes to better circulation in the legs, thereby avoiding oedema and reducing the risk of development of deep vein thrombosis (13, 14).
Pronounced spasticity may contribute to increased functional failure, contractures, incorrect posture, ulcers and pain. To avoid such negative developments, treatment should start as soon as possible (15).
Clinical assessment
When assessing spasticity, it is essential to have a thorough description of the extent and degree of the spasticity, plus its effect on the patient’s day-to-day functioning and quality of life (16). Spasticity can prevent transfer, affect the placing of legs in wheelchairs or cars, make manual hygiene difficult, cause problems with catheterisation and cause pain. If the patient cannot give a personal account of his or her problems, information should be obtained from those who know the patient best. When assessing different treatment options, an assessment of what it might be possible to achieve should also be included.
In the clinical examination it is important to assess the range of active and passive movements combined with any complications such as pain and/or abnormal limb positions. The most widely used assessment scales are the Ashworth Scale and the modified Ashworth Scale (17). With the Ashworth Scale, the resistance in the limb to passive movements is measured (18). This score says little about the loss of function that is related to spasticity and it is therefore not suitable for assessing the effect of treatment. There are a number of other scales, but they measure loss of function to only a limited degree and are therefore of limited clinical value (19) – (22). Individual assessment, preferably with the aid of video clips from before and after treatment, may be useful for assessing effectiveness. One important parameter will always be whether the aims of the treatment were fulfilled.
Electromyography (EMG) can be used to identify spastic muscles, but cannot be used to assess degree of spasticity or to determine an indication for treatment (8, 15).
Treatment
Spasticity can be influenced by a number of factors. Patients with SCI have a partial or complete loss of sensibility below the injury level. Increasing spasticity may be a reaction to disease or injury below the injury level. The first step is therefore to assess and treat all conditions that may lead to an exacerbation of the spasticity (exacerbating factors), such as urinary tract infection, pneumonia, obstipation, menstrual pain, ingrown nails, pressure ulcers etc. Acute, serious infections and syringomyelia may cause both increased spasticity and a sudden absence of spasticity (Fig. 1).

Spasticity is counteracted by systematic motor stimulation. The patient’s own physical activity and treatment involving stretching and bending as well as standing exercises may therefore modulate spasticity. This should be started immediately with all patients with SCI and spasticity (23).
Peroral medication
Spasticity can be treated medicinally, and peroral medication is the first-line choice for general, regional and local spasticity (Fig. 1). Peroral medication has a limited effect, but is simple to administer. Baclofen tablets are the most widely used medication, and the only pure spasmolytic drug registered in Norway. Baclofen is a GABA analogue and inhibits monosynaptic and polysynaptic spinal reflexes. Baclofen binds to GABAB receptors that are linked to potassium and calcium channels both pre- and post-synaptically (24).
The effect on spasticity was demonstrated in a controlled study of patients with SCI, while another study failed to show this effect (25, 26). The limited effect of baclofen is due to the fact that only a small portion of the active substance penetrates the blood-brain barrier (24). There are frequently side effects, with sedation, nausea, dizziness and difficulty in breathing as the most common (24). Side effects can be avoided to some extent by a slow stepwise increase of the dose. Epileptic seizures, psychosis and hyperthermia have been described in connection with sudden termination of the drug (27).
Another drug that may affect spasticity is tizanidine. This drug is not registered in Norway, and is therefore used only when baclofen is not effective. Tizanidine is an imidazole derivative and a central ?-adrenergic agonist which inhibits the release of excitatory amino acids in spinal interneurons (24). A study demonstrated a pronounced muscle relaxant effect and suppression of polysynaptic reflexes in cases of complete SCI (28). Tizanidine has proved to be effective in placebo-controlled studies of patients with SCI (29, 30). Tizanidine and baclofen can be combined, but the drugs have the same potential side effects, and these may be more pronounced when the drugs are used in combination.
Benzodiazepines may be effective for reducing spasticity (24). The effect is a result of the GABAA receptor system being affected. Sedation and cognitive modulation are common side effects. When used concurrently with baclofen, benzodiazepines have an additive effect on spasticity, but must be administered in small doses because of similar side effects that may be more pronounced than with monotherapy (24). Benzodiazepines are often used in acute cases where a rapid effect is desired and side effects are a secondary consideration.
Clonidine and gabapentin may have the effect of reducing spasticity. Their use as spasmolytics in clinical practice is limited, and clinical studies of patients with SCI have not been performed (24, 30).
Cannabis products have been tested as medication for pain and spasticity, but the results of the studies have been conflicting. These products are not recommended for treating spasticity because of the narrow therapeutic range and risk of side effects and dependence (31).
Local injections with botulinum toxin
If spasticity is restricted to a few muscles or a delimited muscle group, injection with botulinum toxin may be a treatment option. Botulinum toxin is a product of Clostridium botulinum and blocks presynaptic release of acetyl choline from nerve terminals so that the connection between nerve terminal and muscle fibre is cut off for a period. Botulinum toxin weakens muscle tone, and paresis may thus become more pronounced. Seven immunologically distinct toxins are known (types A–G). Treatment with type A is most common (Botox, Xeomin, Dysport), but type B (Neurobloc) can also be used. A prerequisite for successful treatment is that a precise analysis and description of function is made in advance and an assessment made of what can be achieved by means of injection into particular muscles.
It must also be ensured that the injections are given into the correct muscles. The muscles can be identified by means of a one-channel EMG system, stimulator or ultrasound (8). The effect develops gradually in the course of 1 – 3 days after the injection, and maximum effect can be seen after 5 – 14 days. The effect wanes gradually after 12 – 16 weeks (24). Because of the risk of antibody development, the treatment can be repeated after three months at the earliest. Treatment with botulinum toxin must be limited to a few muscles, and no more than 50 units of Botox or Xeomin (150 units of Dysport) should be injected in the same place (33). The total dose per treatment should not exceed 500 units of Botox or Xeomin (1500 units of Dysport). The best effect is obtained by combining botulinum toxin treatment with physiotherapy (34, 35). Botulinum toxin treatment has a stable and predictable effect (36) which has been demonstrated in patients with SCI and a number of other conditions (36, 37). However, there are no randomised studies of the effect of botulinum toxin on persons with SCI. Randomised studies are beset by ethical challenges, because they would mean many injections of placebo into muscles.
Intrathecal infusion of baclofen
In cases of regional spasticity, particularly in the lower limbs, treatment with intrathecal infusion of baclofen via a programmable pump will be more effective than treatment with peroral medicines (8) (Fig. 2). Baclofen is pumped directly into the subarachnoid space by means of a programmable pump via a catheter system. Systemic effects are thereby reduced, and the risk of side effects is considerably lower (24). Before the baclofen pump can be implanted, the effect of intrathecal baclofen must be tested by administering a bolus dose to the patient via a normal spinal puncture needle, alternatively continuous infusion via an external pump. After implantation, the dose can be adjusted by a programmer, and the pump reservoir must be refilled with baclofen at regular intervals. The treatment can be administered over a long period, and the effect is sustained (38, 39). There is no absolute upper limit for the dosing. Daily doses of over 500 g may cause side effects, first and foremost in the form of sedation.
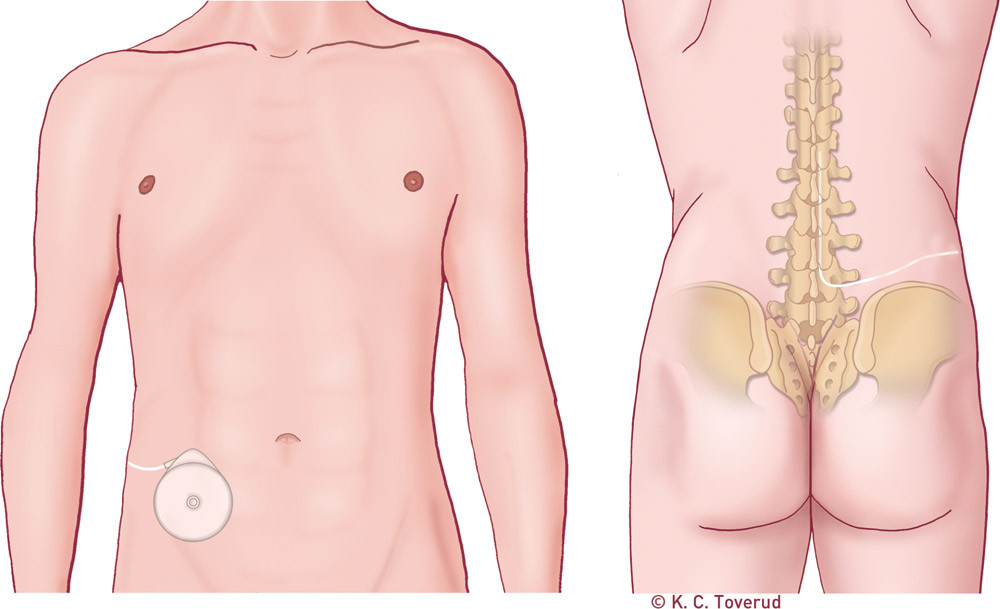
In cases of extensive spasticity in all extremities, the effect of intrathecal baclofen on the upper limbs has proved to be only about 25 % of the effect on the legs (40). In cases of spasticity in both upper and lower limbs, combination treatment with a baclofen pump for spasticity in the lower limbs and botulinum injections for spasticity in the upper limbs may be a good alternative. In order to preserve the walking function, a combination of a low dose of intrathecal baclofen and botulinum toxin can be tried in some muscles in the lower limbs. Spasticity can thereby be dampened in a more targeted way so that the patient gains better control over musculature and movement and thereby preserves walking function.
Surgical treatment of spasticity
Surgical treatment of spasticity entails irreversible changes and can often be avoided if other steps are taken at an early stage. Orthopaedic interventions can be used either as a sole measure or in combination with physiotherapy and botulinum toxin injections in cases of local spasticity (41). Tendon extension, tendon plasty or osteotomy can be considered for selected patients with intractable local spasticity with joint deformation, as in equinus contracture. In cases of pronounced regional spasticity, posterior selective rhizotomy may be an option (43). This treatment is most suitable for patients with non-progressive conditions.
Practical management
Spasticity is not a static phenomenon and should be followed up both during and after stays in hospital. All patients with spasticity should be urged to exercise. If this is not sufficiently effective, the patient should have physiotherapy with guidelines for exercises that counteract the spasticity.
Peroral drugs can be prescribed by the primary doctor. For treatment with botulinum toxin the patient should be referred to the nearest specialist who can offer this treatment. Assessment, testing and implantation of baclofen pumps are carried out by university clinics. Observation and problem description by the patient’s primary doctor and physiotherapist and information about the patient’s other illnesses and conditions are important for enabling the correct treatment strategy to be chosen. The use of warfarin and conditions that result in a higher risk of haemorrhaging may be a relative contraindication that should be considered before botulinum toxin is used or intrathecal baclofen is tried. Refilling and other adjustment of pumps are carried out at a number of hospitals in Norway. The patient should receive an offer from the closest hospital to avoid having to travel far. All other measures should be attempted before orthopaedic or neurosurgical intervention is considered.
Tabell
| Main points |
|
- 5.
Lance JW. The control of muscle tone, reflexes, and movement: Robert Wartenberg Lecture. Neurology 1980; 30: 1303 – 13. [PubMed]
- 9.
Saval A, Chiodo AE. Intrathecal baclofen for spasticity management: a comparative analysis of spasticity of spinal vs cortical origin. J Spinal Cord Med 2010; 33: 16 – 21. [PubMed]
- 17.
Ashworth B. Preliminary trial of carisopodol in multiple sclerosis. Practitioner 1964; 192: 540 – 2.
- 19.
Tardieu G, Shentoub S, Delarue R. A la recherché d’une technique de mesure de la spasticité. Rev Neurol 1954; 91: 143 – 4. [PubMed]
- 23.
Gracies JM. Pathophysiology of impairment in patients with spasticity and use of stretch as a treatment of spastic hypertonia. Phys Med Rehabil Clin N Am 2001; 12: 747 – 68. [PubMed]
- 26.
Burke D, Gillies JD, Lance JW. An objective assessment of a gamma aminobutyric acid derivative in the control of spasticity. Proc Aust Assoc Neurol 1971; 8: 131 – 4. [PubMed]
- 29.
Nance PW, Bugaresti J, Shellenberger K et al. Efficacy and safety of tizanidine in the treatment of spasticity in patients with spinal cord injury. Neurology 1994; 44 (suppl 9): S44 – 51. [PubMed]
- 30.
Taricco M, Pagliacci MC, Telaro E et al. Pharmacological interventions for spasticity following spinal cord injury: results of a Cochrane systematic review. Eura Medicophys 2006; 42: 5 – 15. [PubMed]
- 35.
Smedal T, Gjelsvik B, Lygren H et al. Botulinum toksin A- effekt på spastisitet. Tidsskr Nor Lægeforen 2001; 121: 3277 – 80. [PubMed]
- 39.
Koulousakis A, Kuchta J. Intrathecal antispastic drug application with implantable pumps: results of a 10 years follow-up study. Acta Neurochir Suppl (Wien) 2007; 97: 181 – 4. [CrossRef]
- 42.
Smyth MD, Peacock WJ. The surgical treatment of spasticity. Muscle Nerve 2000; 23: 153 – 63. [PubMed]
- 43.
Grønning M, Svendsen F, Skeidsvoll H et al. Spastisitet behandlet med selektiv bakre rhizotomi. Tidsskr Nor Lægeforen 2002; 122: 1190 – 1. [PubMed]