Several decades of effective cancer treatment have resulted in higher patient survival in Norway, but the treatment may entail a higher risk of infertility (1). Fertility preservation is therefore important for young cancer survivors, who all want to live a full life. Advances in reproductive medicine have given us a number of methods to preserve fertility, and these should always be considered alongside the cancer treatment itself. Several studies have demonstrated that failing to discuss the negative effects of cancer treatment can result in greater emotional stress and a poorer quality of life after remission (2). Patients who are to undergo cytotoxic treatment should be informed as early as possible about the risk of infertility and be referred to fertility centres that offer fertility preservation treatment if it is relevant. In Norway, all fertility treatment is regulated by the Biotechnology Act (3).
The purpose of this article is to provide an overview of infertility following successful cancer treatment in girls and young women and of current and future fertility-preserving methods for treating these patients.
Material and methods
The article is based on literature searches in the medical databases Medline, Pubmed and Scopus and the experience of the Nordic Network of Gonadal Preservation after Cancer Treatment in Children and Young Adults.
Fertility and effect of cancer treatment in women
Infertility is defined as the failure to achieve pregnancy after more than one year of regular sexual intercourse without contraception. Fertility in women presupposes normal anatomy and normal functioning of the sexual organs. Gonatropin-releasing hormone (GnRH) is secreted by the hypothalamus and stimulates secretion of follicle-stimulating hormone (FSH) and luteinising hormone (LH) from the hypophysis. In the ovaries, these gonadotropins stimulate follicle development and cause further maturation of the oocytes. Despite recent hypotheses to the effect that oocytes are also developed post-natally, the theory that a woman is born with a more or less fixed reservoir of immature oocytes still prevails (4). The reduction in the number of immature oocytes in the ovaries takes place through apoptosis, and proceeds both before and after birth. From puberty, the loss of oocytes is assumed to be between 500 and 1 000 in each menstrual cycle (5). Oocyte quality declines with increasing age, and is more pronounced from the age of about 35. By menopause the ovarian reserve is almost empty.
Women who have been treated for cancer become infertile after surgical operations, cytostatics treatment or radiation of the cervix, uterus or ovaries (table 1) (6). High-dose cytostatics treatment, particularly with alkylating cytostatics, and full body radiation as part of the conditioning regimen prior to haematopoietic stem cell transplantation may induce apoptosis in the follicles of the ovaries and result in a reduction of the oocyte reserve. The result may be premature menopause (menopause before the age of 40) (7). Radiation of the central nervous system with doses of ≥ 40 Gy may harm the hypothalamus and hypophysis and result in hypogonadotropic hypogonadism.
Table 1
Risk of amenorrhoea/premature ovarian failure after cancer treatment of girls and women
| Risk |
Treatment |
| High risk (≥ 80 %) |
Radiation treatment of whole abdomen or pelvis with ≥ 6 Gy in adult women, ≥ 15 Gy in prepubertal girls and > 10 Gy in post-pubertal girls. |
| Full body radiation |
|
| Alkylating chemotherapy (including cyclophosphamide ≥ 7.5 g/m² women < 20 years old |
|
| High-dose melphalan chemotherapy with stem cell support (HMAS) |
|
| Protocols for treating lymphoma that contain procarbazine: BEACOPP¹, cyclo-phosphamide, vincristine, procarbazine, prednisone-dacarbazine (COPP), nitrogen mustard, vincristine, procarbazine, prednisone (MOPP) |
|
| Radiation of the brain ≥ 40 Gy |
|
| Intermediate risk (30 – 70 %) |
Cyclophosphamide, epirubicin, fluorouracil (FEC): 30 – 39 years old |
| Radiation treatment of whole abdomen or pelvis at 10 – 15 Gy in prepubertal girls, 5 – 10 Gy in post-pubertal girls |
|
| Cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) |
|
| Cisplatin |
|
| Radiation of the central nervous system ≥ 25 Gy |
|
| Low risk (< 20 %) |
Doxorubicin and/or cyclophosphamide in women aged 30 – 39 |
| Cyclophosphamide, epirubicin, fluorouracil (FEC) in women aged < 30 |
|
| Doxorubicin, bleomycin, vinblastine, dacarbazine (ABVD) |
|
| Anthracyclines |
|
| Cytarabine |
|
| [i] | |
[i] ¹ BEACOPP = bleomycin, vincristine, cyclophosphamide, doxorubicin, etoposide, procarbazine, prednisone
Continued menstruation after cancer treatment is no guarantee that fertility has been preserved. The oocyte reserve may be severely reduced even if menstruation continues immediately after treatment. The oocyte reserve can be predicted by measuring the volume of the ovaries, number of antral follicles, FSH level, inhibin B, oestradiol and anti-Müller hormone (AMH). Follow-up studies of young cancer survivors show that the AMH level in the serum, as an indicator of the number of small pre-antral follicles in the ovaries, is not affected by the menstrual cycle or use of oral contraceptives and appears to be a better marker of the residual activity of the ovaries than the FSH or inhibin B-level (8).
Fertility-preserving treatment of children is still experimental, as the technical aspects are under development and there is less experience of these methods, also worldwide. This treatment should only be carried out at specialised clinics and with parental consent (9). Some serious benign diseases in children, such as aplastic anaemia, thalassaemia, sickle cell anaemia, Langerhans cell histiocytosis, haemophagocytic lymphohistiocytosis, Wegener’s granulomatosis and Turner’s syndrome may also cause infertility, due either to the disease itself or to gonadotoxic treatment of the underlying disease. Consequently, fertility-preserving measures may also be relevant with these diseases (10). Today this is an option only for patients who receive gonadotoxic treatment for the underlying disease, not for those whose fertility has been reduced by the underlying disease itself.
Figs 1 and 2 provide an overview of currently available fertility-preserving treatments for girls and women.
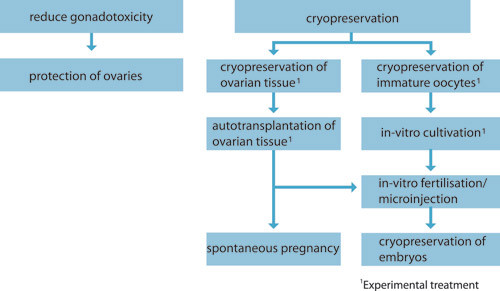
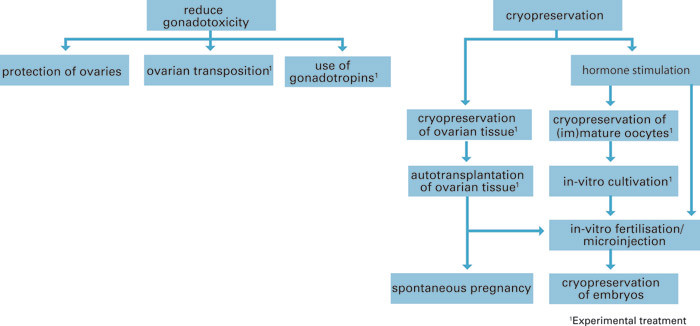
Embryo cryopreservation
In-vitro fertilisation (IVF) and intracytoplasmic sperm injection are the only established clinical methods of assisted fertilisation in Norway. These are only offered to women living in a stable relationship. After hormone stimulation, mature oocytes are harvested transvaginally and fertilised in the laboratory. Embryos can be cryopreserved for later thawing and placed back in the uterus when the patient has been pronounced recovered.
According to the Norwegian Biotechnology Act, cryopreserved embryos can be kept for up to five years. Cryopreservation of embryos implies 2 – 6 weeks’ postponement of cancer treatment, depending on the woman’s menstrual cycle. With the aid of a new type of stimulation protocol, initiated during the luteal phase, oocytes can be harvested over a two week period (11). The short time frame means that this can be done before the start of cancer treatment.
Cryopreservation of mature oocytes
Cryopreservation of unfertilised mature oocytes is an option for women who do not live in a stable partnership and for post-pubertal girls. Very poor survival rates have been achieved on thawing oocytes that have undergone cryopreservation by traditional freezing methods (slow freezing).
Ultra-rapid freezing, or vitrification, is a new method whereby the cells reach the «glass phase» without the harmful intracellular ice formation that takes place in slow freezing. The fertilisation rate of oocytes after vitrification is now comparable with that of fresh oocytes (12). However, the procedure entails hormone stimulation of the ovaries as in normal in-vitro fertilisation and means that cancer treatment must be postponed for at least two weeks. Cryopreservation of mature oocytes by means of vitrification has been performed in Sweden, inter alia, since 2005, but because of limited experience the method is not yet being used clinically in Norway.
Cryopreservation of immature oocytes
In the future, vitrification of immature oocytes may also be an option for women who cannot postpone cancer treatment, women without partners, hormone-receptor-positive breast cancer patients and prepubertal girls. The disadvantage is that relatively few oocytes are harvested and that it may be difficult to freeze immature oocytes for thawing and future maturation in the laboratory. The advantage is that it entails only mild or no hormone stimulation, without postponement of cancer treatment, as the oocytes can also be harvested in the luteal phase (13).
Cryopreservation and further cultivation of immature oocytes is still regarded as experimental, but international research shows promising results. The method is not used in Norway as yet.
Use of GnRH analogues.
GnRH analogues affect the hypothalumus-hypophysis-ovary axis, either by a sudden, massive but brief release of gonadotropins, or by down-regulating GnRH receptors and inhibiting the functioning of the ovary completely when used for a long period. The evidence accumulated so far is not sufficient to permit recommendation of GnRH analogues as a safe procedure for fertility-preserving treatment in cancer patients. More studies are necessary.
Ovary transposition
Transposition of the ovaries can be used separately from or concurrently with cryopreservation of oocytes or ovarian tissue. The ovaries are very sensitive to high dosage radiation treatment, but lower doses over a long period can also cause reduced fertility or premature menopause. Before radiation treatment of the pelvis, one or both ovaries can be moved out of the pelvis by means of laparoscopy or laparotomy (14). When the treatment is finished, the ovaries can be moved back. An increased occurrence of ovarian cysts and ischaemia has been documented with this procedure, and the ovaries often move back before radiation treatment is completed.
Ovarian transposition is not a treatment option in Norway today. However, all radiation treatment is individually adapted, and the patient’s gonads are protected from both direct and indirect radiation by means of lead shielding and precisely calculated effective doses.
Cryopreservation and reimplantation of ovarian tissue
Freezing of ovarian tissue from cancer patients with a view to later reimplantation has been carried out since 1996 (15). In Norway, the upper age limit for cryopreservation is 35, because of the natural reduction of the oocyte reserve. The probability of permanent infertility must be more than 50 %. Ovarian tissue is harvested by means of laparoscopy or laparotomy where either several ovarian tissue biopsies or an ovary are removed. After preparation, small pieces of the ovarian cortex are cryopreserved. After remission, pieces of tissue can be thawed and reimplanted in the remaining ovary or in a pocket created in the peritoneum. Hormone stimulation is not necessary, and planned cancer treatment can therefore start almost immediately. The treatment is also suitable for children and women without a partner (16).
Prepubertal girls are particularly susceptible to radiation damage of the uterus in the form of inhibition of normal growth and development, which can make it difficult to complete a future pregnancy. Before the start of radiation treatment of the pelvis, it is possible to mark the ovaries with clips during a laparascopy with concurrent removal of ovarian tissue. This makes it simpler to relocate and protect the ovaries during radiation treatment.
Autotransplantation of cryopreserved ovarian tissue is not suitable for patients with leukaemia, ovarian cancer or hereditary breast cancer (BRAC1-2) because of the increased risk of reimplanting malignant cells after remission (17). Some types of childhood cancer, such as neuroblastoma, Wilms’ tumour, osteosarcoma, Ewing’s sarcoma, lymphoma and extragenital rhabdomyosarcoma require special caution because metastasis to the ovaries cannot be excluded. This risk should therefore be assessed and discussed in advance.
Several studies are in progress to improve techniques for further cultivation of isolated, immature follicles from fresh or frozen ovarian tissue to mature oocytes in the final maturation phase, to prevent metastasis in patients' ovarian tissue (18). There are descriptions of freezing ovarian tissue from children down to the age of three, and so far this is the only fertility preservation method that can be offered to prepubertal girls.
Norwegian experiences
In Norway, only Oslo University Hospital offers cryopreservation of ovarian tissue following approval by the Directorate of Health. Since the method was first used in 2004 and up to 2011, ovarian tissue from 100 patients has been cryopreserved. Autotransplantation has not yet been carried out in Norway, but the application to do so has been approved by the Directorate of Health. There are several hundred patients worldwide with frozen ovarian tissue, but only a small percentage of them have requested reimplantation. There are reports from Denmark of cryopreservation of ovarian tissue from some 440 patients, reimplantation in 16 and three babies, two of them borne by the same woman (19). Fifteen live babies have been borne worldwide from ovarian tissue that has been frozen, thawed and reimplanted (Claus Y. Andersen, personal communication).
So far only three women have contacted the Oslo University Hospital for reimplantation of frozen ovarian tissue. The fact that fertility-preserving treatment is still regarded as experimental may frighten many women who have recently been diagnosed as having cancer. The patient must have been pronounced free of cancer for at least five years before reimplantation is a possibility. There is now increasing cooperation between Europe and the USA on fertility-preserving methods and the improvement of established and development of new methods.
Ethical and legal aspects
Fertility-preserving treatment is offered to girls and young women before the start of gonadotoxic chemotherapy and radiation therapy. The methods for preserving fertility are regarded as experimental, with the exception of cryopreservation of embryos. Practical, ethical and legal dilemmas should be resolved before treatment commences. The risk of damage as a result of postponing cancer treatment, invasive surgery, a weakened general condition and reimplantation of cancer cells after remission require thorough evaluation. The advantages of an intervention or an active decision not to intervene should be weighed against the short- and long-term disadvantages.
«Valid consent» means that consent must be given voluntarily by an informed, legally competent person. Legal competence means that the person giving consent must be capable of understanding the information (7). In Norway it is the parents who give consent for girls under the age of 18. A multidisciplinary approach is necessary to evaluate the practical and ethical aspects of fertility-preserving treatment. It is important to remember that this may improve the quality of life of young girls who are long-term survivors – a steadily growing group.
Conclusion
Girls and young women with recently diagnosed cancer have to deal with many questions and decisions in connection with their diagnosis. These include treatment and the side effects of treatment, including the risk of infertility. Developments in reproductive medicine and fertility-preserving treatment methods offer hope for many future cancer survivors who want to be able to have children. Multidisciplinary cooperation is necessary to permit the dissemination of up-to-date information and practical guidelines concerning the fertility of cancer patients and their families.
We should like to thank Gudvor Ertzeid, Nan B. Oldereid, Ritsa Storeng and Tom Tanbo for their thorough reading of the manuscript.
Tabell
| Main points |
|
- 1.
Fosså SD. Medisinske problemer hos kreftoverlevere. I: Fosså SD, Loge JH, Dahl AA, red. Kreftoverlevere. Oslo: Gyldendal, 2009: 58 – 77.
- 3.
Helse- og omsorgsdepartementet. Lov om humanmedisinsk bruk av bioteknologi (bioteknologiloven). LOV-2003 – 12 – 05 – 100. www.regjeringen.no (19.5.2011).
- 4.
Bukovsky A, Caudle MR, Svetlikova M et al. Oogenesis in adult mammals, including humans: a review. Endocrine 2005; 26: 301 – 16. [PubMed]
- 5.
Gougeon A. Regulation of ovarian follicular development in primates: facts and hypotheses. Endocr Rev 1996; 17: 121 – 55. [PubMed]
- 6.
LIVESTRONG. The Lance Armstrong Foundation. www.livestrong.org (19.5.2011).
- 10.
Rodriguez-Wallberg KA, Oktay K. Fertility preservation medicine: options for young adults and children with cancer. J Pediatr Hematol Oncol 2010; 32: 390 – 6. [PubMed]
- 14.
Bloemers MC, Portelance L, Legler C et al. Preservation of ovarian function by ovarian transposition prior to concurrent chemotherapy and pelvic radiation for cervical cancer. A case report and review of the literature. Eur J Gynaecol Oncol 2010; 31: 194 – 7.