Three-dimensional representation of the heart by ultrasound (three-dimensional echocardiography) has in principle been available for the past two decades. The method has been assessed at Oslo University Hospital, Rikshospitalet, since the mid-1990s (1, 2), with the aim of developing better techniques for calculating volume and left ventricular mass. These are important objectives for measuring the left ventricular function which have significance for treatment and the patient’s prognosis. In the past few years there has been an exponential technological development – the use of new ultrasound probes (multi-linear probes) in particular has made it possible to achieve real-time imaging of the heart with better temporal and spatial resolution (3). In addition, more advanced tools have been developed for off-line analysis.
It is now also possible to do three-dimensional imaging using transoesophageal echocardiography. This permits an even more accurate anatomical representation of structures, which is particularly useful in diagnosing mitral valve disease. In the following, we will review the method based on studies of the literature and on our own experience.
Material and method
This article is based on a discretionary selection of articles found by a literature search of PubMed using the following search words: 3D echocardiography and – mitral stenosis (120 hits), – mitral regurgitation (398 hits), – aortic stenosis (191 hits), – aortic regurgitation (398 hits), – tumours (117 hits), – stress echo (33 hits), – ASD (57 hits), – VSD (27 hits). The search was concluded on 30 December 2011. Only literature published in the English language was assessed. The hits were reviewed and a discretionary selection made based chiefly on the findings of randomised, controlled studies and systematic review articles.
How can a representation be «three-dimensional»?
There are several different methods for producing a «three-dimensional» representation or image on the ultrasound screen or work station. The best known method is to view the image through 3D glasses, which allows it to appear in stereoscopic vision. However, many people experience tired eyes wearing 3D glasses, and alternative methods are therefore often preferred.
Colour coding can be used to produce an effect of depth, whereby the structures existing at the same depth are coded with the same colour and intensity. Light-and-shade effects also enable the image to be interpreted more three-dimensionally. Finally, a holographic screen has been developed which gives a true, three-dimensional representation of the heart without the viewer needing to wear 3D glasses (Setred, Forskningsparken, Oslo).
Volume analysis
Automatic volume analysis indicating the left ventricular ejection fraction (EF) can be performed from three orthogonal planes (4) or from the full volume cardiac dataset, with representation of the volume curve in the entire cardiac cycle (Figure 1). Theoretically, 3D echocardiography has an advantage in that the volume is not calculated from a geometric model, but can be estimated directly from a full volume recording. This also avoids cutting through the apex so that the volume is artificially low, which is frequently the case with conventional two-dimensional imaging. The method has been validated against cardiac MRI scans, with good agreement obtained for left ventricular volumes and ejection fraction (2, 4) – (7).
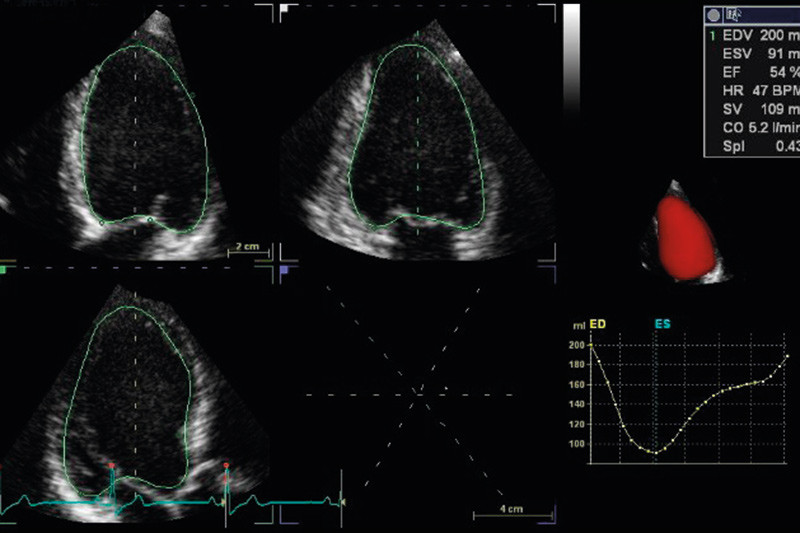
However, it has proved more difficult to estimate right ventricular volume using a three-dimensional technique (8). This is partly because the right ventricle has a more complex geometrical shape, which makes it difficult to obtain the entire volume in one recording, but also in particular because the free wall of the right ventricle is thin and more anteriorly situated, where it is difficult to get good echo signals.
Heart valve disease
The evaluation of heart valve disease using 3D echocardiography has been validated in different studies. To date, the best documented have been diagnostic studies of pathological mitral valves (9) – (12). During the last decade, there has been a change in the surgical treatment of mitral valve disease. New surgical techniques have enabled the repair of mitral defects in many patients who would previously have had to have valve replacement. Reparative surgery of this kind requires a very high degree of precision in diagnostics of the mitral valve’s form and function, to enable the correct surgical method to be employed. In our own department, we use 3D echocardiography to investigate patients with mitral valve regurgitation, to enable us to locate exactly the damage to the valve (Figure 2). We have benefited especially from the so-called «surgeon’s view» from the left atrium, where the entire mitral valve can be represented in the same image as shown in Figure 2.
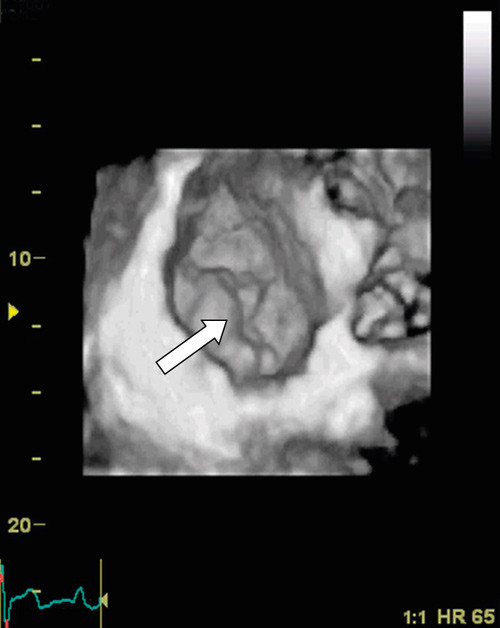
Currently, two-dimensional transoesophageal echocardiography is required in order to determine which surgical technique is desired to treat mitral valve prolapse. Interestingly, it was recently demonstrated that 3D external echocardiography (transthoracic) was equally as good as 2D transoesophageal echocardiography in localising mitral valve prolapse (10). Three-dimensional transoesophageal echocardiography was even more precise in assisting diagnosis of commissural prolapse, which can be difficult to repair. This is important information to have when planning which surgical method is the more appropriate as regards artificial valve versus valve-preserving, reparative surgery. Three-dimensional echocardiography is now routinely performed at a number of European cardiac surgical centres prior to plastic repair of the mitral valve.
Quantitation of regurgitation is also an important part of the diagnostic picture in patients with mitral valve regurgitation. Two-dimensional colour Doppler has clear weaknesses in these diagnostics. Studies show that 3D colour Doppler echocardiography is a more robust measurement for estimating valve regurgitation and calculating the effective regurgitant orifice area (EROA) (13, 14). Three-dimensional colour Doppler echocardiography offers unique possibilities for multiple-plane imaging of vena contracta. A simple technique permits planimetry of the area corresponding to the vena contracta (14, 15).
Several studies have been produced on the assessment of aortic valve stenosis using both direct planimetry (16) – (19) and with 3D colour Doppler echocardiography for determination of stroke volume and derivation of the area with continuity equation (16, 20). Particularly in the presence of septal bulging, which is common among the elderly, the latter method was more precise than using conventional 2D echocardiography (20). Three-dimensional echocardiography also enables quantitation of aortic insufficiency, where two-dimensional echocardiography currently has evident weaknesses (18).
With new techniques, such as percutaneous procedures to treat valve disease (implantation of artificial aortic valve) and repair of mitral valve regurgitation using a transcatheter technique (MitraClip), 3D echocardiography will have a central place in cardiac procedures (21). In the next few years, 3D echocardiography may also be used to assist new percutaneous techniques, not only MitraClip. Techniques are currently being developed (in animal models) for other purposes such as replacement/repair of mitral valve chords and insertion of mitral rings and new percutaneous mitral valves.
In patients with mitral stenosis, the method has also proved to be more precise than the techniques used routinely today (12, 22) – (24). The advantage of 3D echocardiography is the opportunity it offers for defining the image plane «en-face» through the smallest orifice area of the stenosis, so that the true stenosis area can be estimated more precisely.
We have also found 3D echocardiography useful in patients with residual mitral regurgitation after mitral valve surgery. In paravalvular regurgitation, a good overview can be obtained of the localisation and extent of the regurgitation. Other clinical problems for which we employ 3D echocardiography are in diagnosing endocarditis. In addition to more precise localisation, it enables a better overview of the extent of abscesses and any fistula formation (Figure 3).
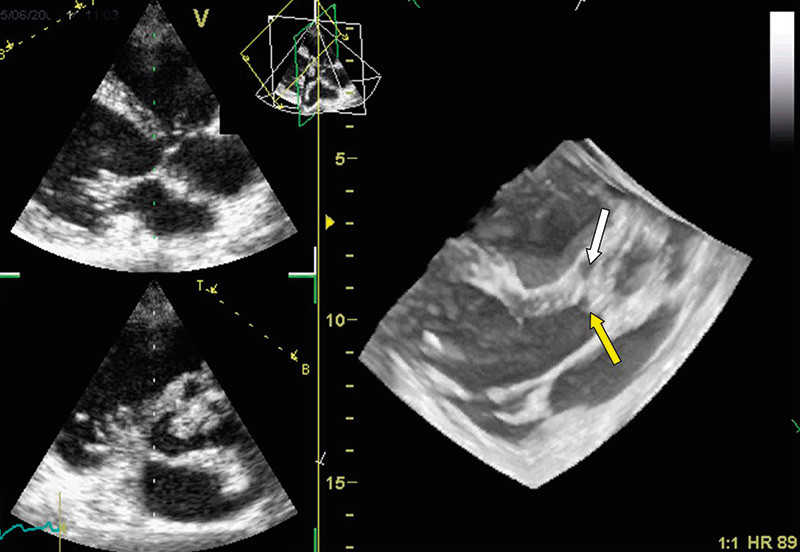
Intracardiac masses
Assessing intracardiac masses can be challenging, even with two-dimensional transoesophageal echocardiography. We have found 3D echocardiography useful in assessing myxomas (the extent of the tumour, whether it is stalked, and in one case the myxoma’s location in the left ventricle in relation to the papillary muscles). It has also enabled us to exclude left auricular thrombus where this could not be excluded by 2D transoesophageal echocardiography.
Apical «masses» can be difficult to interpret, particularly with respect to whether thrombus is present. Here, too, 3D echocardiography can have its advantages compared with 2D echocardiography (25) – (27). Heart valve thrombosis can also be detected with the new technology and, where this is suspected, 3D echocardiography is carried out routinely in our department to identify the condition.
Cardiomyopathies
Using 3D echocardiography with full volume recording, the left ventricle can be represented as multiple short-axis images on the screen (Figure 4). In heart muscle diseases such as hypertrophic cardiomyopathy, a better overview can be obtained of where the hypertrophy is distributed. This is particularly useful with more apical variants, as they can be overlooked in routine echocardiography. Some rarer forms of cardiomyopathy, such as non-compaction cardiomyopathy, have characteristic features which can be reproduced easily with 3D echocardiography (Figure 5). However, the additional value of 3D echocardiography in various cardiomyopathies has not as yet been well documented.
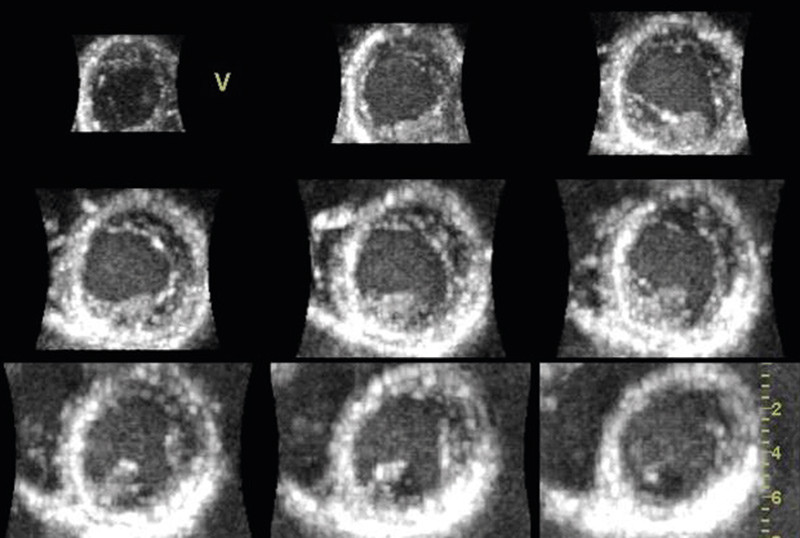
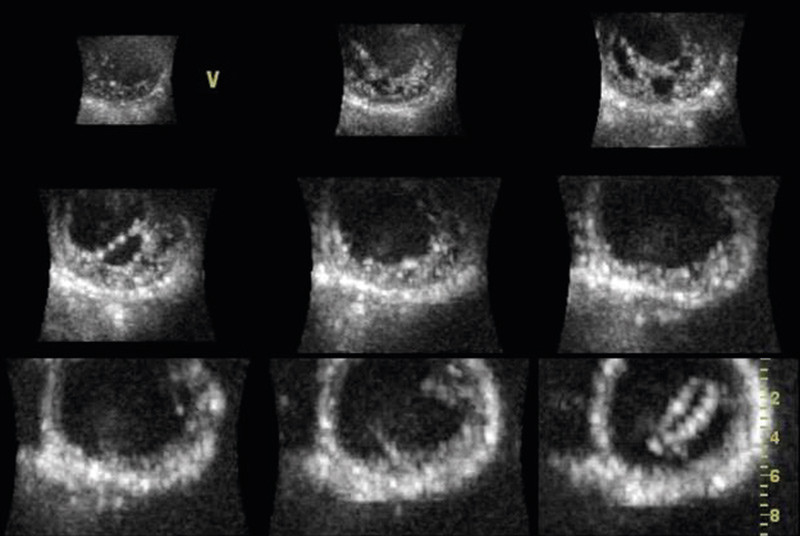
Congenital heart defects
Three-dimensional echocardiography has a number of possible applications in diagnosing patients with congenital heart defects. Assessing atrial septal defect (ASD) (28) – (33) can in some cases be difficult even with transoesophageal echocardiography. This is particularly true when it comes to assessing the scope of the defect and how good the possibilities are of identifying a tissue edge for attaching an artificial plug. Where there is a large defect with only a small tissue edge, one may be best served with surgical closure.
Similarly, three-dimensional ultrasound has been used for diagnostics and for guiding catheter-based closure of muscular ventricular septal defects (34). In more complex heart defects, three-dimensional echocardiography could also theoretically be of benefit.
Myocardial function
In addition to global measurements of left ventricular-function, such as ejection fraction, methods have been developed for calculating regional myocardial function by determining tissue deformation and deformation velocities (35). These calculations have been based on standard two-dimensional echo recordings and are limited to a single plane. Since the heart is a three-dimensional structure with constant mass, deformation in one axis (x) will cause deformation in other axes (y, z). Theoretically, this difficulty can be resolved by using three-dimensional recordings in which the deformation can be calculated in all these axes simultaneously.
The use of 3D echocardiography in diagnostic tests for reversible ischaemia (stress echocardiography) also has a significant theoretical advantage. In the past few years, three-dimensional stress echocardiography has been validated against two-dimensional stress echocardiography (5, 36, 37). Sensitivity is still somewhat lower for 3D echocardiography, particularly as regards the anterior wall and lateral wall. The specificity for the two methods is fairly similar (80 – 90 %). It continues to be a challenge that the temporal resolution is limited, particularly in high cardiac frequencies. Further development of the technology will, it is hoped, improve the temporal and spatial resolution of the 3D echo recordings.
Resource use
In busy clinical practice, it is important to include a cost-benefit analysis in the tests and investigations we undertake. Three-dimensional echocardiography will for the time being be a supplement to complete two-dimensional echocardiography. In our view, all hospitals offering valve-preserving surgery should have 3D echocardiography as part of their range of technical equipment. It is not only tertiary medical centres that will benefit from the technology – the examples above demonstrate the benefits which other hospitals and specialists in private practice may gain from the additional information that 3D echocardiography can provide. It is essential to be dedicated to the task and to spend some time at the start on learning the technique, and maintaining the skills acquired.
Conclusion
With improved technology and hence improved imaging, three-dimensional echocardiography has become an important supplement to today’s standard echocardiography, both transthoracic and transoesophageal. The method is currently best documented for quantitation of left ventricular volume and ejection fraction and for qualitative assessment of mitral valve disease.
Tabell
| Main points |
|
- 2.
Rodevand O, Bjornerheim R, Aakhus S et al. Left ventricular volumes assessed by different new three-dimensional echocardiographic methods and ordinary biplane technique. Int J Card Imaging 1998; 14: 55 – 63. [PubMed]
- 31.
Price MJ, Smith MR, Rubenson DS. Utility of on-line three-dimensional transesophageal echocardiography during percutaneous atrial septal defect closure. Catheter Cardiovasc Interv 2010; 75: 570 – 7. [PubMed]
- 36.
Abdelmoneim SS, Bernier M, Dhoble A et al. Assessment of myocardial perfusion during adenosine stress using real time three-dimensional and two-dimensional myocardial contrast echocardiography: comparison with single-photon emission computed tomography. Echocardiography 2010; 27: 421 – 9. [PubMed] [CrossRef]