The incidence of breast cancer in Norway has increased in the past 50 years (1). The increase has been particularly pronounced since 1995, and in this period has largely affected the group aged 50 – 69. Organised mammography screening for the age group 50 – 69 commenced in 1996 in Akershus, Oslo, Rogaland and Hordaland (the test counties). The incidence of breast cancer in the invited age group was 82 % higher in 1996 – 1997 (first screening round) and 50 % higher in 1998 – 2000 (second and third screening rounds) than in the period 1991 – 1995 (2). A large number of cases of ductal carcinoma in situ (DCIS) were also discovered by mammography (3). We calculated that there would be 770 more breast cancer diagnoses annually once screening was introduced throughout Norway (3).
It has been generally supposed that the primary reason for the increase in the incidence of breast cancer in women who come for screening is that some cancer diagnoses are made earlier and that there will be a fall in incidence later. «Lead time» is the term used to indicate how much the time of diagnosis is believed to have been brought forward. However, the concept is confusing when there are tumours that would never have been diagnosed in the absence of screening (either because they do not grow, or because they disappear spontaneously). The concept of ’brought forward in time’ is meaningless for tumours of this kind.
Overdiagnosis as a result of cancer screening can be defined as the finding of tumours that would never otherwise have been discovered during the patient’s lifetime (4). Overdiagnosis (in contrast to lead time) is defined for all tumours that are diagnosed by mammography and is easy to calculate. The absolute number of over-diagnosed women is calculated by taking the difference between the increase in the number of cancer diagnoses among those who are invited for screening (compared with the expected number for the age group) and the reduction in the number of diagnoses among those who are no longer invited.
We have pointed out previously that because there is no definite reduction in the incidence of breast cancer in the over 70s age group, virtually the whole 50 % increase in the age group 50 – 69 must be overdiagnosis (2). The fact that there was a virtually stable incidence of breast cancer in the age group 40 – 49 (who are not invited for screening) weighs against the possibility that the increase is due instead to a generally increasing incidence of breast cancer in Norwegian women (2). We therefore concluded that one in three cases of breast cancer in the age group 50 – 69 is overdiagnosed tumour disease (2). More than half the other cases of breast cancer (those that were not overdiagnosed) consist of «interval cancer» (cancer that is discovered between two rounds of screening) and of tumours that are diagnosed among the 20 – 25 % of those who are invited but who choose not to come for screening. As a result, there are correspondingly fewer real cases of cancer left to be detected by screening. We have shown previously that when ductal carcinoma in situ is included and interval cancer and cancer in women who do not go for screening are excluded, no less than two of three cancer diagnoses (including DCIS diagnoses) in the mammography screening must represent overdiagnosis.
If early diagnosis is the cause of 770 more cancer diagnoses in the age group 50 – 69, then there must be a very dramatic fall after the age of 69 (5). In the age group 70 – 84, for example, there are only 450 cases of breast cancer a year (1). We have reported previously that there was no statistically significant reduction in the incidence of breast cancer in this age group (2). We have instead suggested that screening leads to massive overdiagnosis, and that overdiagnosed tumours will normally (i.e. in the absence of screening) disappear spontaneously (6, 7).
An important objection to our study in 2004 was that with only three screening rounds we had too short a follow-up time to be able to say anything definite about the level of overdiagnosis. Another objection was that we did not adjust for increased use of hormones against menopausal discomfort. We have therefore now updated our estimate of overdiagnosis with data from seven screening rounds.
Material and method
Aggregated data on the incidence of infiltrating breast cancer and ductal carcinoma in situ diagnosed by the national mammography programme and outside the programme were provided by the Cancer Registry of Norway. The number of women at risk from breast cancer each year in the period 1991 – 2009 was determined using the mean population registered by Statistics Norway (8).
In order to determine the level of overdiagnosis, we used a Poisson regression model to calculate any underlying increase in the incidence of breast cancer for the age group 40 – 79 (we included a linear trend parameter for the whole period) while we simultaneously adjusted for age, county, population growth and the introduction of mammography screening.
Results
Table 1 shows the rates of infiltrating breast cancer and interval cancer, the detection rate and the rate of ductal carcinoma in situ for women who were invited to mammography screening in the period 1998 – 2009 in the four test counties. The rate of interval cancer and the detection rate are both virtually constant in this material. Therefore the incidence of breast cancer as a whole must also be constant in women who go for mammography screening. Because the fall in the number who come for screening is insignificant, the incidence of breast cancer among those who are invited and do not go for mammography screening must also be virtually constant. Nor is there any significant falling trend in the rate of ductal carcinoma in situ, in either the test counties (p > 0.05) or the rest of Norway (p > 0.05) (table 1).
Table 1
Rate of breast cancer, ductal carcinoma in situ (both per 100 000 invited), interval cancer rate (calculated by dividing by two the number of new cancer cases per 100 000 in the first two years after screening start ) and detection rate (number of cancer cases per 100 00 who are really screened) for the invited age group 50 – 69 in the period 1998 – 2009 in the four test counties Oslo, Akershus, Rogaland and Hordaland
| 1998 |
1999 |
2000 |
2001 |
2001 |
2003 |
2004 |
2005 |
2006 |
2007 |
2008 |
2009 |
|
| Breast cancer rate |
309 |
288 |
288 |
307 |
284 |
318 |
294 |
318 |
286 |
290 |
282 |
293 |
| DCIS |
50 |
40 |
52 |
48 |
53 |
39 |
51 |
51 |
49 |
50 |
53 |
–¹ |
| (2nd round) |
(3rd round) |
(4th round) |
(5th round) |
(6th round) |
(7th round) |
|||||||
| Interval cancer rate |
93 |
83 |
100 |
89 |
–¹ |
–¹ |
||||||
| Detection rate |
565 |
622 |
607 |
590 |
639 |
618 |
||||||
| [i] | ||||||||||||
[i] ¹ Data not available
Figure 1 shows the breast cancer incidence rates in the period 1991 – 2009 in the test counties. Data from the first screening round (1996 – 1997) are not included. The average incidence rates in the age group 50 – 69 for each screening round are 297 (1998 – 1999), 298 (2000 – 2001), 301 (2002 – 2003), 306 (2004 – 2005), 286 (2006 – 2007) and 282 (2008 – 2009) per 100 000 women. In the period 1998 – 2009, the incidence rate in the age group 50 – 69 fell by 0.7 % per year (p = 0.064) in the test counties. The incidence rate for the age group 50 – 59 fell by 1.7 % (p < 0.001) per year. The regression analysis shows no significant changes in the incidence rate in the age group 40 – 49 in the period 1991 – 2009 (p = 0.50), but the incidence rate in the age group 70 – 79 fell by 1.3 % per year (p < 0.001) during the same period. Almost the entire reduction occurred in the age group 70 – 74, where the incidence rate fell by 2.4 % per year (p = 0.02) after 1997.
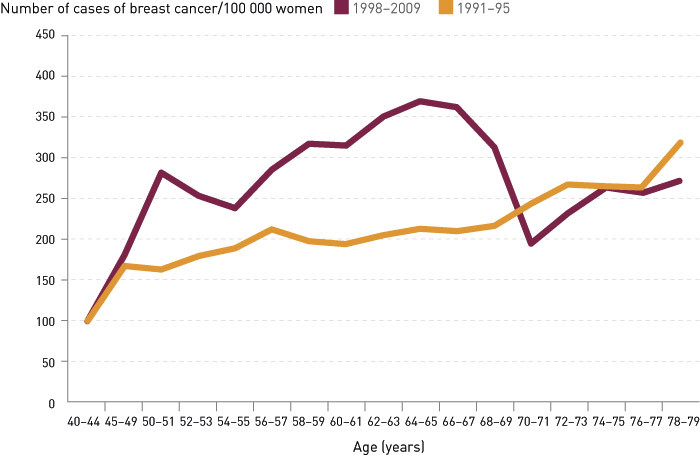
A Poisson regression analysis that includes age, underlying trend and the introduction of mammography screening in 1996 as variables shows that women in the age group 70 – 74 who had been invited to screening when they were younger have a 7 % lower incidence rate than women who were not invited, but this difference was not statistically significant (p = 0.18). This decline corresponds to around ten fewer cases of breast cancer per year. The introduction of mammography screening in 1996 therefore remains the only explanation for the change in the incidence of breast cancer in the period 1991 – 2009 in the test counties – a 50 % increase in the incidence of breast cancer in the age group 50 – 69 and a negligible, brief and non-significant decline in incidence after the age of 69.
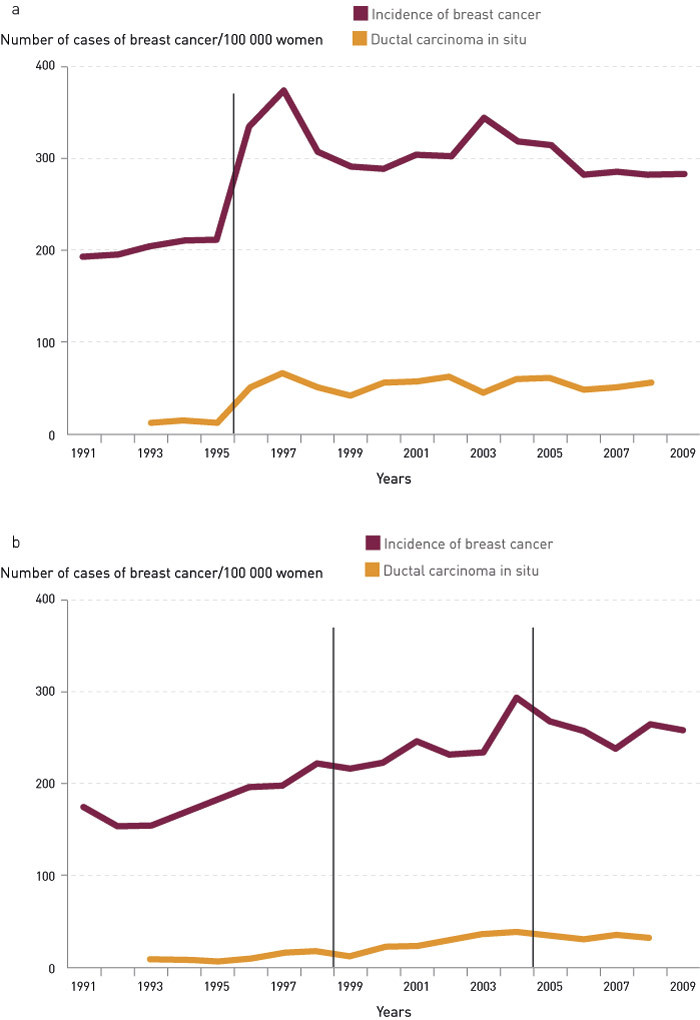
The DCIS rates are an indication of the extent to which women go to screening. Figure 2a shows the incidence rates for breast cancer and ductal carcinoma in situ before and after the start of screening in the test counties. The DCIS rates were nine (1993), 14 (1994) and 12 (1995) per 100 000. After the start of screening, the rate rose immediately to 50 per 100 000. Figure 2b shows rates of incidence for breast cancer and ductal carcinoma in situ in the rest of Norway. The DCIS rate increased from eight per 100 000 before 1996 to about 17 per 100 000 in 1998. When all the other Norwegian counties had established organised screening, the DCIS rate in these counties had reached about 35 per 100 000.
In the years 1991 – 1995, there were 2 000 cases of breast cancer on average in Norway. In 1996, the first year of mammography screening, 2 352 cases were detected. In the same year, 87 DCIS cases were diagnosed in the test counties, 48 of them through the screening programme. In 2009, breast cancer was diagnosed in 2 745 women. In addition, around 300 women were diagnosed annually as having DCIS.
From 1991 – 2009, the number of women in the age group 50 – 69 increased by 35 %, from 403 958 to 542 852. In the early 1990s, there were 700 cases of breast cancer annually in the age group 50 – 69. With an unchanged incidence rate, about 945 cases of breast cancer should have been expected in this age group in 2009, but the Cancer Register recorded 1 473 cases that year. Since the screening programme has expanded to cover the whole country, there have thus been more than 500 extra cases of breast cancer detected each year in the invited age group, and around 300 cases of DCIS. In the age group 70 – 74, there are only about ten fewer cases of cancer each year. The reduction after 69 years is not significant, and it is debatable whether one should adjust for it or not. Ten fewer cases of breast cancer in the age group over 69 years only explain 2 % of the previous increase, however. An adjustment for this is of no significance.
Discussion
After 14 years of screening in the test counties, the incidence of breast cancer among those invited to take part is stable at a level that is 50 % higher than the baseline level from the period before screening started. There has not been any statistically significant reduction in the incidence of breast cancer in women who are no longer invited to screening. If the fall of 7 % in the age group 70 – 74 had been statistically significant, it would only have been able to explain a couple of per cent of the increase of 50 % (2). Thus almost the entire increase in the incidence of breast cancer must be due to overdiagnosis – not early diagnosis.
Our regression analysis of data from the test counties shows that the introduction of the screening programme explains the entire increase in incidence in the test counties in the period 1991 – 2009. The fact that we find no signs of an increase in the incidence rate in age groups that are not screened is a clear indication that there is no general underlying increase in incidence that is independent of the use of hormones (see below). We wish to stress that the data set we have studied is from a population with little migration and with a high, stable level of meeting up for screening, a stable detection rate through screening, a stable incidence of interval cancer and a stable incidence of breast cancer among those who do not come for screening (table 1).
In a systematic review of a number of national screening programmes, Jørgensen & Gøtzsche (9) found that the incidence of breast cancer (including DCIS) increased by 52 %. They describe a lower level of overdiagnosis for Norway than we are reporting here, but their analysis is based on extrapolation of the underlying increase in incidence in the test counties from the period 1980 – 1995 (before organised screening started). Because much of the increase in this period can be explained by non-organised screening, we believe that their estimate of a screening-independent increase in incidence is far too high. Nor do constant or falling breast cancer rates in all western countries for all age groups since the year 2000 suggest that there would have been any rise in incidence in the absence of screening (10) – (13).
In the period 1991 – 1999 there was a sharp rise in the number of women who used hormones for menopausal discomfort. Subsequently, the number of users has fallen just as sharply (13). The reason for the fall can be attributed to the publication of randomised trials that showed that the risk of breast cancer increased by 24 % with long-term use of hormones (14). Reports subsequently came from some countries that the fall in the use of hormones was followed by a slight decline in the incidence of breast cancer (10) – (12). The Norwegian sales figures for 2007 – 2009 for hormone replacement products for systemic treatment (ATC groups G03C and G03F) were around 60 000 DDD/day, which is the same as in the period 1991 – 1993 (15). However, during this period, the number of potential users (i.e. women in the age group 50 – 69) increased by 33 % (8). This means that at the end of the trial period the proportion of hormone replacement users was far lower than at the start. The fact that the incidence of breast cancer (adjusted for mammography screening) has remained stable throughout the period (and particularly during the last eight years, when sales of hormone replacement products have fallen by 70 %), indicates that the use of oestrogen and progestin cannot be a particularly important cause of breast cancer in Norway (10).
In some Norwegian counties, there was a strong increase in the incidence of breast cancer before organised screening started. A simultaneous increase in the DCIS rates in these counties (16) may indicate that much of this increase is due to non-organised screening (the diagnosis DCIS can be made almost exclusively after the use of mammography) (17). However, we cannot exclude the possibility that increased attention to breast cancer by women and doctors has led to more diagnostic mammography scans, which has increased the incidence of both DCIS and cancer. This illustrates that it is difficult to define what is due to greater attention to breast cancer and what is due to non-organised screening.
In Norway the incidence of breast cancer has declined a little in the age group 50 – 69 years since 2001, but only in the counties where screening was introduced in the period 2000 – 2004 (10). This decline can therefore be explained as a prevalence effect – a higher incidence of breast cancer is expected in the first screening round than in the following rounds (5).
Overdiagnosis has normally been explained as being due to the detection of tumours that grow so slowly that the women will die of other causes before the tumours cause clinical symptoms (5, 18). These slow-growing tumours should be very largely detected when women attend the first round of screening, and the detection rate should sink dramatically from the first to the second screening round because this tumour population would then have been eliminated. Similarly, the incidence rate should remain very low for many years to come when women leave the screening programme at the age of 69 (see our initial argument to the effect that there should be hardly any cancer in the age group 70 – 84 if the bulk of the increase due to screening is early diagnosis) (5) – (7, 19). But that is not how it is! We have therefore suggested that overdiagnosis can very largely be explained as being due to the detection of tumours that would have shown spontaneous regression without treatment after a while (6, 7). Fryback et al. (19) came to the same conclusion when they analysed cancer register data from Wisconsin in the USA.
Conclusion
After 14 years of mammography screening in the test counties, there is a 50 % increase in the incidence of breast cancer that cannot be explained as early diagnosis or the use of oestrogen plus progestin products. Rather, the increase must be very largely due to overdiagnosis. This means that about 800 women each year in Norway receive an unnecessary cancer diagnosis and are accordingly given cancer treatment (radiation treatment, chemotherapy and hormone treatment) without deriving any benefit from it.
Limitation of liability: Data from the Norwegian Cancer Register have been used in this study. The interpretation and reporting of these data are the sole responsibility of the authors, and have not been subject to approval by the Cancer Register.
- 1.
Cancer in Norway 2009. Oslo: Kreftregisteret, 2011.
- 3.
Zahl PH, Mæhlen J. Overdiagnostisering ved mammografiscreening. Tidsskr Nor Lægeforen 2004; 124: 2238-9. [PubMed]
- 7.
Zahl PH, Gøtzsche PC, Mæhlen J. Natural history of breast cancers detected in the Swedish mammography screening programme: a cohort study. Lancet Oncol 2011; 12: 1118 – 24. [CrossRef]
- 8.
Statistisk sentralbyrå. http://statbank.ssb.no/statistikkbanken (29.8.2011).
- 10.
Suhrke P, Mæhlen J, Zahl PH. Hormone therapy use and breast cancer incidence by histological subtypes in Sweden and Norway. The Breast Journal 2012 (in press).
- 15.
Legemiddelforbruket i Norge. www.legemiddelforbruk.no (29.8.2011).
- 16.
Zahl PH, Mæhlen J, Welch HG. Breast cancer overdiagnosis with screening mammography – reply. Arch Intern Med 2009; 169: 1000 – 1. [CrossRef]
- 17.
Ernster VL, Barclay J. Increases in ductal carcinoma in situ (DCIS) of the breast in relation to mammography: a dilemma. J Natl Cancer Inst Monogr 1997; nr. 22: 151 – 6.