Intestinal and multivisceral transplantation in patients with chronic intestinal failure
Short bowel syndrome can arise when the length of the small intestine is less than 75 – 100 cm. Patients develop chronic diarrhoea, lose weight and can suffer from serious undernutrition. In children, a bowel length of 50 cm may be sufficient as adaptation of the intestinal mucous membranes takes place after a while (1). Chronic intestinal failure can also arise as a result of intestinal pseudo-obstruction, where the bowel is of normal length but does not absorb necessary nutrients because of motility disorders. Chronic intestinal failure develops only in connection with disease or damage to the small intestine. It is possible to function without the large intestine, but if the large intestine is preserved, the need to supply extra fluid and saline will be reduced (2). Intestinal transplantation is therefore only relevant when the small intestine fails. Up until 1998, the results of this treatment were considerably poorer than for other organ transplantations (3, 4). It is necessary to transplant a large portion of the small intestine (at least 2 m) from a deceased organ donor in order to achieve acceptable intestinal function after the operation.
Patients with chronic intestinal failure can live for many years with an acceptable quality of life with the aid of total parenteral nutrition (TPN) (5). The insertion of a central venous catheter is necessary to provide assured access to the blood stream and is an absolute prerequisite for long-term TPN treatment. Bowel-lengthening surgery should be considered for children with short-bowel syndrome in order to determine which patients can stop using this type of nutrition, or possibly manage with intermittent treatment (6). The treatment may result in serious complications such as thrombosis of central veins, particularly if it lasts for months or years, and catheter infections may cause life-threatening sepsis. TPN-associated cholestasis may also develop because of abnormally viscous bile, as may gallstones, which in time can cause serious, irreversible liver failure.
A small number of intestinal transplantations were performed in the 1960s and 1970s, but the programmes were stopped because none of the patients lived for more than a few months after transplantation (7). The bowel contains a large number of immunologically active cells which caused an early and strong rejection of the graft. Serious infections developed because the intestinal mucous membranes were damaged by the rejection process, enabling bacteria to traverse the intestinal wall and enter the bloodstream, causing serious and life-threatening sepsis. After better immunosuppressive drugs became available (calcineurin inhibitors, CNI), first cyclosporine A from 1983 and then tacrolimus from 1995, transplantation was again attempted. Further immunosuppressive treatment, in addition to calcineurin inhibitor, proved necessary to achieve acceptable results. Immunosuppressive protocols that contained both tacrolimus and a specific antibody against lymphocytes, antithymocyte globulin (ATG), resulted in higher graft and patient survival (8) – (10). Intestinal transplantation is an established treatment today for chronic intestinal failure, and an international register has been established in Ontario, Canada (3). Patient survival after five years is over 60 % at several transplantation centres (10) – (13).
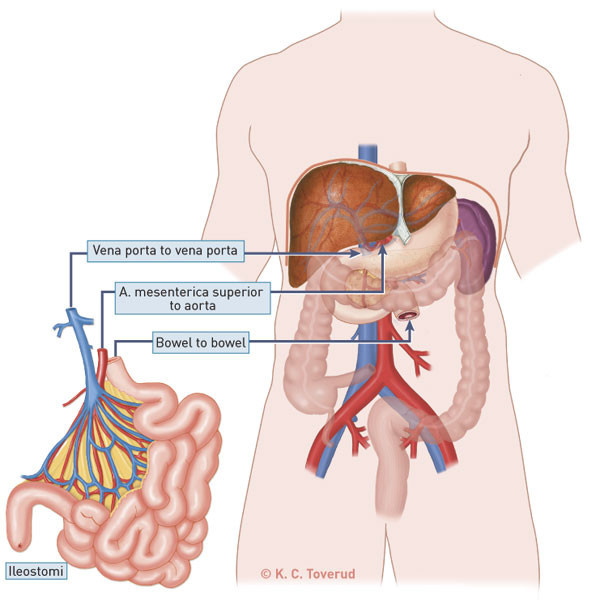
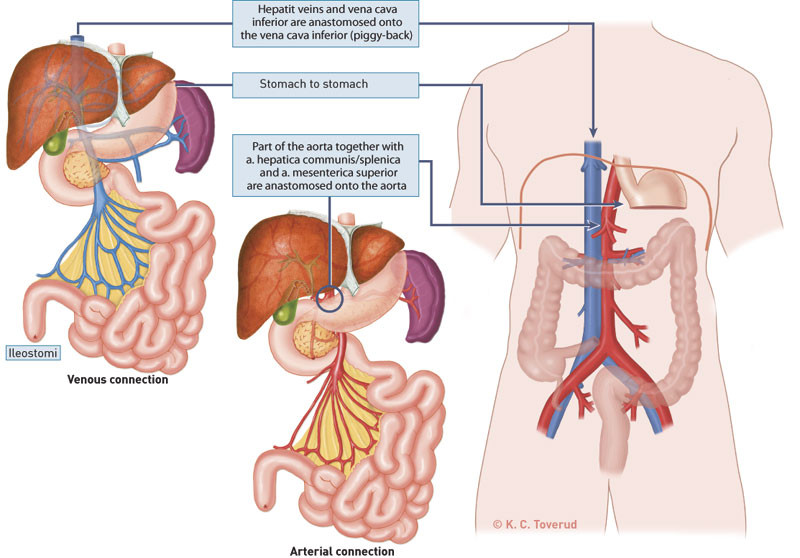
The results for intestinal transplantation have been substantially less successful than for other organ transplantations. Intestinal transplantation is therefore considered only if serious problems arise in connection with treatment with total parenteral nutrition. Isolated intestinal transplantation (Fig. 1) is considered when inadequate vascular access or life-threatening infection episodes occur after long-term treatment. In the event of concurrent chronic liver failure, liver transplantation is considered as well. Multivisceral transplantation which encompasses stomach, duodenum and pancreas as well as the small intestine and liver will often be the best treatment in such cases (Fig. 2). This paper presents experience from seven Norwegian patients who were treated with intestinal/multivisceral transplantation abroad.
Material and method
All the patients were primarily assessed at Oslo University Hospital, Rikshospitalet before they were accepted for bowel transplantation. Clinical information was obtained from the transplantation hospital after the operation. Oslo University Hospital, Rikshospitalet has been responsible for following up patients after the transplantation, in close cooperation with the referring hospital.
Pre-operative assessment
Satisfactory heart and lung function is a prerequisite for patients being entered on Scandiatransplant’s waiting list, which is a joint waiting list for the five Nordic countries. Abdominal vessels must be examined pre-operatively with CT angiography. In cases of liver failure, an ultrasound scan must be performed and an MR of the liver and bile ducts. Multivisceral transplantation is contra-indicated in the case of serious infections.
Patients
Prior to transplantation, all patients had a long case history of numerous laparotomies accompanied by weight loss and poor general condition despite total parenteral nutrition. In three patients, the reason for short-bowel syndrome was pronounced bowel gangrene, which was due to strangulation of the entire small intestine in two patients and thrombosis in intestinal mesenterial veins in one patient. Intestinal pseudo-obstruction was the indication for transplantation in three patients, because of Hirschsprung’s disease with total colon obstruction in one patient (a child), and because of congenital cryptogenic motility disorder of the bowel in the other two patients. A colectomy was also performed on the child with Hirschsprung’s disease.
One patient had Gardner’s syndrome with pronounced colon polyposis and developed multiple, large desmoid tumours intra-abdominally after a total colectomy. An attempt was made to treat them with chemotherapy (Caelyx (doxorubicin) and Dakarbazine) every four weeks. The treatment had a transitory effect on the growth of the tumours, but after one year the patient developed multiple bowel fistulae. After a while the chronic, intestinal obstruction became untenable for the patient.
All patients apart from one had developed chronic irreversible liver failure after long-term total parenteral nutrition (spread 2 – 25 years).
Operation
Multivisceral transplantation (Fig. 2) was performed on all seven patients and included liver transplantation in six. The patient who did not have a liver transplantation also had cholestasis, but the reason proved to be multiple gallstones. The patient with mesenterial vein thrombosis had had a liver transplantation two months earlier. An occluded portal vein with continued portal hypertension and massive ascites production and variceal haemorrhaging from the oesophagus were indications for liver retransplantation. In Miami, the spleen was included in the multivisceral transplantation in both patients and the entire abdominal wall of one patient was transplanted because of scar formation. The inferior epigastric artery and vein of the abdominal wall transplant were anastomosed to the iliac vessels using microsurgery technology. Two patients also had kidney transplants performed because of chronic renal failure.
Post-operative follow-up
There is a very high (90 %) risk of rejection and infection in the months immediately after the transplantation. Daily blood tests with close clinical observation, including measuring the amount of fluid from the ileostomy, are important in the early post-operative phase. Three to four biopsies are initially taken once or twice a week from the transplanted bowel with the aid of a flexible endoscope via the ileostomy; should there be early signs of rejection, appropriate treatment can start immediately. Grading of rejection is based on both endoscopic and microscopic findings which presuppose specialist expertise in endoscopy and pathology (14). Weekly monitoring of CMV PCR and EBV PCR are important for detecting virus reactivation/infection.
Ethics
All patients discussed in the manuscript gave written consent to publication. In the case of patients who died, consent was given by their families. The Norwegian Data Protection Officer authorised publication. The study was not reported to the Regional Ethics Committee (Rek) because it concerns ongoing handling and treatment of patients with chronic intestinal failure where established treatment has been proceeding in Sweden and Miami for a number of years (since 1995).
Results
A total of seven Norwegian patients had multivisceral transplantations performed in the period 2001 – 2009, five in Gothenburg and two in Miami. One patient was a child (girl, four years old) and the other six were adult women and men aged 27 – 48. The operation was often a long one (spread 12 – 40 hours) and complicated due to previous surgical operations in most patients. A chronological overview of the results is shown in Table 1.
Table 1
Chronological overview of Norwegian patients who had intestinal/multivisceral transplantations performed abroad in the period 2001 – 2009
| Patient |
Treatment |
Result |
||||||||
| Age (years) |
Gender |
Diagnosis |
Transplantation |
Place |
Year |
Rejection (n) |
Reopera-tion (n) |
Status (time after transplantation) |
||
| 45 |
♂ |
Bowel harmed by radiationShort-bowel syndrome |
Multivisceral transplantation, incl. kidney |
Miami |
2001 |
0 |
2 |
Alive (120 mo.) |
||
| 27 |
♂ |
Strangulation ileusShort-bowel syndrome |
Multivisceral transplantation |
Gothenburg |
2002 |
1 |
1 |
Dead (1 mo.) |
||
| 33 |
♂ |
Gardner’s Syndrome Desmoid |
Multivisceral transplantation without liver |
Miami |
2005 |
2 |
6 |
Dead (24 mo.) |
||
| 4 |
♀ |
Hirschsprung’s Disease (total) |
Multivisceral transplantation, incl. kidney |
Gothenburg |
2005 |
1 |
0 |
Alive (76 mo.) |
||
| 37 |
♀ |
Pseudo-obstruction |
Multivisceral transplantation, incl. kidney |
Gothenburg |
2005 |
1 |
2 |
Dead (10 mo.) |
||
| 48 |
♂ |
Portal vein thrombosisLater also liver transplantation |
Multivisceral transplantation |
Gothenburg |
2008 |
2 |
1 |
Alive (36 mo.) |
||
| 34 |
♀ |
Pseudo-obstruction |
Multivisceral transplantation |
Gothenburg |
2009 |
1 |
2 |
Alive (24 mo.) |
||
Rejections
All patients had suffered 1 – 2 rejections of small intestinal transplants apart from one patient who had had radiation treatment for testicular seminoma 15 years prior to the transplantation. Acute rejections are treated with high doses of steroids (up to 1 g methylprednisolone daily for four days) and in addition monoclonal anti-CD3-antibody (OKT-3) in the case of steroid-resistant rejection that does not respond to standard steroid treatment.
Other complications
Six of the patients have had 1 – 6 re-operations because of intra-abdominal abscesses (four patients), bile leakage (two patients) and intestinal fistulae (two patients). All the patients had also had several infection episodes, triggered bacterially or by cytomegalovirus (CMV) or Epstein-Barr virus (EBV). One patient developed ulceration of the ventricle two years after the transplantation; a biopsy revealed EBV-positive lymphoma consistent with PTLD (post-transplant lymphoproliferative disease). The lesion healed after immunosuppression had been reduced.
Long-term results
Four of seven patients are alive and their general condition is satisfactory 1 – 10 years after the transplantation. For the most part, these patients take nutrition in the normal manner and are no longer dependent on total parenteral nutrition, but periodically they have to have fluid and electrolytes administered intravenously. Three patients died following a serious infection one, ten and 24 months, respectively, after transplantation took place. Two of the three patients who died had an impaired nutritional status with a BMI of less than 15 kg/m2. The third patient died 24 months after the bowel transplantation as a result of sepsis following renal transplantation.
Discussion
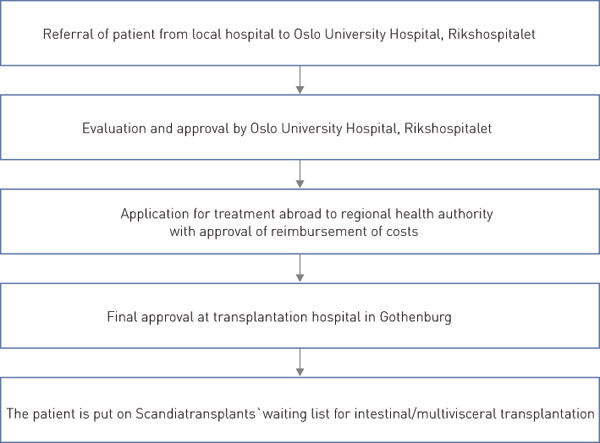
To date, physicians in Norway have no experience of our own of intestinal transplantations. Survival in our patients treated in collaboration with Gothenburg and Miami is on a par with the results reported in the literature (60 % five-year survival) (14). Early referral of patients with chronic intestinal failure to Oslo University Hospital, Rikshospitalet, is crucial for optimising the time of operation to ensure the best possible outcome for the patient (Fig. 3). Isolated bowel transplantation is a smaller and technically simpler operation than multivisceral transplantation, where the supply of organs is also limited. From an immunological point of view, however, bowel transplantation is just as challenging as multivisceral transplantation (12, 13).
The choice in Denmark has been to centralise treatment of chronic intestinal failure in Copenhagen and Århus, since cross-disciplinary teams have to be established to provide optimal follow-up (15). It is particularly important to avoid TPN-associated cholestasis; the primary measure is a reduced supply of intravenously administered lipids (5). Clinical studies have shown that fat emulsions containing a mixture of soya, coconut, olive and fish oil result in less cholestasis, particularly in children (16). Randomised studies of SMOF lipids in connection with chronic intestinal failure are taking place in Europe and the USA (17).
The establishment of a intestinal transplantation programme is contingent on the participation of a number of specialists in several internal medicine specialities (gastroenterology, hepatology, paediatrics) transplantation surgery and anaesthesiology as well as specialised nursing. Many other specialities and professional groups also play an important part in the extensive evaluation and treatment of patients with chronic intestinal failure (medical biochemistry, microbiology, transplantation immunology, radiology, pathology, nutritional physiology, physiotherapy, physical medicine and rehabilitation and social medicine.
The treatment is resource-intensive, with a daily cost at least three times as high as the cost of liver transplantation alone, a minimum of NOK 3 million. The medication is assumed to cost NOK 100 000 annually. After ten years it can be estimated that every extra year of life won has cost at least NOK 400 000. By way of comparison, the costs of treatment with total parenteral nutrition are around NOK 1 million per year. After a successful intestinal transplantation, patients with chronic intestinal failure can have many good years with a satisfactory quality of life. A continuous social assessment must nevertheless be made of whether this is a correct use of medical resources. The treatment is only relevant for a few younger patients (the upper age limit is 50 years). In a debate on priorities, it is an open question which patients with serious and rare disorders should be offered a costly treatment, rather than patients with diseases for which the treatment costs are far lower. Patients and family must be informed that intestinal transplantations are probably the most complex treatment in modern medicine, and that the treatment can take a heavy toll on the patient.
Multivisceral transplantations are particularly resource-intensive, and presuppose that the inter-disciplinary team treats a sufficient number of patients to be able to build up and maintain the necessary expertise. The professional community at Oslo University Hospital, Rikshospitalet is of the view that we should continue to base ourselves on collaboration with Sahlgrenska University Hospital, Gothenburg, as long as they have the capacity to treat both their own and Norwegian patients. In very special cases it may be necessary to refer the patient to transplantation centres in the USA, which have most experience with bowel transplantations. A large population base is required to maintain the expertise necessary for patients with chronic intestinal failure to be offered life-saving treatment with bowel and multivisceral transplantation. Since the need for bowel transplantation in Norway has been 1 – 2 per year so far, the professional community at Oslo University Hospital, Rikshospitalet has agreed not to start a separate national programme.
Tabell
| Main points |
|
- 3.
The Intestinal Transplant Registry. www.lhsc.on.ca/itr/
- 10.
de Ville de Goyet J. Place of transplantation in infant intestinal failure management. Transplantation 2000; 69: 1234 – 6. [PubMed]
- 11.
Herlenius G, Krantz M, Finkel Y et al. Tarmtransplantation–experimentell terapi som blivit realistiskt alternativ. Läkartidningen 2004; 101: 2874 – 8. [PubMed]
- 13.
Tzakis AG, Kato T, Levi DM et al. 100 multivisceral transplants at a single center. Ann Surg 2005; 242: 480 – 90, discussion 491 – 3. [PubMed]