The first committees of research ethics were established in the mid-1960s, in the US and elsewhere, as a countermeasure to unethical research practices. It was recognised that leaving these assessments to the researchers themselves provided an inadequate safeguard (1). Norway, however, was different. The immediate background for looking at the issue of establishing committees of research ethics was the recommendation in the 1975 Declaration of Helsinki to the effect that «any experiment involving human beings (...) must be submitted to an especially appointed, independent committee for review, comment and guidance» (2). The driving forces to achieve this was the Council of Medical Ethics, its chairman Erik Enger (born 1927), and the Council of Medical Research (RMF) under the Research Council of Norway (NAVF).
The Council of Medical Research arranged a number of major seminars, the first one in November 1976, as a result of which the Norwegian Medical Association submitted a specific proposal to establish committees of research ethics, in a letter dated 26 May 1977. As formulated succinctly by Erik Enger: «The revised declaration indicates that even in our country, the time has now come to discuss matters relating to the establishment of such committees and the issue of their function» (4).
Material and methods
We have examined the material that has been submitted to the National Archives by the Research Council of Norway, the Ministry of Social Affairs and the Ministry of Culture and Education. The records of the Regional Committees for Medical and Health Research Ethics (REC) South and East contain correspondence with various ministries, the Norwegian Medical Association and the University of Oslo, as well as a complete archive of all projects that have been reviewed since 1987. Projects that were addressed by the Council of Medical Research before 1987 have been deposited in the National Archives.
Initial preparations prior to 1977
Even though the time did not come until 1977, the initiative and the prevailing interest in research ethics showed that the ground was well prepared. Following a proposal from the Nordic medical associations and research councils, a Nordic working group was appointed with Erik Enger, Clarence Blomquist (1925 – 79) and Povl Riis (born 1925) as members. The group’s mandate was to submit a proposal for a revised Declaration of Helsinki. Its recommendations included a proposal that test protocols should be submitted to an independent committee of ethics (3, 5).
In a memo from the Council of Medical Research, dated 2 November 1977, it transpires that the matter had been put on the agenda in 1974, with a view to establishing «ethical rules for medical research» (3). The Council of Medical Research glanced to Sweden, where institutional committees of ethics had been introduced in 1966 according to the American model. Some similar institutional committees of ethics had been established in Norway as well (5).
Planning and establishment 1976 – 85
The meeting of the Council of Medical Ethics on 6 November 1976 marked the start of the establishment of ethics committees in Norway (3). The authorities were also interested (6). A working group was appointed to prepare a more specific proposal. The working group consisted of representatives from the Directorate of Health and the Norwegian Association of Local and Regional Authorities (KS), as well as medical doctors Egil Gjone (1925 – 99) and Erik Enger and the solicitor Åsmund Hodne (born 1940) (3).
To determine how this could best be addressed in Norway, the working group was especially interested in examining the model that had been introduced in Denmark. The Danish model involved regional committees and one general national committee. These committees should be broadly composed, and at the time there was a notion that they should include four medical doctors, two laypersons and a nurse. The Norwegian model incorporated this approach to a large extent, and the working group submitted a proposal for a system that included three levels of ethical approval (Figure 1). On the lowest level the group proposed the establishment of local ethics committees in the hospitals. These should have an advisory role, and their function should be to assess ethical aspects of research projects at the hospital in question.
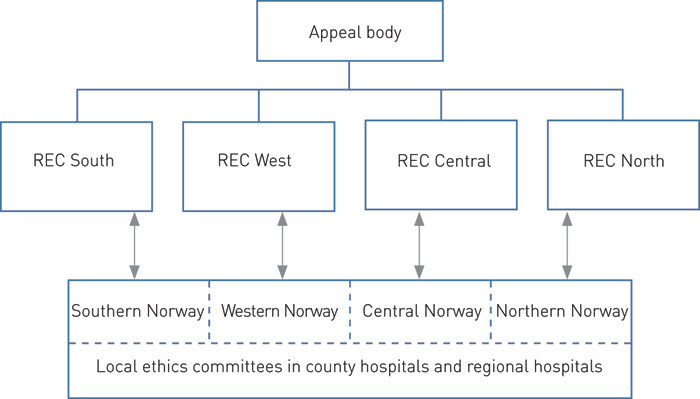
The next level was referred to as supervisory committees of ethics. The group proposed that these should have decision-making authority. The function of these committees should be to decide in matters in which the local committees were in doubt, and act as a body of appeal for researchers who were dissatisfied with the decisions made by the local committee of ethics. In a letter from 1977, the group further elaborated that «in addition to medical research in a narrow sense, these bodies also ought to assume responsibility for assessing research in veterinary medicine, dentistry research, sociological research and other forms of research involving human beings» (3).
Furthermore, the supervisory committees of ethics should be geographically dispersed. They were therefore also referred to as «nationally dispersed committees». The geographical dispersal implied one committee in each of the regions of Southern Norway, Western Norway, Central Norway and Northern Norway. The composition of the committees should include two representatives from the universities, one hospital owner, one representative from the Research Council of Norway, one hospital doctor, one representative of the regional authorities and one layperson. Finally, a national body of appeal for the regional committees was to be established.
There was clear scepticism as to the need for such an ambitious organisational model. Case documents from the Ministry of Social Affairs have a number of notes added in the margin (3). One of them, dated 6 June 1977, says that «this will be too intricate», with the recommendation that «we really ought to find a simpler solution» (3). In a letter to the Norwegian Medical Association dated 7 October 1977, the Ministry of Social Affairs recommends that «the simplest possible model» ought to be tried first (3). And that’s what happened.
The simplest possible model
Following a proposal submitted by the Norwegian Medical Association in a letter dated 21 July 1978, the Research Council of Norway established a separate ethics commission under the Council of Medical Research (5). It was proposed that the commission also should include «external parties», i.e. non-doctors. The Council of Medical Research appointed the commission pending the establishment of «a central committee», and in order to comply with the obligation of prior review of projects by an independent committee of ethics as required by the 1975 Declaration of Helsinki. The Council of Medical Research prepared a mandate and forms to cover «all research that pertains to human health (clinical medicine, dentistry and public health, experimental medicine, etc.)» (5). The first committee consisted of Professor Jarle Ofstad (born 1927) as chairman, Professor Egil Gjone at The National Hospital, Torbjørn Mork, Director of Health (1928 – 92) and the «external parties» – Ingse Stabel (born 1946), Principal Officer in the Ministry of Justice and Professor Jacob Jervell (born 1925) of the Faculty of Theology at the University of Oslo. It held its first meeting on 14 September 1979 (5).
The work nearly comes to a halt
The establishment of the commission under the Council of Medical Research ensured compliance with the requirement for prior approval of projects. This may in itself have been a reason why the matter lost momentum.
In addition, the largest hospitals had established their own local committees. The foremost pioneer in the establishment of Norwegian ethics committees, Erik Enger, chaired the local committee at Ullevål Hospital. In the context of planning the establishment of further local committees in the hospital, he stated that «in practice, this means that most likely there will be no regional committees at all» (7). His opinion was shared by Hans Erik Rugstad, the later chairman of a Regional Committee on Research Ethics: «Work on the regional ethics committees appears to have come to a halt» (8).
In a summary of the efforts to establish regional ethics committees issued by the Ministry of Social Affairs to the Ministry of Church and Education in January 1981 it transpires, however, that the lack of clarity surrounding the financial aspects, rather than the need and the scope, was the key problem (9). There was also disagreement regarding the organisation, the need for a secretariat and the remuneration to committee members. However, the Ministry deemed the proposal from the Research Council of Norway, that the faculties of medicine in the respective regions should take on the function of the secretariats, as «a natural solution» (9). And that’s also how it turned out, eventually.
One of the main reasons for the slow grind of the matter was the report on research and ethical responsibility submitted by a commission under the Main Committee of the Norwegian Research Council in the autumn of 1981, under the leadership of Knut Erik Tranøy (1918 – 2012), Professor of Philosophy. The report complains that Norway «is the Nordic country which is lagging furthest behind with respect to establishment of committees of biomedical research ethics», and it was recommended «in very strong terms» that the main committee should request the Government to let the proposed arrangements «enter into force immediately» (10).
The following year, during question time in the Storting, Grete Knudsen (born 1940) asked «when the minister can promise that the committees will be established» (11). Minister Lars Roar Langslet (born 1936) reported that the universities had taken a positive view on incorporating the secretariats in the medical faculties, and that he was confident that «the Ministry of Social Affairs will support this measure by funding the operating costs». As regards his own ministry, he was confident that «in the very next days to come, we will find an arrangement for the secretariat function». They didn’t, however.
The first period of operation 1985 – 89 in the South and the East
Members were appointed to five regional committees for medical research ethics from 1 July 1985 (12). Each committee was to have seven members with deputies. These included representatives of the medical faculty, the hospital owner, the health authorities and the Norwegian Nurses Association, in addition to representatives with competence in ethics and adjudication and representatives of the medical profession. The first period of operation would turn out to be a difficult one for the committees in the southern and eastern regions. While the other regions started their work relatively shortly after the appointment, the southern and eastern regions would not hold their first meetings until the autumn of 1987.
The main problem in the initial phase concerned the finances. While the other regions quickly could provide a part-time (50 %) secretary per committee, the southern and eastern regions had to make do with a part-time (50 %) position between them (13). Even though Minister Langslet had counted on support from the Ministry of Social Affairs and the universities had taken a positive view, it remained unclear who would be expected to cover the cost. The University of Oslo was definitely not going to contribute. The lack of clarity also rendered it difficult to recruit committee chairmen, since the candidates were worried about the workload that would be placed on them, because the amount of secretarial assistance available would be very modest. This became especially evident when both of the appointed chairmen for the southern and eastern regions resigned before a single meeting had been held. Even though this to some extent was caused by the financial situation, the implications constituted the main problem. In an interview with the Aftenposten newspaper on 24 October 1986, Olav Hilmar Iversen (1923 – 97) and Erik Enger stated: «The issues to be addressed by the committees concern human life and rights and the progress of medical science; these do not lend themselves to imprudent work» (14).
The chairmen of the two committees could be replaced, but the financial problems remained. The Ministry of Culture and Science was reluctant to provide funding, even though funds for a 50 % consultant’s position were produced in May 1986 (15). Neither ability nor willingness appeared much greater in the Faculty of Medicine, which was to provide the «infrastructure» for the work of the committees. In a sharply formulated letter to the Ministry of Culture and Science in November 1987, Tore Wie (born 1944), the first secretary of the southern regional committee, did not mince his words: «Everybody knows that the financial framework for the committees is extremely tight. For this committee, this is further highlighted by the fact that The National Hospital, as our landlord, has declined all financial support towards its physical establishment, apart from putting an empty, tucked-away and completely dilapidated office at our disposal. Not even the electrical fittings are appropriate for our needs» (13).
The first meetings in the South and East
In spite of the tucked-away location and meagre conditions, the first meeting took place in the autumn of 1987. REC East went ahead first and held its initial meeting on 16 September, while REC South followed suit on 11 December (16). The committee faced several challenges during its first period. In REC South’s annual report for 1988, which was the committee’s first full year of activity, several of these are pointed out (13). The budget was the recurring main problem. The limited budget meant that the committee had to restrict its number of meetings. For 1988, this resulted in six ordinary meetings, in addition to one extraordinary meeting. A total of 111 new cases had been submitted for that year. According to the annual report, the large caseload resulted in «several marathon meetings with an infinite number of cases on the agenda».
In March 1988, the new chairman of REC South, Professor Hans Erik Rugstad, wrote a very strongly worded letter to the dean of the faculty of medicine concerning «the financial situation and viability of the committee». He noted that it was «only with a narrow margin that this committee succeeded in being established at all», and he listed the empty office in the attic, no budget and unbearable working conditions for the secretary and the committee. Unless immediate steps were taken, Rugstad announced, «the committee will face a total breakdown with discontinuation as a result» (14). It worked. From 1 July veterinarian Tore Wie was employed in a full-time position by REC South (13), a budget was provided and the infrastructure was improved.
However, there was also time for reflection on content. For example, the minutes of one meeting report that several members were of the opinion that the Norwegian translation of the Declaration of Helsinki was too strict. Jon Wetlesen (born 1940), ethics expert in the committee, was especially concerned that the guidelines had turned into rules through the use of Norwegian equivalents of «shall» and «must», which ought to have been rendered as «should» (17).
The first projects
REC South’s annual report for 1988 gives a good indication of the types of projects that were submitted and assessed (13). Nearly 80 per cent of the projects involved clinical trials of drugs, and approximately 15 per cent concerned epidemiology. It is worth noting that the number of projects relating to clinical research was negligible and that projects from researchers other than medical doctors were totally absent, even though the committee included a representative of the nursing profession.
The first clinical trials that were submitted concerned mild to moderate hypertension, serious infections, rheumatoid arthritis and serious heart failure. In all these cases the committee was mostly concerned with satisfactory insurance, which had to be confirmed by the sponsor. This can be explained by the fact that the Act relating to product liability did not enter into force until 1 January 1989. In one case, the committee imposed requirements for the safeguarding of personal data before their transfer abroad, and it introduced some minor amendments to the information folder. There were no assessments of design, placebo or risk and benefit. Other noteworthy types of research include nutritional trials in cases of infantile autism and sudden infant death syndrome. For the nutritional trial project, the committee required comprehensive patient information in conformity with the Declaration of Helsinki. However, the committee also raised scientific objections to the protocol, including the requirement that it should not characterise the patient biochemically through analysis of urine and serum to identify the peptide patterns, because the project investigator had justified the trial with reference to the correlation between peptide patterns in serum and urine and the patient’s mental condition. The project was approved despite the objections from the committee.
The project on sudden infant death syndrome, which was to be undertaken by a medical student, involved measurement of exogenous and endogenous opioids with the aid of radioimmunoassay in spinal fluid, the corpus vitreum and serum, and using the sandwich technique to make an incision in the brain stem. The project also planned to interview the families concerned on matters such as nutrition and stress factors prior to the incident. The committee had no objections to the analyses of biological material, but did not accept the retrospective interview study. It claimed that the study «may give rise to renewed worries and self-reproach in parents who are already exposed to strong hardship». This concern could be set aside if the likelihood of beneficial results could be documented. It was also noted that «such a sensitive and difficult interview effort (will) hardly be suitable as a student project, since the parents obviously will need types of help and support that require advanced knowledge and experience». This was the first project to be rejected by the committee, and it is interesting to note that the committee’s secretary added «as a personal view» that the decision «does not imply any form of distrust in you as a person or in your academic qualities (...) Our decision is solely based on the sensitive and difficult topics involved. We therefore hope that you will come back in force, and that this little «setback» only may serve as an impetus for renewed efforts».
Conclusion
In posterity, it is interesting to note that in spite of the broad consensus on establishing regional committees of medical research ethics, unclear issues remained with regard to who was going to pay for what. This was the most important reason why the process dragged on for so long. It was not until the adoption of the Research Ethics Act in 2006 that the regional committees were provided with earmarked funding for their operations. Figure 2 shows some of the main events in the process.

It is also interesting to note that the form of organisational model that was originally designed in 1977 (Figure 1) is by and large identical to the one that came into place with the Health Research Act of 2009. The model had clarity of vision and was forward-looking. Even though we currently have no local ethics committees in the sense that was foreseen at the time, the Health Research Act points out that the research institutions have an independent responsibility for ensuring that research activities are organised and subject to quality assurance in an appropriate manner. This has spurred the establishment of local ethics committees, even though they have other names, such as department of research support, commission for research ethics, etc.
From a fairly modest and erratic start, the regional committees of research ethics are currently a professional organisation that enforces the Health Research Act and assesses approximately 1 800 new applications each year between seven committees with a total of 63 members and the same number of deputies, 26 employees in the secretariats and a budget of more than NOK 30 million. Erik Enger could hardly have imagined this in 1976, when the reported that the time had come (4).
Tabell
| Main message |
|
- 1.
Katz J. Experimentation with human beings: the authority of the investigator, subject, professions and state in the human experimentation process. New York, NY: Russel Sage, 1972.
- 2.
Shepherd W, Enger E. Helsinki-deklarasjonen. Tidsskr Nor Laegeforen 1976; 96: 469 – 70.
- 3.
Riksarkivet. Sosialdepartementet, Helsedirektoratet, Legekontoret, H1. Regionale komiteer for forskningsetikk 1970 – 1983.
- 4.
Enger E. Helsinki-deklarasjonen – revidert forskningsetikk. Tidsskr Nor Laegeforen 1976; 96: 467 – 71. [PubMed]
- 5.
Riksarkivet. NAVF. Rådet for medisinsk forskning. 63-arkiv. RMFS utvalg for forskningsetikk. 1974 – 1989.
- 6.
St.meld. nr. 35 (1975 – 76). Om forskningens organisering og finansiering.
- 7.
Brev fra Erik Enger til Egil Gjone 17.3.1982. REK sør-østs arkiv.
- 8.
Brev fra Jacob Jervell og Hans Erik Rugstad til direktøren for Rikshospitalet 4.11.1981. REK sør-østs arkiv.
- 9.
Brev fra Sosialdepartementet 20.1.1981. REK sør-østs arkiv.
- 10.
Hovedkomiteen for norsk forskning. Forskning og etisk ansvar. En rapport fra utvalget for «Forskning og etikk» nedsatt av Hovedkomiteen for norsk forskning. Oslo: NAVF, 1981.
- 11.
Forhandlinger i Stortinget 1982. Spørretime – 8. desember, s. 1886.
- 12.
Riksarkivet. Kultur og vitenskapsdepartementet, Forskningsavdelingen, Infrastruktur spørsmål 1981 – 1986. Utviklingen av medisinsk etikk i Norge 1975 – 81.
- 13.
Årsrapport 1988 for region 2. REK sør-østs arkiv.
- 14.
Riksarkivet. Kultur- og vitenskapsdepartementet, Forskningsavdelingen, Infrastruktur spørsmål 1987 – 1988. Forskningsetikk – regionale etiske forskningskomiteer.
- 15.
Brev fra Kultur- og vitenskapsdepartementet til Det medisinske fakultets sekretariat ved Universitetet i Oslo 7.5.1986. REK sør-østs arkiv.
- 16.
Dagsorden for REK sørs møte 11.12.1987. Referat fra REK østs møte 16.9.1987. REK sør-østs arkiv.
- 17.
Wetlesen J. Helsinki-deklarasjonen – misvisende norsk oversettelse? Tidsskr Nor Legeforen 1989; 109: 1179 – 80.