A number of boys and men who contract cancer survive the disease and subsequently live full lives. Among children aged less than 18, leukaemia, lymphoma and tumours of the central nervous system are the most prevalent forms of cancer. In the age group 19 – 30 years, tumours in the male gonads account for about a quarter of all cases of cancer, followed by lymphoma, leukaemia and tumours of the central nervous system (Tini van Dijk, Cancer Register, personal communication). Some cancer treatments entail a higher risk of reduced fertility or of infertility, so that young men of fertile age risk not being able to father children when they have recovered. For some of these patients, fertility treatment may be a possibility of reducing this risk.
This article provides an overview of the risk of infertility in boys and young men after cancer treatment, and of current and potential fertility-preserving treatment methods. The objective is that doctors who are involved in following up these patients should be capable of providing helpful information to patient and family.
Material and methods
The article is based on literature searches in the medical databases Medline, Pubmed and Scopus and the experience of the Nordic Network of Gonadal Preservation after Cancer Treatment in Children and Young Adults.
Male fertility and effect of cancer treatment.
Infertility is defined as the failure to achieve pregnancy after more than one year of regular sexual intercourse without contraception. As is the case with women, fertility in men is contingent on a normal anatomy and normal functioning of the gonads. The testicles produce sperm and sex hormones, including testosterone, under the control of gonadotropins. Sperm production starts at puberty. Men produce sperm from stem cells throughout their adult lives in a continuous cycle that takes about 70 days (1).
Infertility in men with cancer may be caused by the disease itself and/or by surgery, chemotherapy and radiation treatment (table 1) (2). With testicular cancer and Hodgkin’s lymphoma, sperm quality may deteriorate and the sperm may suffer DNA damage even before the cancer treatment starts (3). Transient infertility induced by various types of cytostatics may last for several years after the completion of treatment. Permanent infertility occurs most commonly after high doses of cytostatics treatment (alkylating cytostatics), after radiation of the testicles with doses of > 1.2 Gy and after full body radiation prior to haematopoietic stem cell transplantation (4). Chemotherapy and radiation therapy affect both the sterol cells and the lending cells in the testicles, but the germinal epithelium is more prone to cell damage than the lending cells, so that infertility is a more frequent side effect than endocrine hypogonadism. Radiation treatment of the central nervous system with doses of ≥ 40 Gy can result in hypogonadotropic hypogonadism. Cancer treatment can damage the nerves and blood supply of the pelvis, which can cause problems with ejaculation and/or erection.
Table 1
Risk of azoospermia due to cancer treatment (2)
| Risk |
Treatment |
| High risk |
Full body radiation |
| Irradiation of testicles ≥ 2.5, Gy men, ≥ 6 Gy prepubertal boys |
|
| Alkylating chemotherapy (including cyclophosphamide ≥ 7.5 g/m² |
|
| High-dose melphalan chemotherapy with stem cell support (HMAS) |
|
| Protocols for treating lymphoma that contain procarbazine: BEACOPP¹, cyclophosphamide, vincristine, procarbazine, prednisone-dacarbazine (COPP), nitrogen mustard, vincristine, procarbazine, prednisone (MOPP) |
|
| Radiation of the brain ≥ 40 Gy |
|
| Intermediary risk |
Bleomycin, etoposide, cisplatin (BEP) with testicular cancer |
| Cisplatin |
|
| Cyclophosphamide, doxorubicin, vincristine, prednisone (CHOP) |
|
| Carboplatin < 2 g/m² |
|
| Low risk |
Doxorubicin, bleomycin, vinblastine, dacarbazine (ABVD) |
| Vincristine, etoposide, prednisone, doxorubicin, (OEPA) |
|
| Irradiation of testicles 0.2 – 0.7 Gy |
|
| [i] | |
[i] ¹ BEACOPP = bleomycin, vincristine, cyclophosphamide, doxorubicin, etoposide, procarbazine and prednisone
In some men, there has been a documented decline in normal testicular function after radiation treatment and various types of chemotherapy several years after treatment has finished, depending on the dose and type of treatment (Table 1). As with women, protection of the gonads during radiation treatment is therefore standard practice. With boys, some serious benign diseases, for example aplastic anaemia, thalassaemia, sickle cell anaemia, Langherhans cell histiocytosis, haemophagocytic lymphohistiocytosis, Wegener’s granulomatosis and Klinefelter’s syndrome, may also result in infertility due to the disease itself or to gonadotoxic treatment of the underlying disease. Fertility-preserving treatment may also be relevant for these patients, but at present it is only offered to those who are receiving gonadotoxic treatment for their underlying disease.
In Norway, all fertility treatment is regulated by the Biotechnology Act (5). Fertility preservation is an important topic for many young cancer survivors (6). Several studies have demonstrated that failing to discuss the negative effects of cancer treatment can result in greater emotional stress and a poorer quality of life after remission (7).
Fertility-preserving treatment should always be considered for boys and young men who are going to undergo cancer treatment (8) and patients must receive clear information about the possible side effects of the therapy, including fertility-related topics. Figures 1 and 2 provide an overview of fertility-preserving treatment for boys and young men.
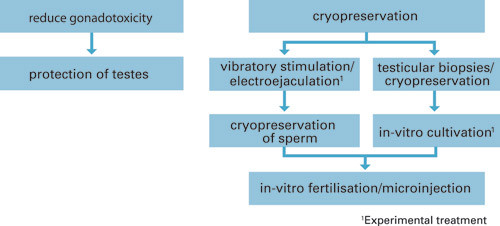
Cryopreservation of semen
There is a standard fertility-preserving option for men and post-pubertal boys. Cryopreservation of semen is today a well established procedure at five clinics in Norway (Tromsø, Trondheim, Bergen, Haugesund and Oslo). The doctor in charge of treatment should therefore discuss the possibility of cryopreservation of semen with the patient. This is a simple procedure and should always be recommended, also where cancer treatment has already been initiated.
Spermatogenesis in men takes about 70 days, and negative effects will probably not be observed before several weeks after the start of chemotherapy. However, a higher risk of DNA damage has been reported for sperm that has been ejaculated and stored after cytotoxic treatment has begun (9, 10), and long-term follow-up of the children of these patients is therefore necessary. Patients with testicular cancer and malignant lymphoma often have poorer sperm quality even before the start of the cancer treatment. If sperm are observed in the ejaculate, it is important to freeze them, as subsequent treatment using assisted fertilisation does not require many living sperm in order to achieve pregnancy. Major improvements in cryopreservation techniques and techniques for assisted fertilisation with intracytoplasmic sperm injection (ICSI) have resulted in successful pregnancies. Stored sperm appears to be viable for up to 28 years (11).
It may be difficult for many youngsters to produce a semen sample through masturbation. Testicle volume, not age, is directly correlated with sperm production (12). With Tanner’s puberty stages P2 – 3 and/or testicle volume > 6 ml, sperm production will very probably have started. In cases where the patient himself very much wants to cryopreserve his sperm, but fails with masturbation, vibratory stimulation of the penis is a possibility that should perhaps be considered more often, but always in collaboration with urologists and paediatricians. Electroejaculation may also be an alternative, but the procedure requires a general anaesthetic (13). The operation may be carried out concurrently with other procedures, such as the implantation of a central venous catheter before the start of chemotherapy, without postponing the start of treatment. This form of treatment is not used for cancer patients in Norway.
Cryopreservation of testicular biopsies
In the case of absence of sperm in the ejaculate (azoospermia) or very poor sperm quality, testicular biopsies can be frozen. Ideally, testicular tissue from prepubertal boys can be frozen before the start of cancer treatment and then thawed when the patient has recovered. The stored stem cells from the tissue samples are reimplanted in the patient’s own testicle, where they mature. The procedures are established in Sweden, but the Biotechnology Act places restrictions on the establishment of the method for cancer patients in Norway (14). From the tissue samples it is possible to isolate undifferentiated spermatogonia (stem cells) which lend themselves to further cultivation. Xenotransplantation or further cultivation of spermatogonia in the laboratory may be necessary in the case of patients with a higher risk of metastasis, but at present this treatment is not an option (15) – (17).
The Nordic countries are considering the possibility of centralising cryopreservation of testicular biopsies from the few prepubertal boys who are too young to produce sperm samples. Centralising this type of collaboration on the development of clinical treatment is a great advantage, as there are currently no fertility-preserving options for this patient group (18) (Petersen, C., Jahnukainen, K., Rechnitzer, C. et al. Nordic recommendations on fertility preservation in boys and young men. Abstract to the International Society of Paediatric Oncology, SIOP 2011).
Ethical and legal aspects
As with fertility-preserving options for girls and young women, most fertility-preserving methods for boys and young men, with the exception of cryopreservation of semen, are experimental. Practical, ethical and legal dilemmas should be resolved before treatment commences. Taking biopsies of the testicles is an invasive procedure, and the advantages and disadvantages must be weighed against each other.
A Belgian study describes an option for prepubertal boys of cryopreserving testicular tissue before the start of cancer treatment. Over 90 % of these patients and their parents accepted the offer and considered it a favourable option (19). A multidisciplinary approach is often necessary. The desire for a full life with retained fertility is very important to many young long-term survivors of cancer.
Conclusion
Cryopreservation of sperm is standard procedure for post-pubertal boys and adult men with cancer prior to cancer treatment. Cryopreservation of testicular biopsies and further cultivation or transplantation of male sex cells are still to be regarded as experimental methods. Extensive Nordic collaboration is in progress in this area.
We should like to thank Gudvor Ertzeid, Nan B. Oldereid, Ritsa Storeng and Tom Tanbo for their thorough reading of the manuscript.
Tabell
| Main points |
|
- 1.
Sharpe RM. Regulation of spermatogenesis. I: Knobil ENJD, red. The physiology of reproduction. 2. utg. New York: Raven Press, 1994: 1335 – 63.
- 2.
LIVESTRONG. The Lance Armstrong Foundation. www.livestrong.org (19.5.2011).
- 5.
Helse- og omsorgsdepartementet. Lov om humanmedisinsk bruk av bioteknologi (bioteknologiloven). LOV-2003-12-05-100. www.regjeringen.no (9.5.2011).
- 6.
Nieman CL, Kazer R, Brannigan RE et al. Cancer survivors and infertility: a review of a new problem and novel answers. J Support Oncol 2006; 4: 171 – 8. [PubMed]