From global to local: a new understanding of cardiac electromechanical coupling
Since the publication of Sydney Ringer’s seminal article in 1883, it has been known that Ca2+ plays a key role in the contraction of the heart (1). Research in the 1900s demonstrated that the contractility of the heart is adapted to the organism’s needs through the regulation of the concentration of Ca2+ in the heart muscle cells. However, it took a hundred years before Ringer’s discovery of this regulation was explained at molecular level. The electromechanical coupling of the heart has now been described in detail, but we still have no explanation for and cannot predict ordinary phenomena such as impaired contractility and arrhythmias in connection with heart failure.
This paper describes a cardiophysiological approach that has attracted growing attention over the past decade. The approach studies the interaction between proteins in delimited «microdomains» in cells. The binding protein ankyrin-B and the Ca2+ channel IP₃R (inositol 1,4,5 triphosphate receptor) are examples of proteins that probably have to be understood in this connection.
Method
The paper is based on the authors' own research in the field and on a discretionary selection of articles found by means of a literature search in PubMed. The following search terms were used: «ANK2», «ankyrin-B», «ankyrin-B syndrome», «Long QT syndrome type 4», «IP₃R». This resulted in 140 hits in PubMed on 15 February 2012.
Since the focus of the article is on Ca2+ homeostasis in ventricular cells, we placed emphasis on articles that mainly concern ventricular cells or ventricular arrhythmias. The hypotheses formulated in the article concerning the function of ankyrin-B and IP₃R are our own, but in line with recent review articles in the field.
Classical understanding of the heart’s electromechanical coupling
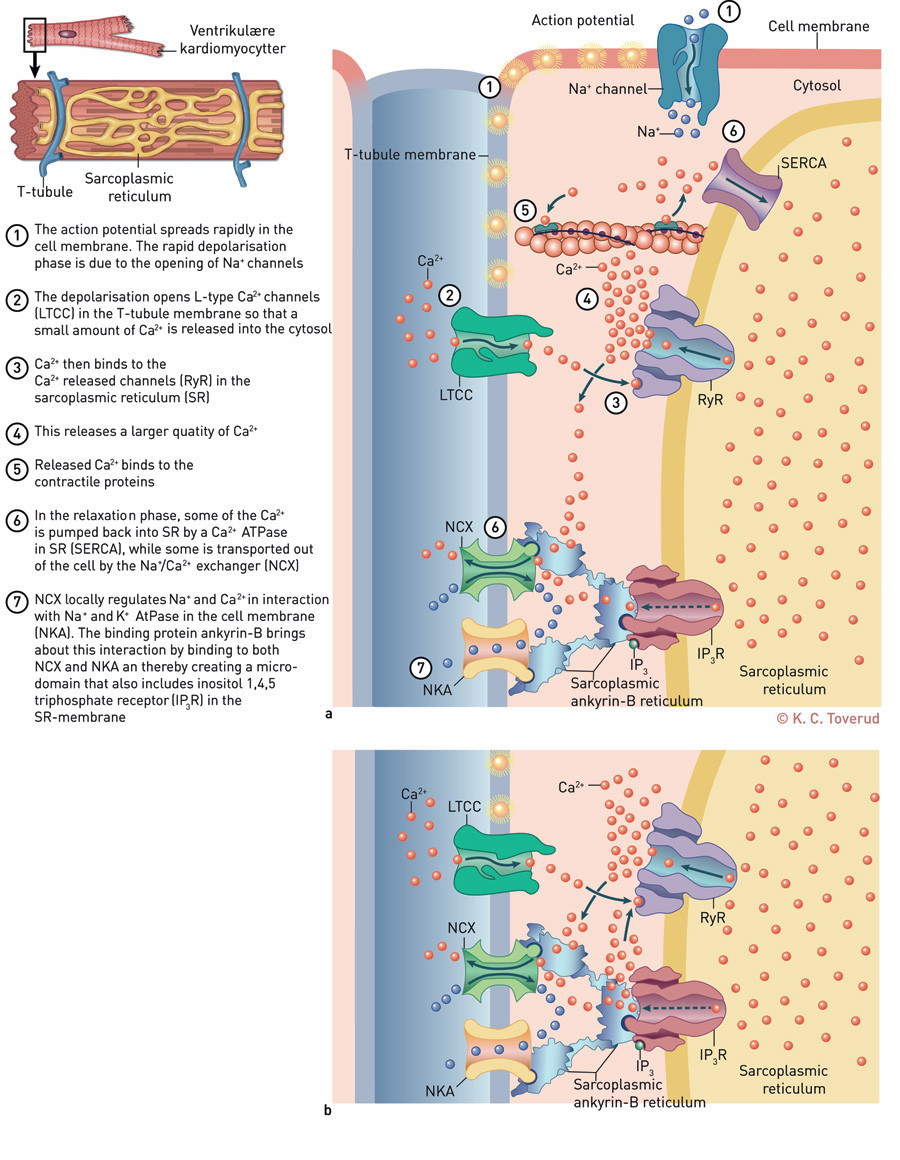
The theory of Ca2+ -induced Ca2+ release in the heart muscle cells describes a system for reinforcing the Ca2+ signal that has several control points (Fig. 1a) (2). On each activation (depolarisation) a small amount of Ca2+ is released into the heart muscle cells through L-type Ca2+ channels in the cell membrane. Entering Ca2+ binds to a Ca2+ release channel in the heart muscle cells’ Ca2+ reservoir, the sarcoplasmic reticulum. These Ca2+ release channels, the ryanodine receptors (RyR), can be opened by a small amount of Ca2+ in the cytosol. When the RyR opens, Ca2+ flows out into the cytosol, where its concentration is therefore multiplied 5 – 15 times compared with the diastole. Ca2+ then binds to the contractile proteins and thereby starts the muscle contraction. The systolic concentration of Ca2+ in the cytosol determines the strength of the contraction. The coupling of Ca2+ and contraction thus have four important control points (3): First, the actual influx of Ca2+ through the cell membrane, which starts the process, is subject to regulation.
This is important, since a large influx will result in larger releases from the Ca2+ reservoir. Second, the sensitivity of the RyR can be altered so that more Ca2+ is required for a large release. Third, the quantity of Ca2+ in the sarcoplasmic reticulum can be adjusted. This is an important control point, since the quantity of Ca2+ that is released on each activation increases exponentially with the quantity of Ca2+ in the sarcoplasmic reticulum. Fourth, the ability of the contractile proteins to react to Ca2+ can be adapted to the organism’s need for an increased or reduced stroke volume.
We have gradually learned much about these control points and how they are regulated in physiological and pathological situations. Over the past two decades, particular attention has been focused on RyR and its role in the pathophysiology of both weakened contraction and arrhythmias (4).
From «global» to «local» Ca2+ handling
The description of the connection between Ca2+ and contractions is based on an understanding of the interaction between proteins in the heart muscle cells. It has long been known that the diffusion of Ca2+ in cytosol is restricted because of the buffer effect of Ca2+ -binding proteins and geometrical obstacles (5). The diffusion coefficient of Ca2+ in water is 1 000 µm²/s, whereas in cytosol it is estimated to be 10 – 20 µm²/s (6). Theoretically, such diffusion would still be enough for Ca2+ released from a single Ca2+ channel in the cell membrane to reach large parts of the cell in the course of one contraction cycle.
However, one problem with a system based on diffusion from few channels in the cell membrane would be large concentration differences within the cell and regional time differences for activation of the Ca2+ release channels. The extremely rapid (~1 ms from activation of the cell) and normally very homogeneous Ca2+ release in different areas points to a closer connection between membrane activation and Ca2+ release (7). There was therefore good reason to assume that depolarisation-contraction coupling must proceed very «locally», but at the same time in synchrony throughout the cell.
This process has largely been studied through phenomena that can be observed «globally», for example the total concentration of Ca2+ in the cytoplasm and the overall flux from all membrane channels. In the 1990s, however, improvements in microscope technology made it possible to describe the local processes in greater detail. Thus, it was confirmed that the rapid increase of Ca2+ in the cytosol when the heart muscle cells are activated is a result of many RyR channels opening simultaneously (8).
These RyR channels occur in well-defined «clusters» in the sarcoplasmic reticulum. L-type Ca2+ channels in the surface membrane are closely linked to a RyR cluster. This local structure constitutes the «dyad», the ultrastructural basic unit in the coupling between depolarisation and Ca2+ release in the heart muscle cells. The rapid spread of the action potential in the cell membrane (the whole cell is depolarised in the course of ~ 0.1 ms) (9) activates L-type Ca2+ channels in the whole cell almost simultaneously, and the close coupling with RyR in the dyads ensures synchronicity and homogenous Ca2+ release in the cell. It has been demonstrated that the dyad’s structure can be disturbed, for example by heart failure, and that this results in heterogeneous, dyssynchronous and slower Ca2+ release (7, 10).
Dyads – one of many microdomains?
The structure of the dyad can thus explain essential physiological and pathophysiological phenomena associated with the Ca2+ homeostasis of heart muscle cells. However, the development of new molecular technologies has shown that the dyad is not the only local unit in the heart muscle cells, but rather one of a number of microdomains (11). An example of new discoveries that have accelerated this kind of thinking is the ankyrin-B syndrome, or long QT syndrome type 4.
A large number of sudden cardiac deaths as a result of arrhythmias in a French family led to genetic analyses with identification of mutations in the gene (ANK2) that encodes the ankyrin-B protein (12). Experimental animal studies later confirmed that a reduced quantity of ankyrin-B in the heart entails a higher risk for ventricular arrhythmias and sudden death triggered by increased sympathetic activity (13).
It was shown that cells from the ventricles of mice with a reduced amount of ankyrin-B more frequently depolarised spontaneously during the repolarisation phase (Phase3) and the resting phase (Phase 4) of the action potential. Such early and delayed afterdepolarisations may be caused by disturbances in Ca2+ homeostasis and spontaneous Ca2+ release from the sarcoplasmic reticulum that activates the Na+/Ca2+ ion exchanger (NCX). The main task of NCX is to transport one Ca2+ ion out of the cell in exchange for three Na+ ions during the cell’s relaxation phase. This creates a net charge influx that has a depolarising effect on the cell membrane. This depolarisation is balanced by other ion fluxes having the opposite effect, and the surplus Na+ is removed by the Na+/K+-ATPase. However, by releasing Ca2+ from the sarcoplasmic reticulum in phase 3 or phase 4 of the action potential, the NCX flux may depolarise the cell membrane sufficiently to trigger a spontaneous action potential. Spontaneous action potentials are seen as ventricular extrasystoles and may start arrhythmias. This causal chain of events was demonstrated during β-adrenergic stimulation in mice with reduced ankyrin-B: Spontaneous Ca2+ release caused afterdepolarisations, which triggered spontaneous action potentials. The mice also had an increased incidence of extrasystoles and sustained ventricular arrhythmias.
Not all mutations in ANK2 predispose patients for arrhythmias; the clinical effect of the mutation depends on the significance of the mutation for the ankyrin-B function (14, 15). Despite this insight into the mechanism of the disease, prophylaxis of ankyrin-B syndrome is limited to β-adrenergic receptor agonists and implantable defibrillators, although other measures such as sympathetic denervation have also been proposed (16). More specific treatment will depend on the answer to the remaining question: How does reduced ankyrin-B function bring about changes in the Ca2+ homeostasis that increase the risk of arrhythmia?
Ankyrin-B and IP₃R – new kids on the block, or a whole new neighbourhood?
Ankyrin-B is a binding protein, ensuring that proteins involved in Ca2+ handling of heart muscle cells are retained in particular microdomains (17). Interestingly, proteins in the classic dyad are not among these. Thus, Ankyrin-B ensures local interaction between other proteins. The loss of this local interaction may be of great significance for Ca2+ homeostasis, at worst with fatal consequences. Thus more microdomains than the dyad have a bearing on Ca2+ handling.
Ankyrin-family proteins stabilise and bind proteins in the sarcolemma and the sarcoplasmic reticulum to the cytoskeleton of the cells (17). Ankyrin-B binds three proteins that are involved in Ca2+ homeostasis: NCX and the Na+/K+-pump (NKA) in the cell membrane and also the inositol 1,4,5 triphosphate receptor (IP₃R) in the sarcoplasmic reticulum (18) (Fig. 1a). It has long been known from the effect of digitalis that the interaction between NCX and NKA is essential for Ca2+ homeostasis in the heart: digitalis inhibits NKA, and therefore causes an accumulation of Na+ in cytosol. This prevents NCX from exchanging Ca2+ with Na+ from the extracellular fluid. As a result, the Ca2+ concentration in the cytosol increases. We have previously shown that NKA is co-localised with NCX (19, 20). As a result of the binding to ankyrin-B, these proteins form part of a microdomain with other proteins, including IP₃R. Interestingly, IP₃R is also a Ca2+ channel in the sarcoplasmic reticulum (21). Thus RyR is not the only route for Ca2+ release from the sarcoplasmic reticulum.
IP₃R is stimulated by IP₃, which is released when phospholipase C (PLC)-linked receptors in the cell membrane are activated by endothelin-1, angiotensin II or noradrenaline (22). The result is that IP₃R opens and releases Ca2+ from the sarcoplasmic reticulum to the cytosol. This process does not require prior depolarisation of the cell membrane. This Ca2+ release is therefore distinct from the Ca2+-induced Ca2+ release from the RyR channels. In view of the fact that there are normally far fewer IP₃R than RyR, approx. 1: 50 (23), and Ca2+ release from each IP₃R is far slower and more limited than the Ca2+ release from RyR, one wonders what function the release from IP₃R has. This is an unanswered question, but there is reason to believe that the answer involves Ca2+ signalling in the IP₃R microdomain, and that the connection with heart disease may be due to internal changes in the latter or in the interaction with other microdomains (24).
IP₃R – regulator of RyR or safety valve?
Although it is known that IP₃R is also found in the membrane of the sarcoplasmic reticulum, its localisation in relation to the RyR clusters remains undetermined. The localisation and functional interaction with RyR are crucial for the significance of abnormal IP₃R function in the ankyrin-B syndrome and heart failure.
We have formulated two hypotheses regarding the role of IP₃R (Fig. 1a, Fig. 1b): If IP₃R is co-localised with RyR, it is possible that it plays a part as regulator of the Ca2+-induced Ca2+ release. Ca2+ release from IP₃R can change the local Ca2+ concentration around RyR and thereby influence RyR’s sensitivity to Ca2+ (Fig. 1b). This would be an example illustrating that an interaction between different proteins in the same microdomain in the heart muscle cells can result in more precise regulation of the contraction.
Alternatively, IP₃R may play a part as a «safety valve» in the sarcoplasmic reticulum (Fig. 1a): Localisation of IP₃R in the same microdomain as NKA and NCX means that Ca2+ released through IP₃R can be easily transported out of the cell in a controlled manner. It is well known that accumulation of Ca2+ in the heart muscle cells has a positive effect in the form of increased inotropy. But if the accumulation becomes too large, Ca2+ will «leak» in an uncontrolled manner from the sarcoplasmic reticulum through RyR. As previously described, such RyR leakage could activate NCX, lead to afterdepolarisations and trigger arrhythmias. In recent years, however, it has been found that leakage from the sarcoplasmic reticulum may also take place «silently», independently of RyR (25). It is conceivable that such leakage could take place through IP₃R. It is therefore very interesting that heart failure, the end-point of many heart diseases, is associated with an increased amount of IP₃R in the heart (22, 26).
Heart failure is also associated with increased activity in the sympathetic nervous system and in the endothelin and renin-angiotensin systems. Since this means increased production of IP₃, IP₃R and its microdomain may play a larger part in heart failure than in healthy hearts (22). At present it is not known whether this is a protective mechanism to prevent over-filling of the sarcoplasmic reticulum, whether it contributes to weakened contractility by reducing the Ca2+ reservoir, or whether it increases the risk of arrhythmias by making RyR «hypersensitive». Clearly, both this and other microdomains in heart muscle cells should be studied further to enable an understanding of how the heart functions under normal circumstances and during illness.
Conclusion
Since Ringer’s discovery of the importance of Ca2+ for muscle contraction, heart physiology has explained an increasing number of physiological phenomena by moving the focus from organ to cell to protein level. The research of recent decades has demonstrated the need to also study structure and function in delimited and adjacent microdomains in heart muscle cells. Examples of new insight into the significance of ankyrin-B and IP₃R show that these may be of great importance to an understanding of diseases with weakened contractility and increased risk of arrhythmias.
Tabell
| Main points |
|
- 1.
Ringer S. A further contribution regarding the influence of the different constituents of the blood on the contraction of the heart. J Physiol 1883; 4: 29 – 42.3. [PubMed]
- 2.
Fabiato A. Calcium-induced release of calcium from the cardiac sarcoplasmic reticulum. Am J Physiol 1983; 245: C1 – 14. [PubMed]
- 19.
Swift F, Sjaastad I, Sejersted OM. Er endret regulering av Na+ årsak til svekket kontraktilitet i myokard ved hjertesvikt? Tidsskr Nor Lægeforen 2003; 123: 3036-40. [PubMed]
- 24.
Louch WE, Sejersted OM, Swift F. There goes the neighborhood: pathological alterations in T-tubule morphology and consequences for cardiomyocyte Ca2+ handling. J Biomed Biotechnol 2010; 2010: 503906.